
Journal of Inorganic Materials ›› 2026, Vol. 41 ›› Issue (4): 409-420.DOI: 10.15541/jim20250307
• REVIEW • Next Articles
PENG Dezhao1,2( ), LI Rui1,2, WANG Wenhong1,2, WANG Zirui1,2, ZHANG Zhizhen1,2(
), LI Rui1,2, WANG Wenhong1,2, WANG Zirui1,2, ZHANG Zhizhen1,2( )
)
Received:2025-07-19
Revised:2025-09-17
Published:2026-04-20
Online:2025-10-17
Contact:
ZHANG Zhizhen, associate professor. E-mail: zhangzhzh28@mail.sysu.edu.cnAbout author:PENG Dezhao (1999-), male, PhD candidate. E-mail: pengdzh5@mail2.sysu.edu.cn
Supported by:CLC Number:
PENG Dezhao, LI Rui, WANG Wenhong, WANG Zirui, ZHANG Zhizhen. Research Progress on Sodium Chloride Solid Electrolytes[J]. Journal of Inorganic Materials, 2026, 41(4): 409-420.
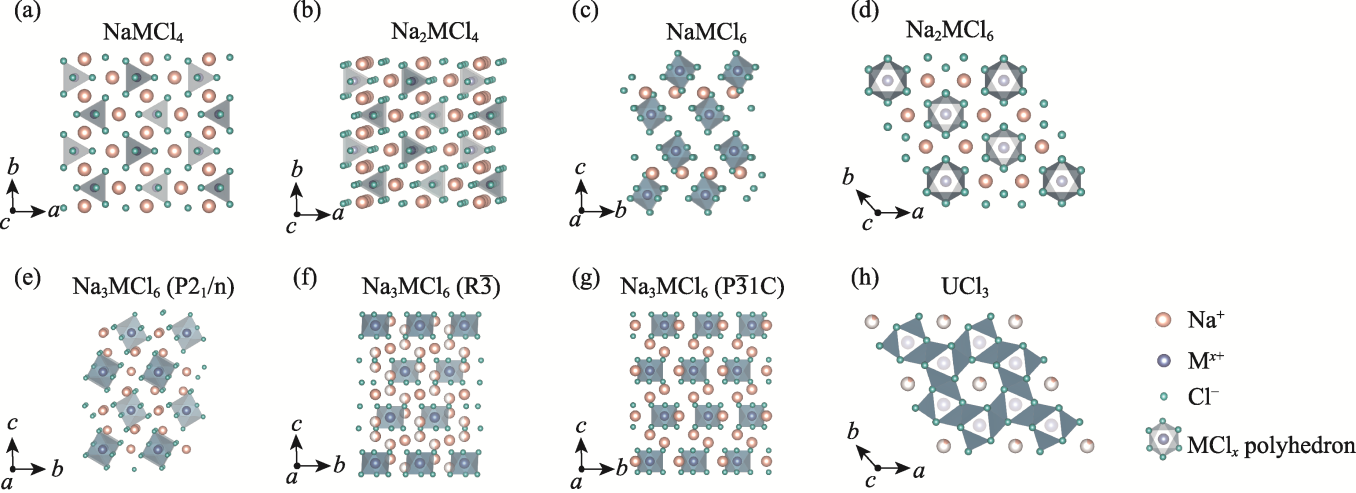
Fig. 2 Crystal structures of different sodium chloride solid electrolytes (a) NaMCl4; (b) Na2MCl4; (c) NaMCl6; (d) Na2MCl6; (e) Na3MCl6 (P21/n); (f) Na3MCl6 ($\text{R}\overline{3}$); (g) Na3MCl6 ($\text{P}\overline{3}1\text{c}$); (h) UCl3
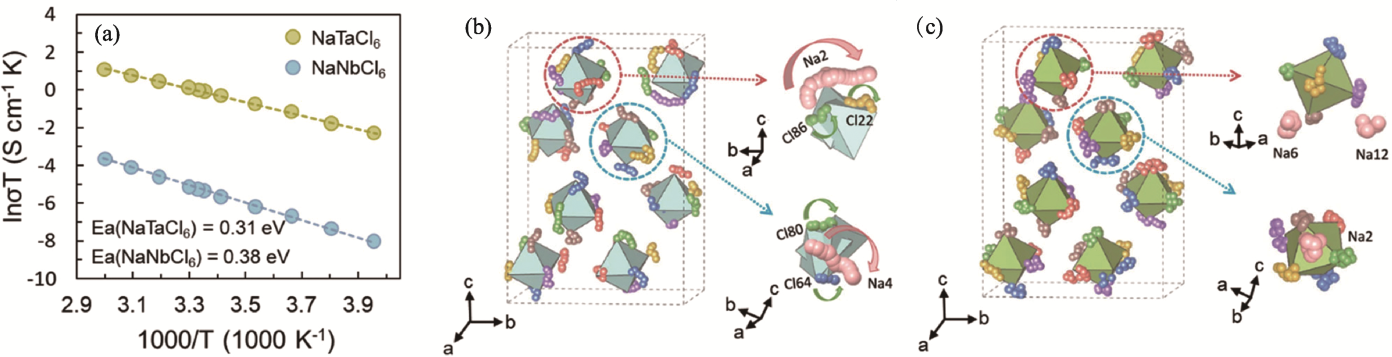
Fig. 3 Ion transport properties of NaMCl6 solid electrolytes[52] (a) Arrhenius plots of NaTaCl6 and NaNbCl6; (b, c) Atomic trajectories of Na and Cl atoms from ab initio molecular dynamics (AIMD) simulations for (b) NaTaCl6 and (c) NaNbCl6
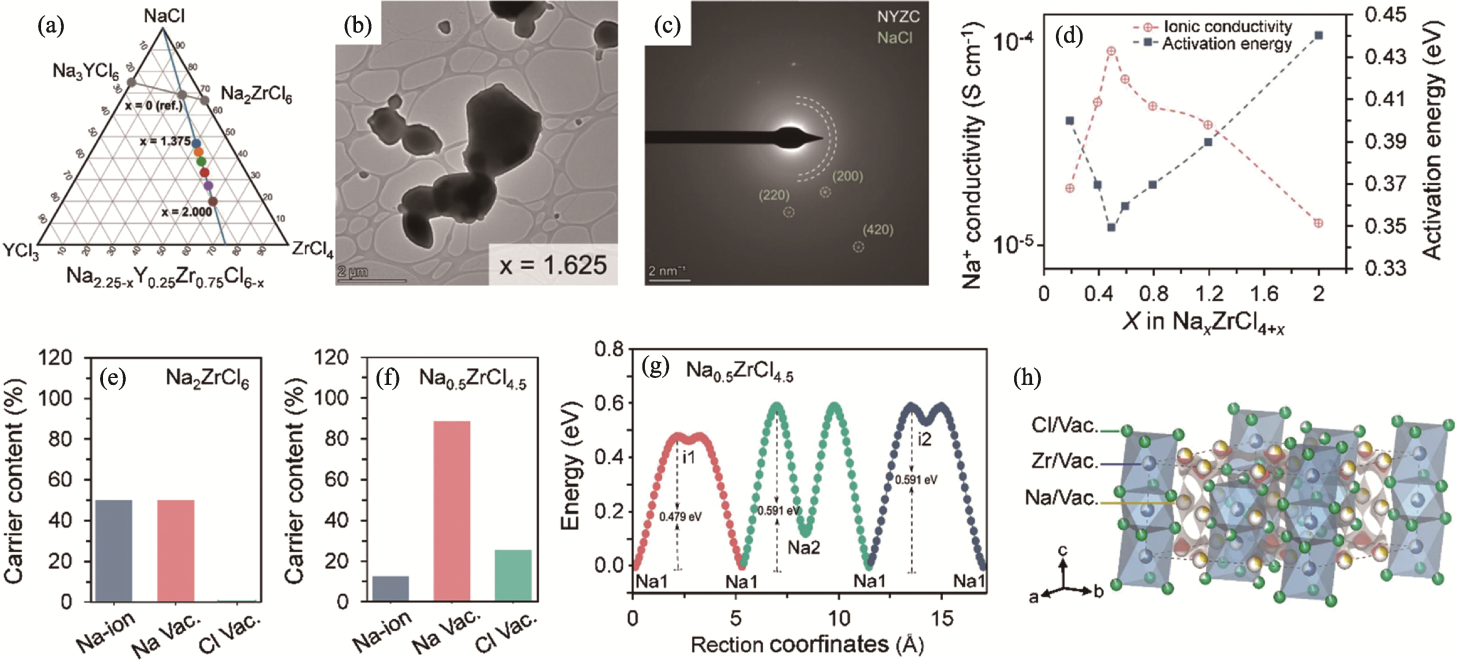
Fig. 4 Phase diagram, ion transport properties and structures of Na3-xMCl6-x/Na2-xMCl6-x solid electrolytes[36,59] (a) Phase diagram of NaCl-YCl3-ZrCl4[59]; (b) Transmission electron microscope image of Na0.625Y0.25Zr0.75Cl4.375[59]; (c) Selected area electron diffraction pattern of Na0.625Y0.25Zr0.75Cl4.375[59]; (d) Ionic conductivity and activation energy of NaxZrCl4+x at 25 ℃[36]; (e, f) Na+ and vacancy contents in (e) Na2ZrCl6 and (f) Na0.5ZrCl4.5[36]; (g) Energy profiles along the Na+ transport pathways in Na0.5ZrCl4.5[36]; (h) Crystal structure of Na0.5ZrCl4.5[36]
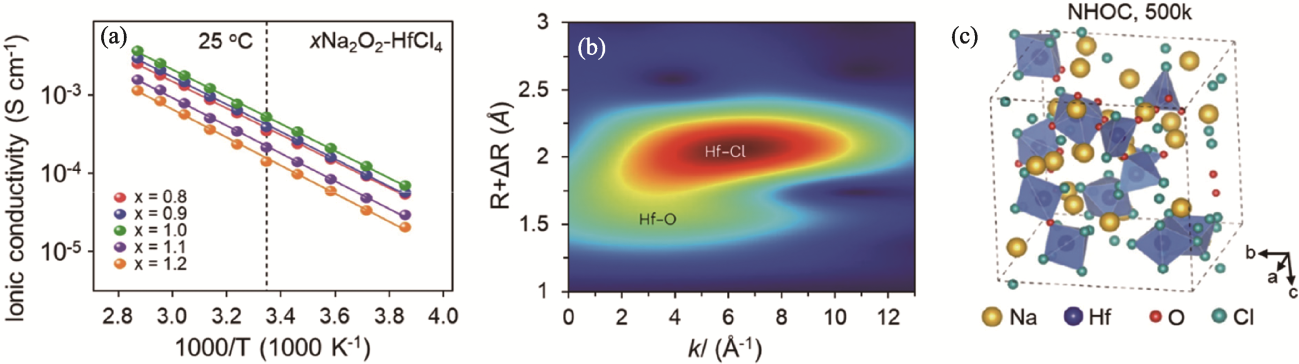
Fig. 5 Ion transport property and structure of xNa2O2-HfCl4 solid electrolytes[39] (a) Arrhenius plots of xNa2O2-HfCl4; (b) Wavelet-transformed EXAFS contour plots of Na2O2-HfCl4 at Hf L3-edge[39];(c) AIMD-generated structure of amorphous Na2O2-HfCl4 at 500 K[39]

Fig. 6 Ion transport property and structure of xUCl3-(1-x)NaTaCl6 solid electrolytes[69] (a) Arrhenius plots of 0.62Na0.75Sm1.75Cl6-0.38NaTaCl6 and 0.57Na0.75La1.75Cl6-0.43NaTaCl6; (b, c) Transmission electron microscope images of 0.62Na0.75Sm1.75Cl6-0.38NaTaCl6
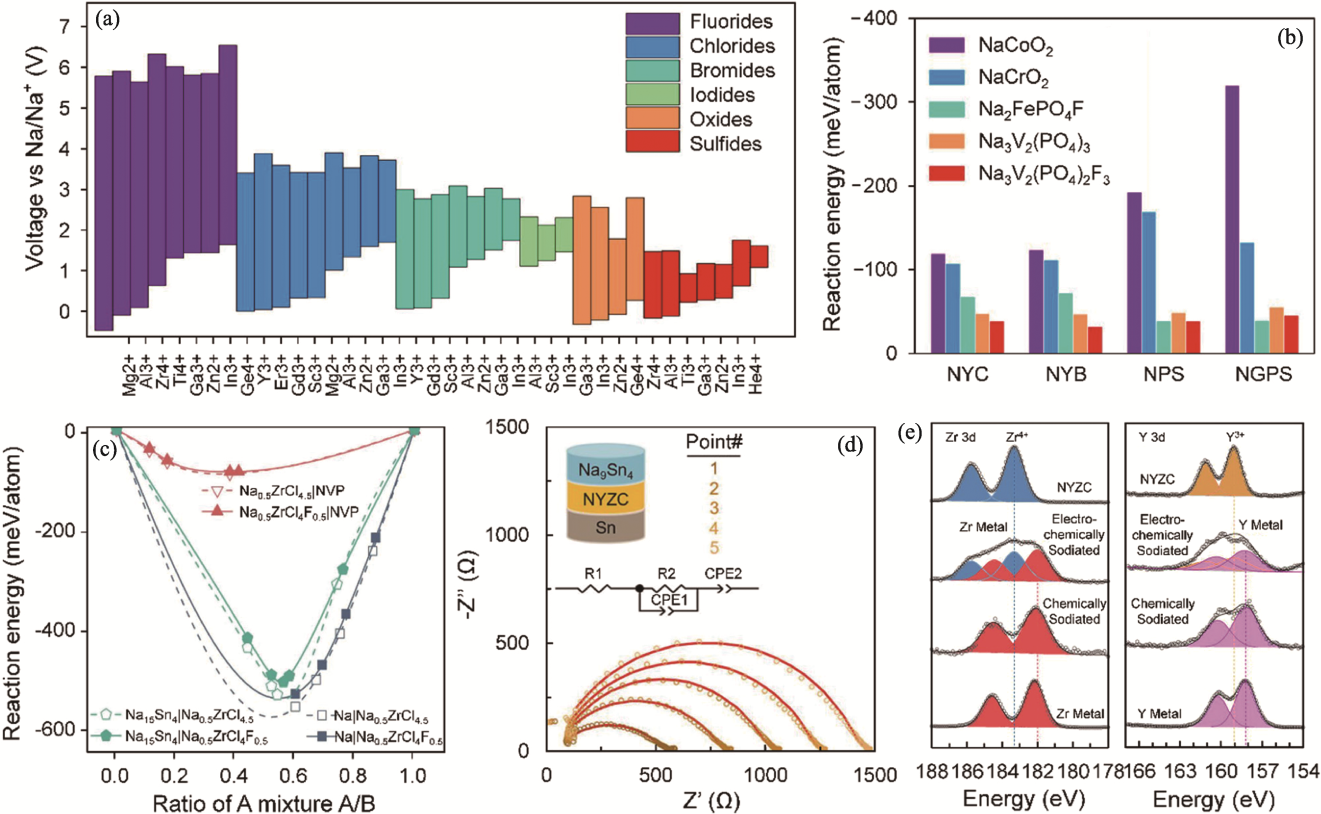
Fig. 7 Electrochemical stability of sodium chloride solid electrolytes and their compatibilities with electrode materials[36,71,74] (a) Calculated electrochemical stability windows of Na-M-X ternary compounds, including fluorides, chlorides, bromides, iodides, oxides, and sulfides[71]; (b) Calculated reaction energies between different cathodes and solid electrolytes for Na3YCl6 (NYC), Na3YBr6 (NYB), Na3PS4 (NPS), and Na10GeP2S12 (NGPS)[71]; (c) Calculated reaction energies between Na0.5ZrCl4.5, Na0.5ZrCl4F0.5 solid electrolytes and Na3V2(PO4)3 (NVP) cathode, Na anode and Na15Sn4 anode[36]; (d) Impedance evolution of Na9Sn4|Na2.25Y0.25Zr0.75Cl6|Na cells with the time increasing[74]; (e) Zr3d and Y3d XPS spectra of Na2.25Y0.25Zr0.75Cl6 obtained from Na9Sn4|Na2.25Y0.25Zr0.75Cl6|Na cells after charging and discharging[74]
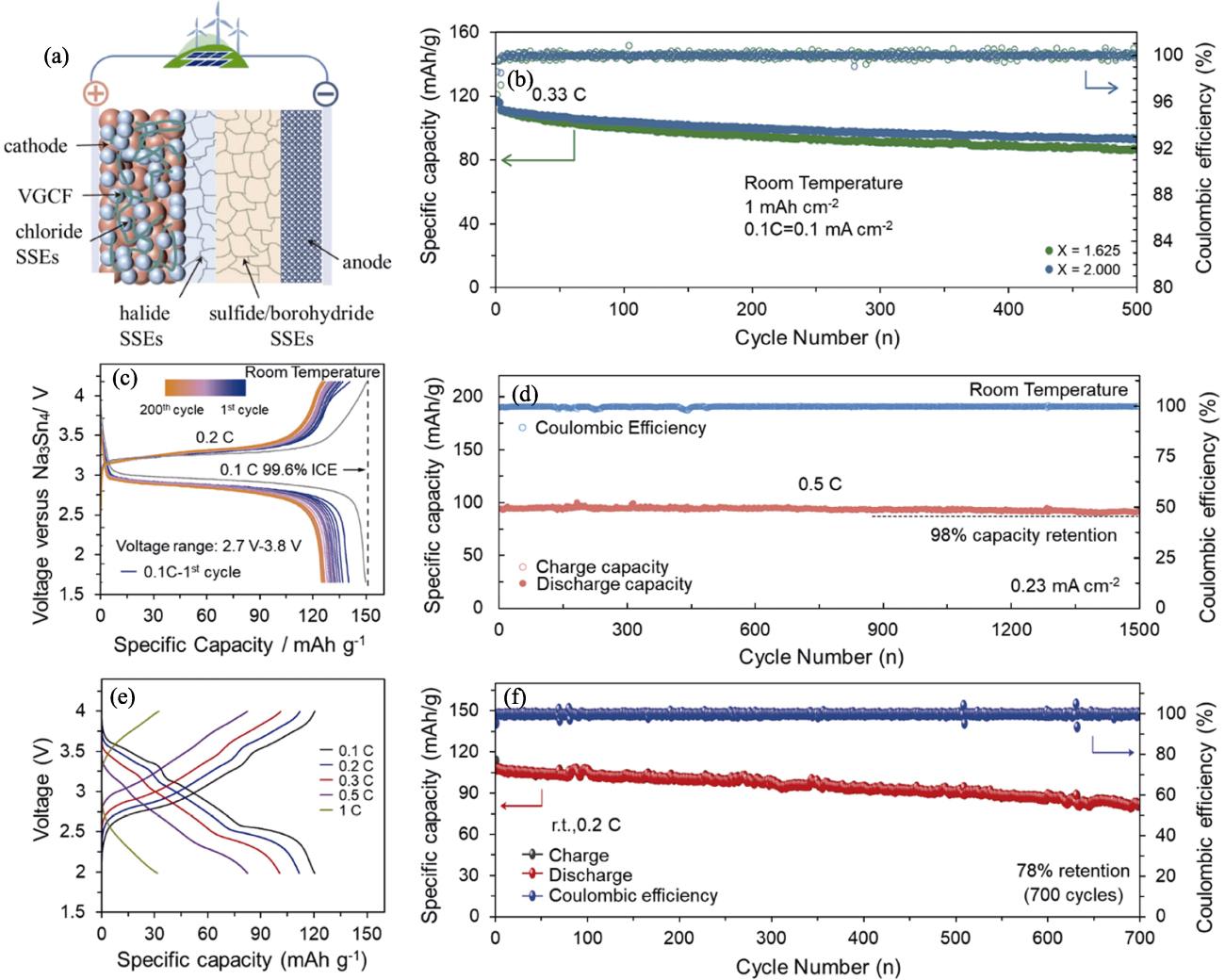
Fig. 8 Electrochemical performance of chloride all-solid-state sodium-ion batteries[36-37,39,59] (a) Schematic illustration of halide-based all-solid-state battery[36]; (b) Long cycling profiles of NaCrO2|Na2.25Y0.25Zr0.75Cl6|Na2(B10H10)0.5(B12H12)0.5|Na9Sn4 cells[59]; (c) Charge-discharge curves of Na3V2(PO4)3|NaTaCl6|Na3PS4|Na15Sn4 cells during cycling[37];(d) Long cycling profiles of Na3V2(PO4)3|NaTaCl6|Na3PS4|Na15Sn4 cells[37]; (e) Charge-discharge curves of Na0.85Mn0.5Ni0.4Fe0.1O2|Na2O2-HfCl4|Na3PS4|Na15Sn4 cells with different rate currents[39] and corresponding (f) long cycling profiles[39]
| [1] |
GOODENOUGH J B. Evolution of strategies for modern rechargeable batteries. Acc. Chem. Res., 2013, 46(5): 1053.
DOI URL |
| [2] |
GOODENOUGH J B, KIM Y. Challenges for rechargeable Li batteries. Chem. Mater., 2009, 22(3): 587.
DOI URL |
| [3] | XU J, CAI X, CAI S, et al. High-energy lithium-ion batteries: recent progress and a promising future in applications. Energy Environ. Mater., 2023, 6(5): 35. |
| [4] |
GAO Y L, PAN Z H, SUN J G, et al. High-energy batteries: beyond lithium-ion and their long road to commercialisation. Nano-Micro Lett., 2022, 14(1): 94.
DOI PMID |
| [5] | VAALMA C, BUCHHOLZ D, WEIL M, et al. A cost and resource analysis of sodium-ion batteries. Nat. Rev. Mater., 2018, 3(4): 3443. |
| [6] |
KUNDU D, TALAIE E, DUFFORT V, et al. The emerging chemistry of sodium Ion batteries for electrochemical energy storage. Angew. Chem. Int. Ed., 2015, 54(11): 3431.
DOI PMID |
| [7] |
HAYASHI A, NOI K, SAKUDA A, et al. Superionic glass-ceramic electrolytes for room-temperature rechargeable sodium batteries. Nat. Commun., 2012, 3: 856.
DOI |
| [8] |
CHE H, CHEN S, XIE Y, et al. Electrolyte design strategies and research progress for room-temperature sodium-ion batteries. Energy Environ. Sci., 2017, 10(5): 1075.
DOI URL |
| [9] |
SUN B, XIONG P, MAITRA U, et al. Design strategies to enable the efficient use of sodium metal anodes in high-energy batteries. Adv. Mater., 2019, 32(18): 1903891.
DOI URL |
| [10] |
ZHANG X, LI L, FAN E, et al. Toward sustainable and systematic recycling of spent rechargeable batteries. Chem. Soc. Rev., 2018, 47(19): 7239.
DOI PMID |
| [11] |
YAO X, HUANG B, YIN J, et al. All-solid-state lithium batteries with inorganic solid electrolytes: review of fundamental science. Chin. Phys. B, 2016, 25(1): 018802.
DOI URL |
| [12] |
HIRSH H S, LI Y, TAN D H S, et al. Sodium-ion batteries paving the way for grid energy storage. Adv. Energy Mater., 2020, 10(32): 2001274.
DOI URL |
| [13] |
SAND S C, RUPP J L M, YILDIZ B. A critical review on Li-ion transport, chemistry and structure of ceramic-polymer composite electrolytes for solid state batteries. Chem. Soc. Rev., 2025, 54(1): 178.
DOI PMID |
| [14] |
JANEK J, ZEIER W G. A solid future for battery development. Nat. Energy, 2016, 1: 24.
DOI |
| [15] |
MANTHIRAM A, YU X W, WANG S F. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater., 2017, 2(4): 16103.
DOI |
| [16] |
CHEN R, LI Q, YU X, et al. Approaching practically accessible solid-state batteries: stability issues related to solid electrolytes and interfaces. Chem. Rev., 2020, 120(14): 6820.
DOI PMID |
| [17] |
YU T, YANG X, YANG R, et al. Progress and perspectives on typical inorganic solid-state electrolytes. J. Alloys Compd., 2021, 885: 161013.
DOI URL |
| [18] |
LU P, XIA Y, SUN G, et al. Realizing long-cycling all-solid-state Li-In||TiS2 batteries using Li6+xMxAs1-xS5I (M=Si, Sn) sulfide solid electrolytes. Nat. Commun., 2023, 14: 4077.
DOI |
| [19] |
KATO Y, HORI S, SAITO T, et al. High-power all-solid-state batteries using sulfide superionic conductors. Nat. Energy., 2016, 1(4): 16030.
DOI |
| [20] | NIE X, HU J, LI C. Halide-based solid electrolytes: the history, progress, and challenges. Interdiscip. Mater., 2023, 2(3): 365. |
| [21] | WANG C, LIANG J, KIM J T, et al. Prospects of halide-based all-solid-state batteries: from material design to practical application. Sci. Adv., 2022, 8(36): eadc9516. |
| [22] |
WANG Q, ZHOU Y, WANG X, et al. Designing lithium halide solid electrolytes. Nat. Commun., 2024, 15(1): 1050.
DOI PMID |
| [23] | ASANO T, SAKAI A, OUCHI S, et al. Solid halide electrolytes with high lithium-ion conductivity for application in 4 V class bulk-type all-solid-state batteries. Adv. Mater., 2018, 30(44): e1803075. |
| [24] | WEI Z, NAZAR L F, JANEK J. Emerging halide solid electrolytes for sodium solid-state batteries: structure, conductivity, paradigm of applications. Batteries Supercaps, 2024, 7(7): e202400005. |
| [25] |
LI X N, LIANG J W, YANG X F, et al. Progress and perspectives on halide lithium conductors for all-solid-state lithium batteries. Energy. Environ. Sci., 2020, 13(5): 1429.
DOI URL |
| [26] |
TUO K, SUN C W, LIU S Q. Recent progress in and perspectives on emerging halide superionicconductors for all-solid-state batteries. Electrochem. Energy Rev., 2023, 6(1): 17.
DOI |
| [27] | HUANG L J, ZHANG L, BI J Y, et al. An insight into halide solid-state electrolytes: progress and modification strategies. Energy Mater. Adv., 2024, 5: 35632. |
| [28] |
LIANG J, LI X, ADAIR K R, et al. Metal halide superionic conductors for all-solid-state batteries. Acc. Chem. Res., 2021, 54(4): 1023.
DOI URL |
| [29] |
KWAK H, WANG S, PARK J, et al. Emerging halide superionic conductors for all-solid-state batteries: design, synthesis, and practical applications. ACS Energy Lett., 2022, 7(5): 1776.
DOI URL |
| [30] | KWAK H, LYOO J, PARK J, et al. Na2ZrCl6 enabling highly stable 3 V all-solid-state Na-ion batteries. Energy Storage Mater., 2023, 37: 47. |
| [31] |
ZHAO T, SOBOLEV A N, SCHLEM R, et al. Synthesis-controlled cation solubility in solid sodium ion conductors Na2+xZr1-xInxCl6. ACS Appl. Energy Mater., 2023, 6(8): 4334.
DOI URL |
| [32] |
SEBTI E, QI J, RICHARDSON P M, et al. Synthetic control of structure and conduction properties in Na-Y-Zr-Cl solid electrolytes. J. Mater. Chem. A, 2022, 10(40): 21565.
DOI URL |
| [33] | STEINER H J, LUTZ H D. Neue schnelle ionenleiter vom Typ MⅠ3MⅢCl6 (MⅠ=Li, Na, Ag; MⅢ=In, Y). Z. Anorg. Allg. Chem., 1992, 613(7): 26. |
| [34] |
SCHLEM R, BANIK A, ECKARDT M, et al. Na3-xEr1-xZrxCl6--a halide-based fast sodium-ion conductor with vacancy-driven ionic transpor. ACS Appl. Energy Mater., 2020, 3(10): 10164.
DOI URL |
| [35] |
WU E A, BANERJEE S, TANG H, et al. A stable cathode-solid electrolyte composite for high-voltage, long-cycle-life solid-state sodium-ion batteries. Nat. Commun., 2021, 12: 1256.
DOI PMID |
| [36] | WU M, LIU X, LIU H, et al. Fluorinated amorphous halides with improved ionic conduction and stability for all-solid-state sodium-ion batteries. Nat. Commun., 2025, 16: 2808. |
| [37] |
HU Y, FU J, XU J, et al. Superionic amorphous NaTaCl6 halide electrolyte for highly reversible all-solid-state Na-ion batteries. Matter, 2024, 7(3): 1018.
DOI URL |
| [38] | ZHAO T, SAMANTA B, DE IRUJO-LABALDE X M, et al. Sodium metal oxyhalides NaMOCl4 (M=Nb, Ta) with high ionic conductivities. ACS Mater. Lett., 2024, 6(8): 3683. |
| [39] |
LIN X, ZHANG S, YANG M, et al. A family of dual-anion-based sodium superionic conductors for all-solid-state sodium-ion batteries. Nat. Mater., 2024, 24(1): 83.
DOI PMID |
| [40] | LIN X, ZHAO Y, WANG C, et al. A dual anion chemistry-based superionic glass enabling long-cycling all-solid-state sodium-ion batteries. Angew. Chem. Int. Ed., 2023, 13: e202314181. |
| [41] |
KWAK H, KIM J S, HAN D, et al. Boosting the interfacial superionic conduction of halide solid electrolytes for all-solid-state batteries. Nat. Commun., 2023, 14: 2459.
DOI PMID |
| [42] |
BAENZIGER N C. The crystal structure of NaAlCl4. Acta Cryst., 1951, 4(3): 216.
DOI URL |
| [43] |
PARK J, SON J P, KO W, et al. NaAlCl4: new halide solid electrolyte for 3 V stable cost-effective all-solid-state Na-ion batteries. ACS Energy Lett., 2022, 7(10): 3293.
DOI URL |
| [44] |
HAFNER M, BIANCHINI M. Exploring cationic substitutions in the solid electrolyte NaAlCl4 with density functional theory. J. Phys. Chem. C, 2024, 128(47): 19978.
DOI URL |
| [45] | GOODYEAR J, ALI S A D, STEIGMANN G A. The crystal structure of Na2MnCl4. Acta Cryst., 1971, B27(8): 1672. |
| [46] | VAN LOON C J J, IJDO D J W. The crystal structure of Na6MnCl8 and Na2Mn3Cl8 and some isostructural compounds. Acta Cryst., 1975, B31(3): 770. |
| [47] | VAN LOON C J J, VISSER D. Chlorides with the chrysoberyl structure: Na2CoCl4 and Na2ZnCl4. Acta Cryst., 1977, B33(1): 188. |
| [48] | LUTZ H D, WUSSOW K, KUSKE P. Ionic conductivity, structural, IR and Raman spectroscopic data of olivine, Sr2PbO4, and Na2CuF4 type lithium and sodium chlorides Li2ZnCl4 and Na2MCl4 (M = Mg, Ti, Cr, Mn, Co, Zn, Cd). Z Naturforsch., 1987, 42b(11): 1379. |
| [49] | GUO H, HAFNER M, GRUNINGER H, et al. Structure and ionic conductivity of halide solid electrolytes based on NaAlCl4 and Na2ZnCl4. Adv. Sci., 2025, 12(30): e07224. |
| [50] |
SADOWAY D R, FLENGAS S N. The synthesis and properties of the hexachloroniobates and hexachlorotantalates of Na, K, Rb, and Cs. Can. J. Chem., 1978, 56(15): 2013.
DOI URL |
| [51] | HENKE H. Zur kristallchemischen einordnung von NaSbCl6, NaNbCl6 und NaTaCl6. Z. Krist.-Cryst. Mater., 1992, 198(1/2): 1. |
| [52] |
LI R, XU K Q, WEN S H, et al. A sodium superionic chloride electrolyte driven by paddle wheel mechanism for solid state batteries. Nat. Commun., 2025, 16: 6633.
DOI |
| [53] |
KIPOUROS G J, FLENGAS S N. Equilibrium decomposition pressures of the compounds Na2ZrCl6 and Na2HfCl6. Can. J. Chem., 1981, 59(6): 990.
DOI URL |
| [54] |
ZHAO T, SOBOLEV A N, DE IRUJO LABALDE X M, et al. On the influence of the coherence length on the ionic conductivity in mechanochemically synthesized sodium-conducting halides, Na3-xIn1-xZrxCl6. J. Mater. Chem. A, 2024, 12(12): 7015.
DOI URL |
| [55] |
ZHOU L, ZHANG S, LI W, et al. Amorphous-nanocrystalline fluorinated halide electrolytes with high ionic conductivity and high-voltage stability. J. Am. Chem. Soc., 2025, 147(18): 15136.
DOI URL |
| [56] |
PARK D, KIM K, CHUN G H, et al. Materials design of sodium chloride solid electrolytes Na3MCl6 for all-solid-state sodium-ion batteries. J. Mater. Chem. A, 2021, 9(40): 23037.
DOI URL |
| [57] |
YU S, KIM K, WOOD B C, et al. Structural design strategies for superionic sodium halide solid electrolytes. J. Mater. Chem. A, 2022, 10(45): 24301.
DOI URL |
| [58] |
NIU X, DOU X, FU C, et al. Sodium halide solid state electrolyte of Na3YBr6 with low activation energy. RSC Adv., 2024, 14(21): 14716.
DOI URL |
| [59] |
RIDLEY P, NGUYEN L H B, SEBTI E, et al. Amorphous and nanocrystalline halide solid electrolytes with enhanced sodium-ion conductivity. Matter, 2024, 7(2): 485.
DOI URL |
| [60] | LUO J D, ZHANG Y, CHENG X, et al. Halide superionic conductors with non-close-packed anion frameworks. Angew. Chem. Int. Ed., 2024, 63(17): e202400424. |
| [61] |
LISSNER F, KRÄMER K, SCHLEID T, et al. Die chloride Na3xM2-xCl6 (M=La, Sm) and NaM2Cl6 (M=Nd, Sm): derivate des UCl3-Typs. Synthese, kristallstruktur und röntgenabsorptionsspektroskopie (XANES). Z. Anorg. Allg. Chem., 1994, 620(3): 444.
DOI URL |
| [62] |
WICKLEDER M S, MEYER G. Neue derivate des UCl3-typs: die chloride and bromide A(SrSm)Cl6, A(SrEu)Cl6 und A(BaLa)X6 (A=Na, Ag; X=Cl, Br). Z. Anorg. Allg. Chem., 1999, 624(10): 1577.
DOI URL |
| [63] |
FU C, LI Y, XU W, et al. LaCl3-based sodium halide solid electrolytes with high ionic conductivity for all-solid-state batteries. Nat. Commun., 2024, 15: 5646.
DOI |
| [64] |
DAI T, WU S Y, LU Y X, et al. Inorganic glass electrolytes with polymer-like viscoelasticity. Nat. Energy, 2023, 8(11): 1221.
DOI |
| [65] |
MOTOHASHI K, TSUKASAKI H, MORI S, et al. Fast sodium-ion conducting amorphous oxychloride embedding nanoparticles. Chem. Mater., 2024, 36(19): 9914.
DOI URL |
| [66] |
ZHOU L, BAZAK J D, LI C, et al. 4 V Na solid state batteries enabled by a scalable sodium metal oxyhalide solid electrolyte. ACS Energy Lett., 2024, 9(8): 4093.
DOI URL |
| [67] |
LIANG C C. Conduction characteristics of the lithium iodide-aluminum oxide solid electrolytes. J. Electrochem. Soc., 1973, 120(10): 1289.
DOI URL |
| [68] |
MERCIER R, MALUGANI J P, FAHYS B, et al. Superionic conduction in Li2S-P2S5-LiI-glasses. Solid State Ionics, 1981, 5: 663.
DOI URL |
| [69] | FU J, WANG S, WU D, et al. Halide heterogeneous structure boosting ionic diffusion and high-voltage stability of sodium superionic conductors. Adv. Mater., 2024, 36(3): e2308012. |
| [70] | YU Q, HU J, XU Y, et al. Mesoporous enhanced heterostructured halide solid electrolytes with high air stability and high abundance for sustainable sodium metal batteries. Angew. Chem. Int. Ed., 2025, 64(26): e202425503. |
| [71] |
QIE Y, WANG S, FU S, et al. Yttrium-sodium halides as promising solid-state electrolytes with high ionic conductivity and stability for Na-ion batteries. J. Phys. Chem. Lett., 2020, 11(9): 3376.
DOI PMID |
| [72] |
PARK J, HAN D, SON J P, et al. Extending the electrochemical window of Na+ halide nanocomposite solid electrolytes for 5 V- class all-solid-state Na-ion batteries. ACS Energy Lett., 2024, 9(5): 2222.
DOI URL |
| [73] |
GOODWIN L E, ZIEGLER M, TILL P, et al. Halide and sulfide electrolytes in cathode composites for sodium all-solid-state batteries and their stability. ACS Appl. Mater. Interfaces, 2024, 16(15): 19792.
DOI URL |
| [74] |
DEYSHER G, CHEN Y T, SAYAHPOUR B, et al. Evaluating electrolyte-anode interface stability in sodium all-solid-state batteries. ACS Appl. Mater. Interfaces, 2022, 14(42): 47706.
DOI URL |
| [75] |
ZHANG J, YU Z, ZHU Y, et al. Configuration design and interface reconstruction to realize the superior high-rate performance for sodium layered oxide cathodes. Adv. Energy Mater., 2025, 15(23): 2405951.
DOI URL |
| [76] |
ZHANG Z, ZHANG Q, SHI J, et al. A self-forming composite electrolyte for solid-state sodium battery with ultralong cycle life. Adv. Energy Mater., 2017, 7(4): 1601196.
DOI URL |
| [77] | DONG Z L, GAN Y, MARTINS V, et al. Novel sulfide-chloride solid-state electrolytes with tunable anion ratio for highly stable solid-state sodium-ion batteries. Adv. Mater., 2025, 37(30): e2503107. |
| [78] |
XU J, LI Y, LU P, et al. Water-stable sulfide solid electrolyte membranes directly applicable in all-solid-state batteries enabled by superhydrophobic Li+-conducting protection layer. Adv. Energy Mater., 2021, 12(2): 2102348.
DOI URL |
| [1] | LI Xuan, YE Kuicai, FENG Jiayin, QIU Jiajun, QIAN Wenhao, XING Min. Surface Modification of Titanium-based Dental Implants for Soft Tissue Sealing: A Review [J]. Journal of Inorganic Materials, 2026, 41(4): 432-444. |
| [2] | CHEN Kun, JIANG Yonggang, FENG Junzong, LI Liangjun, HU Yijie, FENG Jian. Research Progress on Lanthanum Zirconate Porous Materials for Thermal Insulation [J]. Journal of Inorganic Materials, 2026, 41(4): 421-431. |
| [3] | WEI Lianjin, QI Zhijie, WANG Xin, ZHU Junwu, FU Yongsheng. Modification of Nanodiamond and Its Application in Electrocatalytic Oxygen Reduction Reaction [J]. Journal of Inorganic Materials, 2026, 41(3): 273-288. |
| [4] | LIU Zhanyi, LI Mian, OUYANG Xiaoping, CHAI Zhifang, HUANG Qing. Recent Progress on Removal of Sr/Cs from Molten Salt in Dry Reprocessing [J]. Journal of Inorganic Materials, 2026, 41(2): 150-158. |
| [5] | SUN Lian, ZHANG Leilei, XUE Zexu, WU Kun, CHEN Ye, LI Zhiyuan, WANG Lukai, WANG Zungang. Research Progress on Zero-dimensional Metal Halide Scintillators towards Radiation Detection Applications [J]. Journal of Inorganic Materials, 2026, 41(2): 159-176. |
| [6] | REN Xianpei, LI Chao, HU Qiwei, XIANG Hui, PENG Yuehong. Research Progress on Mott-Schottky Hydrogen Evolution Catalysts Based on Metal/Transition Metal Compounds [J]. Journal of Inorganic Materials, 2026, 41(2): 137-149. |
| [7] | FAN Yuzhu, WANG Yuan, WANG Linyan, XIANG Meiling, YAN Yuting, LI Benhui, LI Min, WEN Zhidong, WANG Haichao, CHEN Yongfu, QIU Huidong, ZHAO Bo, ZHOU Chengyu. Graphene Oxide-based Adsorbents for Pb(II) Removing in Water: Progresses on Synthesis, Performance and Mechanism [J]. Journal of Inorganic Materials, 2026, 41(1): 12-26. |
| [8] | XU Jintao, GAO Pan, HE Weiyi, JIANG Shengnan, PAN Xiuhong, TANG Meibo, CHEN Kun, LIU Xuechao. Recent Progress on Preparation of 3C-SiC Single Crystal [J]. Journal of Inorganic Materials, 2026, 41(1): 1-11. |
| [9] | YU Shengyang, SU Haijun, JIANG Hao, YU Minghui, YAO Jiatong, YANG Peixin. A Review of Pore Defects in Ultra-high Temperature Oxide Ceramics by Laser Additive Manufacturing: Formation and Suppression [J]. Journal of Inorganic Materials, 2025, 40(9): 944-956. |
| [10] | WEN Shenhao, PENG Dezhao, LIN Zheyu, GUO Xia, HUANG Peixin, ZHANG Zhizhen. Interface Engineering for the Anode in Solid-state Lithium Batteries Based on LLZTO Electrolyte [J]. Journal of Inorganic Materials, 2025, 40(9): 1013-1021. |
| [11] | LIU Jiangping, GUAN Xin, TANG Zhenjie, ZHU Wenjie, LUO Yongming. Research Progress on Catalytic Oxidation of Nitrogen-containing Volatile Organic Compounds [J]. Journal of Inorganic Materials, 2025, 40(9): 933-943. |
| [12] | XIAO Xiaolin, WANG Yuxiang, GU Peiyang, ZHU Zhenrong, SUN Yong. Advances in Regulation of Damaged Skin Regeneration by Two-dimensional Inorganic Materials [J]. Journal of Inorganic Materials, 2025, 40(8): 860-870. |
| [13] | MA Jingge, WU Chengtie. Application of Inorganic Bioceramics in Promoting Hair Follicle Regeneration and Hair Growth [J]. Journal of Inorganic Materials, 2025, 40(8): 901-910. |
| [14] | ZHANG Hongjian, ZHAO Ziyi, WU Chengtie. Inorganic Biomaterials on Regulating Neural Cell Function and Innervated Tissue Regeneration: A Review [J]. Journal of Inorganic Materials, 2025, 40(8): 849-859. |
| [15] | AI Minhui, LEI Bo. Micro-nanoscale Bioactive Glass: Functionalized Design and Angiogenic Skin Regeneration [J]. Journal of Inorganic Materials, 2025, 40(8): 921-932. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||