无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 409-420.DOI: 10.15541/jim20250307 CSTR: 32189.14.10.15541/jim20250307
• 综述 • 下一篇
彭德招1,2( ), 李瑞1,2, 王文鸿1,2, 王梓瑞1,2, 章志珍1,2(
), 李瑞1,2, 王文鸿1,2, 王梓瑞1,2, 章志珍1,2( )
)
收稿日期:2025-07-19
修回日期:2025-09-17
出版日期:2026-04-20
网络出版日期:2025-10-17
通讯作者:
章志珍, 副教授. E-mail: zhangzhzh28@mail.sysu.edu.cn作者简介:彭德招(1999-), 男, 博士研究生. E-mail: pengdzh5@mail2.sysu.edu.cn
基金资助:
PENG Dezhao1,2( ), LI Rui1,2, WANG Wenhong1,2, WANG Zirui1,2, ZHANG Zhizhen1,2(
), LI Rui1,2, WANG Wenhong1,2, WANG Zirui1,2, ZHANG Zhizhen1,2( )
)
Received:2025-07-19
Revised:2025-09-17
Published:2026-04-20
Online:2025-10-17
Contact:
ZHANG Zhizhen, associate professor. E-mail: zhangzhzh28@mail.sysu.edu.cnAbout author:PENG Dezhao (1999-), male, PhD candidate. E-mail: pengdzh5@mail2.sysu.edu.cn
Supported by:摘要:
钠离子电池具有成本低廉、钠资源丰富等优势, 是锂离子电池的潜在替代技术。开发兼具高离子电导率与宽电化学窗口的钠离子固态电解质对推动钠离子固态电池的发展与应用具有重要意义。在诸多固态电解质中, 氯化物电解质因具有高离子电导率、高氧化电位以及良好的塑性, 近年来受到广泛关注。本文系统梳理了钠氯化物固态电解质的发展脉络, 重点总结了固态电解质元素组成、晶体结构与离子电导率之间的内在关联, 并阐述了通过阳/阴离子掺杂、非晶化及异质结构复合等策略调控电解质离子输运性质的作用机制。此外, 本文讨论了钠氯化物固态电解质的电化学稳定性及其与正极材料的化学/电化学兼容性, 总结了其与钠金属负极的界面失效机制, 并简要概述了钠氯化物全固态电池的研究进展。最后, 本文凝练了氯化物全固态钠离子电池在实际应用中面临的关键挑战, 并对未来研究方向进行展望, 为该类材料在能源转换与储存领域中的实际应用提供指导。
中图分类号:
彭德招, 李瑞, 王文鸿, 王梓瑞, 章志珍. 钠氯化物固态电解质研究进展[J]. 无机材料学报, 2026, 41(4): 409-420.
PENG Dezhao, LI Rui, WANG Wenhong, WANG Zirui, ZHANG Zhizhen. Research Progress on Sodium Chloride Solid Electrolytes[J]. Journal of Inorganic Materials, 2026, 41(4): 409-420.
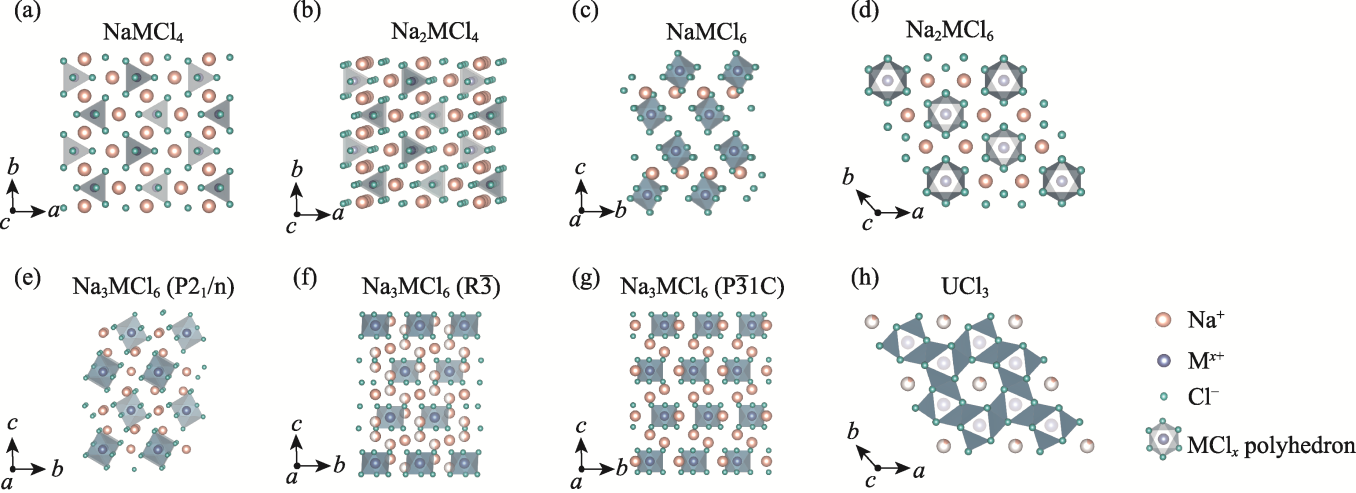
图2 不同类型钠氯化物固态电解质的晶体结构
Fig. 2 Crystal structures of different sodium chloride solid electrolytes (a) NaMCl4; (b) Na2MCl4; (c) NaMCl6; (d) Na2MCl6; (e) Na3MCl6 (P21/n); (f) Na3MCl6 ($\text{R}\overline{3}$); (g) Na3MCl6 ($\text{P}\overline{3}1\text{c}$); (h) UCl3
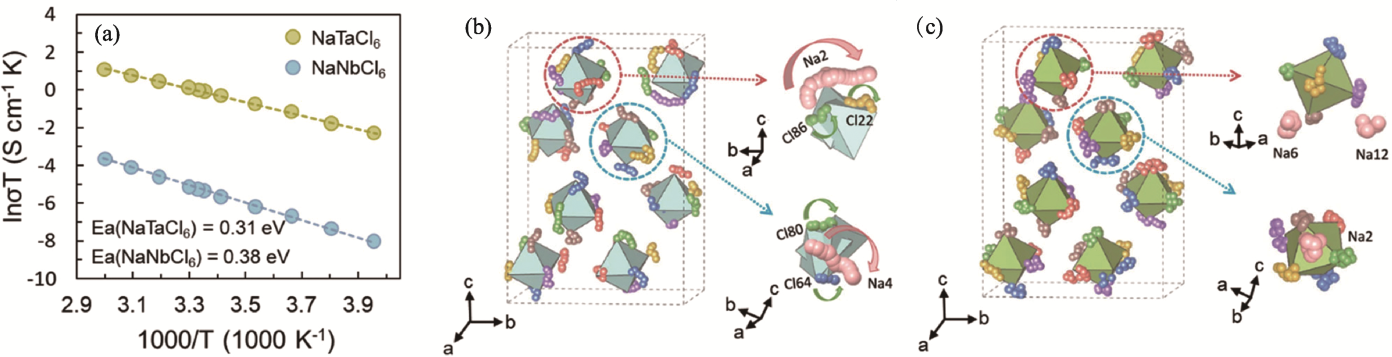
图3 NaMCl6型固态电解质的离子输运性质[52]
Fig. 3 Ion transport properties of NaMCl6 solid electrolytes[52] (a) Arrhenius plots of NaTaCl6 and NaNbCl6; (b, c) Atomic trajectories of Na and Cl atoms from ab initio molecular dynamics (AIMD) simulations for (b) NaTaCl6 and (c) NaNbCl6
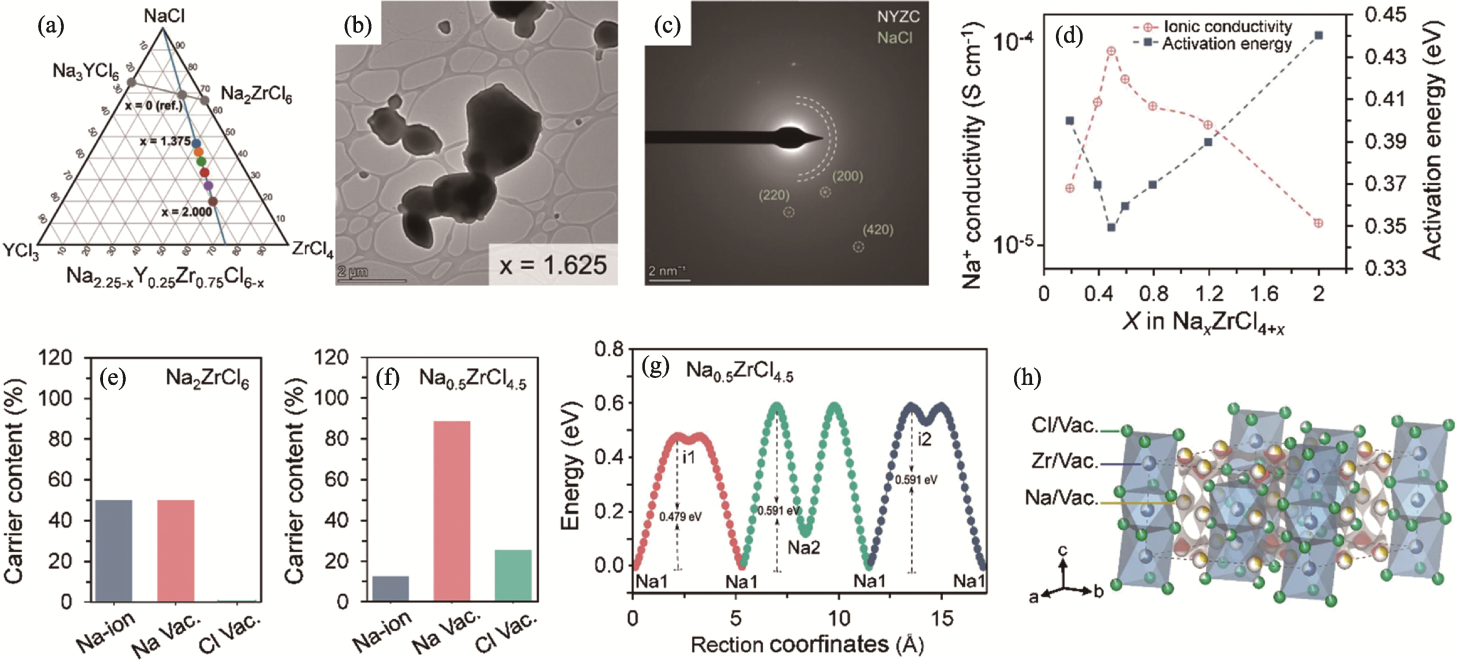
图4 Na3-xMCl6-x/Na2-xMCl6-x型固态电解质的相图、离子输运性质及结构[36,59]
Fig. 4 Phase diagram, ion transport properties and structures of Na3-xMCl6-x/Na2-xMCl6-x solid electrolytes[36,59] (a) Phase diagram of NaCl-YCl3-ZrCl4[59]; (b) Transmission electron microscope image of Na0.625Y0.25Zr0.75Cl4.375[59]; (c) Selected area electron diffraction pattern of Na0.625Y0.25Zr0.75Cl4.375[59]; (d) Ionic conductivity and activation energy of NaxZrCl4+x at 25 ℃[36]; (e, f) Na+ and vacancy contents in (e) Na2ZrCl6 and (f) Na0.5ZrCl4.5[36]; (g) Energy profiles along the Na+ transport pathways in Na0.5ZrCl4.5[36]; (h) Crystal structure of Na0.5ZrCl4.5[36]
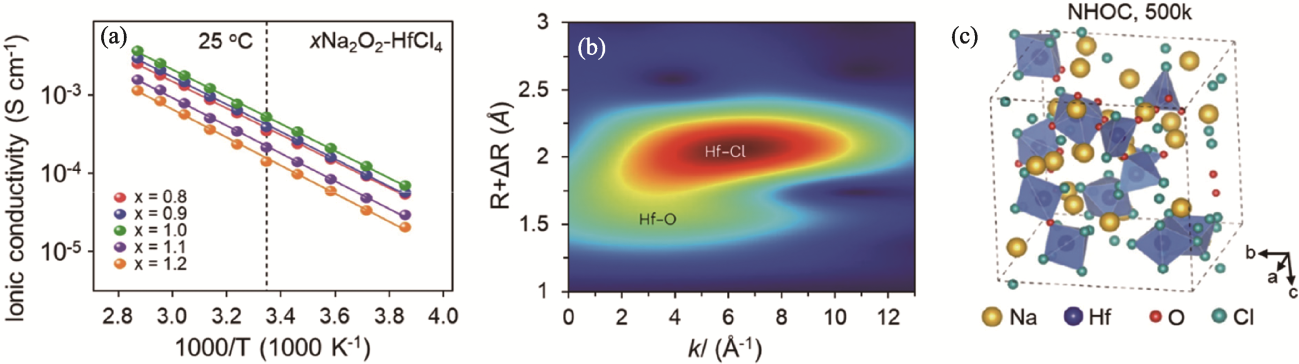
图5 xNa2O2-HfCl4型固态电解质的离子输运性质及结构[39]
Fig. 5 Ion transport property and structure of xNa2O2-HfCl4 solid electrolytes[39] (a) Arrhenius plots of xNa2O2-HfCl4; (b) Wavelet-transformed EXAFS contour plots of Na2O2-HfCl4 at Hf L3-edge[39];(c) AIMD-generated structure of amorphous Na2O2-HfCl4 at 500 K[39]

图6 xUCl3-(1-x)NaTaCl6型固态电解质的离子输运性质及结构[69]
Fig. 6 Ion transport property and structure of xUCl3-(1-x)NaTaCl6 solid electrolytes[69] (a) Arrhenius plots of 0.62Na0.75Sm1.75Cl6-0.38NaTaCl6 and 0.57Na0.75La1.75Cl6-0.43NaTaCl6; (b, c) Transmission electron microscope images of 0.62Na0.75Sm1.75Cl6-0.38NaTaCl6
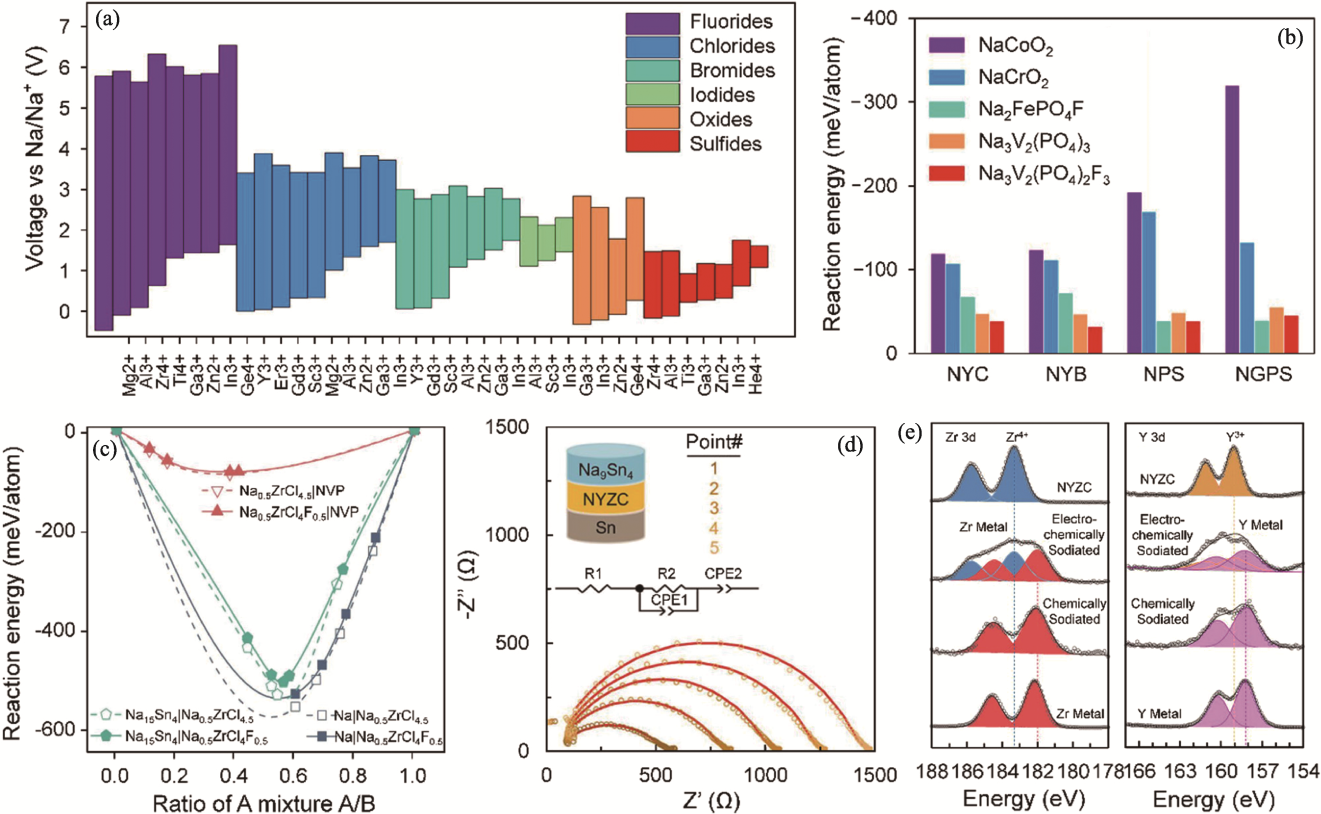
图7 钠氯化物固态电解质的电化学稳定性及其与电极材料的兼容性[36,71,74]
Fig. 7 Electrochemical stability of sodium chloride solid electrolytes and their compatibilities with electrode materials[36,71,74] (a) Calculated electrochemical stability windows of Na-M-X ternary compounds, including fluorides, chlorides, bromides, iodides, oxides, and sulfides[71]; (b) Calculated reaction energies between different cathodes and solid electrolytes for Na3YCl6 (NYC), Na3YBr6 (NYB), Na3PS4 (NPS), and Na10GeP2S12 (NGPS)[71]; (c) Calculated reaction energies between Na0.5ZrCl4.5, Na0.5ZrCl4F0.5 solid electrolytes and Na3V2(PO4)3 (NVP) cathode, Na anode and Na15Sn4 anode[36]; (d) Impedance evolution of Na9Sn4|Na2.25Y0.25Zr0.75Cl6|Na cells with the time increasing[74]; (e) Zr3d and Y3d XPS spectra of Na2.25Y0.25Zr0.75Cl6 obtained from Na9Sn4|Na2.25Y0.25Zr0.75Cl6|Na cells after charging and discharging[74]
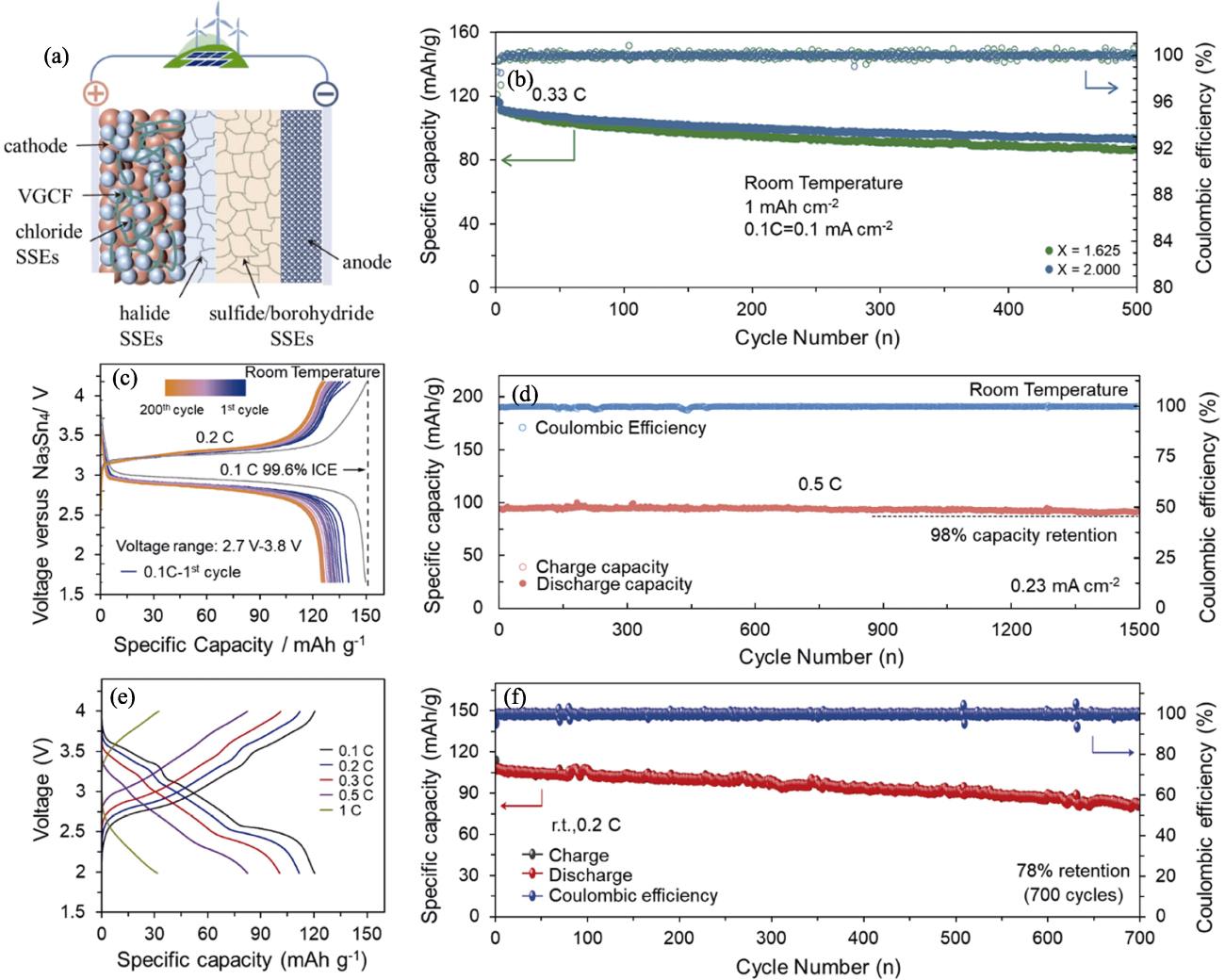
图8 氯化物全固态钠离子电池的电化学性能[36-37,39,59]
Fig. 8 Electrochemical performance of chloride all-solid-state sodium-ion batteries[36-37,39,59] (a) Schematic illustration of halide-based all-solid-state battery[36]; (b) Long cycling profiles of NaCrO2|Na2.25Y0.25Zr0.75Cl6|Na2(B10H10)0.5(B12H12)0.5|Na9Sn4 cells[59]; (c) Charge-discharge curves of Na3V2(PO4)3|NaTaCl6|Na3PS4|Na15Sn4 cells during cycling[37];(d) Long cycling profiles of Na3V2(PO4)3|NaTaCl6|Na3PS4|Na15Sn4 cells[37]; (e) Charge-discharge curves of Na0.85Mn0.5Ni0.4Fe0.1O2|Na2O2-HfCl4|Na3PS4|Na15Sn4 cells with different rate currents[39] and corresponding (f) long cycling profiles[39]
| [1] |
GOODENOUGH J B. Evolution of strategies for modern rechargeable batteries. Acc. Chem. Res., 2013, 46(5): 1053.
DOI URL |
| [2] |
GOODENOUGH J B, KIM Y. Challenges for rechargeable Li batteries. Chem. Mater., 2009, 22(3): 587.
DOI URL |
| [3] | XU J, CAI X, CAI S, et al. High-energy lithium-ion batteries: recent progress and a promising future in applications. Energy Environ. Mater., 2023, 6(5): 35. |
| [4] |
GAO Y L, PAN Z H, SUN J G, et al. High-energy batteries: beyond lithium-ion and their long road to commercialisation. Nano-Micro Lett., 2022, 14(1): 94.
DOI PMID |
| [5] | VAALMA C, BUCHHOLZ D, WEIL M, et al. A cost and resource analysis of sodium-ion batteries. Nat. Rev. Mater., 2018, 3(4): 3443. |
| [6] |
KUNDU D, TALAIE E, DUFFORT V, et al. The emerging chemistry of sodium Ion batteries for electrochemical energy storage. Angew. Chem. Int. Ed., 2015, 54(11): 3431.
DOI PMID |
| [7] |
HAYASHI A, NOI K, SAKUDA A, et al. Superionic glass-ceramic electrolytes for room-temperature rechargeable sodium batteries. Nat. Commun., 2012, 3: 856.
DOI |
| [8] |
CHE H, CHEN S, XIE Y, et al. Electrolyte design strategies and research progress for room-temperature sodium-ion batteries. Energy Environ. Sci., 2017, 10(5): 1075.
DOI URL |
| [9] |
SUN B, XIONG P, MAITRA U, et al. Design strategies to enable the efficient use of sodium metal anodes in high-energy batteries. Adv. Mater., 2019, 32(18): 1903891.
DOI URL |
| [10] |
ZHANG X, LI L, FAN E, et al. Toward sustainable and systematic recycling of spent rechargeable batteries. Chem. Soc. Rev., 2018, 47(19): 7239.
DOI PMID |
| [11] |
YAO X, HUANG B, YIN J, et al. All-solid-state lithium batteries with inorganic solid electrolytes: review of fundamental science. Chin. Phys. B, 2016, 25(1): 018802.
DOI URL |
| [12] |
HIRSH H S, LI Y, TAN D H S, et al. Sodium-ion batteries paving the way for grid energy storage. Adv. Energy Mater., 2020, 10(32): 2001274.
DOI URL |
| [13] |
SAND S C, RUPP J L M, YILDIZ B. A critical review on Li-ion transport, chemistry and structure of ceramic-polymer composite electrolytes for solid state batteries. Chem. Soc. Rev., 2025, 54(1): 178.
DOI PMID |
| [14] |
JANEK J, ZEIER W G. A solid future for battery development. Nat. Energy, 2016, 1: 24.
DOI |
| [15] |
MANTHIRAM A, YU X W, WANG S F. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater., 2017, 2(4): 16103.
DOI |
| [16] |
CHEN R, LI Q, YU X, et al. Approaching practically accessible solid-state batteries: stability issues related to solid electrolytes and interfaces. Chem. Rev., 2020, 120(14): 6820.
DOI PMID |
| [17] |
YU T, YANG X, YANG R, et al. Progress and perspectives on typical inorganic solid-state electrolytes. J. Alloys Compd., 2021, 885: 161013.
DOI URL |
| [18] |
LU P, XIA Y, SUN G, et al. Realizing long-cycling all-solid-state Li-In||TiS2 batteries using Li6+xMxAs1-xS5I (M=Si, Sn) sulfide solid electrolytes. Nat. Commun., 2023, 14: 4077.
DOI |
| [19] |
KATO Y, HORI S, SAITO T, et al. High-power all-solid-state batteries using sulfide superionic conductors. Nat. Energy., 2016, 1(4): 16030.
DOI |
| [20] | NIE X, HU J, LI C. Halide-based solid electrolytes: the history, progress, and challenges. Interdiscip. Mater., 2023, 2(3): 365. |
| [21] | WANG C, LIANG J, KIM J T, et al. Prospects of halide-based all-solid-state batteries: from material design to practical application. Sci. Adv., 2022, 8(36): eadc9516. |
| [22] |
WANG Q, ZHOU Y, WANG X, et al. Designing lithium halide solid electrolytes. Nat. Commun., 2024, 15(1): 1050.
DOI PMID |
| [23] | ASANO T, SAKAI A, OUCHI S, et al. Solid halide electrolytes with high lithium-ion conductivity for application in 4 V class bulk-type all-solid-state batteries. Adv. Mater., 2018, 30(44): e1803075. |
| [24] | WEI Z, NAZAR L F, JANEK J. Emerging halide solid electrolytes for sodium solid-state batteries: structure, conductivity, paradigm of applications. Batteries Supercaps, 2024, 7(7): e202400005. |
| [25] |
LI X N, LIANG J W, YANG X F, et al. Progress and perspectives on halide lithium conductors for all-solid-state lithium batteries. Energy. Environ. Sci., 2020, 13(5): 1429.
DOI URL |
| [26] |
TUO K, SUN C W, LIU S Q. Recent progress in and perspectives on emerging halide superionicconductors for all-solid-state batteries. Electrochem. Energy Rev., 2023, 6(1): 17.
DOI |
| [27] | HUANG L J, ZHANG L, BI J Y, et al. An insight into halide solid-state electrolytes: progress and modification strategies. Energy Mater. Adv., 2024, 5: 35632. |
| [28] |
LIANG J, LI X, ADAIR K R, et al. Metal halide superionic conductors for all-solid-state batteries. Acc. Chem. Res., 2021, 54(4): 1023.
DOI URL |
| [29] |
KWAK H, WANG S, PARK J, et al. Emerging halide superionic conductors for all-solid-state batteries: design, synthesis, and practical applications. ACS Energy Lett., 2022, 7(5): 1776.
DOI URL |
| [30] | KWAK H, LYOO J, PARK J, et al. Na2ZrCl6 enabling highly stable 3 V all-solid-state Na-ion batteries. Energy Storage Mater., 2023, 37: 47. |
| [31] |
ZHAO T, SOBOLEV A N, SCHLEM R, et al. Synthesis-controlled cation solubility in solid sodium ion conductors Na2+xZr1-xInxCl6. ACS Appl. Energy Mater., 2023, 6(8): 4334.
DOI URL |
| [32] |
SEBTI E, QI J, RICHARDSON P M, et al. Synthetic control of structure and conduction properties in Na-Y-Zr-Cl solid electrolytes. J. Mater. Chem. A, 2022, 10(40): 21565.
DOI URL |
| [33] | STEINER H J, LUTZ H D. Neue schnelle ionenleiter vom Typ MⅠ3MⅢCl6 (MⅠ=Li, Na, Ag; MⅢ=In, Y). Z. Anorg. Allg. Chem., 1992, 613(7): 26. |
| [34] |
SCHLEM R, BANIK A, ECKARDT M, et al. Na3-xEr1-xZrxCl6--a halide-based fast sodium-ion conductor with vacancy-driven ionic transpor. ACS Appl. Energy Mater., 2020, 3(10): 10164.
DOI URL |
| [35] |
WU E A, BANERJEE S, TANG H, et al. A stable cathode-solid electrolyte composite for high-voltage, long-cycle-life solid-state sodium-ion batteries. Nat. Commun., 2021, 12: 1256.
DOI PMID |
| [36] | WU M, LIU X, LIU H, et al. Fluorinated amorphous halides with improved ionic conduction and stability for all-solid-state sodium-ion batteries. Nat. Commun., 2025, 16: 2808. |
| [37] |
HU Y, FU J, XU J, et al. Superionic amorphous NaTaCl6 halide electrolyte for highly reversible all-solid-state Na-ion batteries. Matter, 2024, 7(3): 1018.
DOI URL |
| [38] | ZHAO T, SAMANTA B, DE IRUJO-LABALDE X M, et al. Sodium metal oxyhalides NaMOCl4 (M=Nb, Ta) with high ionic conductivities. ACS Mater. Lett., 2024, 6(8): 3683. |
| [39] |
LIN X, ZHANG S, YANG M, et al. A family of dual-anion-based sodium superionic conductors for all-solid-state sodium-ion batteries. Nat. Mater., 2024, 24(1): 83.
DOI PMID |
| [40] | LIN X, ZHAO Y, WANG C, et al. A dual anion chemistry-based superionic glass enabling long-cycling all-solid-state sodium-ion batteries. Angew. Chem. Int. Ed., 2023, 13: e202314181. |
| [41] |
KWAK H, KIM J S, HAN D, et al. Boosting the interfacial superionic conduction of halide solid electrolytes for all-solid-state batteries. Nat. Commun., 2023, 14: 2459.
DOI PMID |
| [42] |
BAENZIGER N C. The crystal structure of NaAlCl4. Acta Cryst., 1951, 4(3): 216.
DOI URL |
| [43] |
PARK J, SON J P, KO W, et al. NaAlCl4: new halide solid electrolyte for 3 V stable cost-effective all-solid-state Na-ion batteries. ACS Energy Lett., 2022, 7(10): 3293.
DOI URL |
| [44] |
HAFNER M, BIANCHINI M. Exploring cationic substitutions in the solid electrolyte NaAlCl4 with density functional theory. J. Phys. Chem. C, 2024, 128(47): 19978.
DOI URL |
| [45] | GOODYEAR J, ALI S A D, STEIGMANN G A. The crystal structure of Na2MnCl4. Acta Cryst., 1971, B27(8): 1672. |
| [46] | VAN LOON C J J, IJDO D J W. The crystal structure of Na6MnCl8 and Na2Mn3Cl8 and some isostructural compounds. Acta Cryst., 1975, B31(3): 770. |
| [47] | VAN LOON C J J, VISSER D. Chlorides with the chrysoberyl structure: Na2CoCl4 and Na2ZnCl4. Acta Cryst., 1977, B33(1): 188. |
| [48] | LUTZ H D, WUSSOW K, KUSKE P. Ionic conductivity, structural, IR and Raman spectroscopic data of olivine, Sr2PbO4, and Na2CuF4 type lithium and sodium chlorides Li2ZnCl4 and Na2MCl4 (M = Mg, Ti, Cr, Mn, Co, Zn, Cd). Z Naturforsch., 1987, 42b(11): 1379. |
| [49] | GUO H, HAFNER M, GRUNINGER H, et al. Structure and ionic conductivity of halide solid electrolytes based on NaAlCl4 and Na2ZnCl4. Adv. Sci., 2025, 12(30): e07224. |
| [50] |
SADOWAY D R, FLENGAS S N. The synthesis and properties of the hexachloroniobates and hexachlorotantalates of Na, K, Rb, and Cs. Can. J. Chem., 1978, 56(15): 2013.
DOI URL |
| [51] | HENKE H. Zur kristallchemischen einordnung von NaSbCl6, NaNbCl6 und NaTaCl6. Z. Krist.-Cryst. Mater., 1992, 198(1/2): 1. |
| [52] |
LI R, XU K Q, WEN S H, et al. A sodium superionic chloride electrolyte driven by paddle wheel mechanism for solid state batteries. Nat. Commun., 2025, 16: 6633.
DOI |
| [53] |
KIPOUROS G J, FLENGAS S N. Equilibrium decomposition pressures of the compounds Na2ZrCl6 and Na2HfCl6. Can. J. Chem., 1981, 59(6): 990.
DOI URL |
| [54] |
ZHAO T, SOBOLEV A N, DE IRUJO LABALDE X M, et al. On the influence of the coherence length on the ionic conductivity in mechanochemically synthesized sodium-conducting halides, Na3-xIn1-xZrxCl6. J. Mater. Chem. A, 2024, 12(12): 7015.
DOI URL |
| [55] |
ZHOU L, ZHANG S, LI W, et al. Amorphous-nanocrystalline fluorinated halide electrolytes with high ionic conductivity and high-voltage stability. J. Am. Chem. Soc., 2025, 147(18): 15136.
DOI URL |
| [56] |
PARK D, KIM K, CHUN G H, et al. Materials design of sodium chloride solid electrolytes Na3MCl6 for all-solid-state sodium-ion batteries. J. Mater. Chem. A, 2021, 9(40): 23037.
DOI URL |
| [57] |
YU S, KIM K, WOOD B C, et al. Structural design strategies for superionic sodium halide solid electrolytes. J. Mater. Chem. A, 2022, 10(45): 24301.
DOI URL |
| [58] |
NIU X, DOU X, FU C, et al. Sodium halide solid state electrolyte of Na3YBr6 with low activation energy. RSC Adv., 2024, 14(21): 14716.
DOI URL |
| [59] |
RIDLEY P, NGUYEN L H B, SEBTI E, et al. Amorphous and nanocrystalline halide solid electrolytes with enhanced sodium-ion conductivity. Matter, 2024, 7(2): 485.
DOI URL |
| [60] | LUO J D, ZHANG Y, CHENG X, et al. Halide superionic conductors with non-close-packed anion frameworks. Angew. Chem. Int. Ed., 2024, 63(17): e202400424. |
| [61] |
LISSNER F, KRÄMER K, SCHLEID T, et al. Die chloride Na3xM2-xCl6 (M=La, Sm) and NaM2Cl6 (M=Nd, Sm): derivate des UCl3-Typs. Synthese, kristallstruktur und röntgenabsorptionsspektroskopie (XANES). Z. Anorg. Allg. Chem., 1994, 620(3): 444.
DOI URL |
| [62] |
WICKLEDER M S, MEYER G. Neue derivate des UCl3-typs: die chloride and bromide A(SrSm)Cl6, A(SrEu)Cl6 und A(BaLa)X6 (A=Na, Ag; X=Cl, Br). Z. Anorg. Allg. Chem., 1999, 624(10): 1577.
DOI URL |
| [63] |
FU C, LI Y, XU W, et al. LaCl3-based sodium halide solid electrolytes with high ionic conductivity for all-solid-state batteries. Nat. Commun., 2024, 15: 5646.
DOI |
| [64] |
DAI T, WU S Y, LU Y X, et al. Inorganic glass electrolytes with polymer-like viscoelasticity. Nat. Energy, 2023, 8(11): 1221.
DOI |
| [65] |
MOTOHASHI K, TSUKASAKI H, MORI S, et al. Fast sodium-ion conducting amorphous oxychloride embedding nanoparticles. Chem. Mater., 2024, 36(19): 9914.
DOI URL |
| [66] |
ZHOU L, BAZAK J D, LI C, et al. 4 V Na solid state batteries enabled by a scalable sodium metal oxyhalide solid electrolyte. ACS Energy Lett., 2024, 9(8): 4093.
DOI URL |
| [67] |
LIANG C C. Conduction characteristics of the lithium iodide-aluminum oxide solid electrolytes. J. Electrochem. Soc., 1973, 120(10): 1289.
DOI URL |
| [68] |
MERCIER R, MALUGANI J P, FAHYS B, et al. Superionic conduction in Li2S-P2S5-LiI-glasses. Solid State Ionics, 1981, 5: 663.
DOI URL |
| [69] | FU J, WANG S, WU D, et al. Halide heterogeneous structure boosting ionic diffusion and high-voltage stability of sodium superionic conductors. Adv. Mater., 2024, 36(3): e2308012. |
| [70] | YU Q, HU J, XU Y, et al. Mesoporous enhanced heterostructured halide solid electrolytes with high air stability and high abundance for sustainable sodium metal batteries. Angew. Chem. Int. Ed., 2025, 64(26): e202425503. |
| [71] |
QIE Y, WANG S, FU S, et al. Yttrium-sodium halides as promising solid-state electrolytes with high ionic conductivity and stability for Na-ion batteries. J. Phys. Chem. Lett., 2020, 11(9): 3376.
DOI PMID |
| [72] |
PARK J, HAN D, SON J P, et al. Extending the electrochemical window of Na+ halide nanocomposite solid electrolytes for 5 V- class all-solid-state Na-ion batteries. ACS Energy Lett., 2024, 9(5): 2222.
DOI URL |
| [73] |
GOODWIN L E, ZIEGLER M, TILL P, et al. Halide and sulfide electrolytes in cathode composites for sodium all-solid-state batteries and their stability. ACS Appl. Mater. Interfaces, 2024, 16(15): 19792.
DOI URL |
| [74] |
DEYSHER G, CHEN Y T, SAYAHPOUR B, et al. Evaluating electrolyte-anode interface stability in sodium all-solid-state batteries. ACS Appl. Mater. Interfaces, 2022, 14(42): 47706.
DOI URL |
| [75] |
ZHANG J, YU Z, ZHU Y, et al. Configuration design and interface reconstruction to realize the superior high-rate performance for sodium layered oxide cathodes. Adv. Energy Mater., 2025, 15(23): 2405951.
DOI URL |
| [76] |
ZHANG Z, ZHANG Q, SHI J, et al. A self-forming composite electrolyte for solid-state sodium battery with ultralong cycle life. Adv. Energy Mater., 2017, 7(4): 1601196.
DOI URL |
| [77] | DONG Z L, GAN Y, MARTINS V, et al. Novel sulfide-chloride solid-state electrolytes with tunable anion ratio for highly stable solid-state sodium-ion batteries. Adv. Mater., 2025, 37(30): e2503107. |
| [78] |
XU J, LI Y, LU P, et al. Water-stable sulfide solid electrolyte membranes directly applicable in all-solid-state batteries enabled by superhydrophobic Li+-conducting protection layer. Adv. Energy Mater., 2021, 12(2): 2102348.
DOI URL |
| [1] | 李璇, 叶奎材, 冯佳音, 邱家军, 钱文昊, 邢敏. 钛基牙种植体表面改性促进软组织封闭的研究进展[J]. 无机材料学报, 2026, 41(4): 432-444. |
| [2] | 陈坤, 姜勇刚, 冯军宗, 李良军, 胡艺洁, 冯坚. 锆酸镧多孔隔热材料研究进展[J]. 无机材料学报, 2026, 41(4): 421-431. |
| [3] | 韦连金, 齐志杰, 汪信, 朱俊武, 付永胜. 纳米金刚石改性及其在电催化氧还原反应中的应用[J]. 无机材料学报, 2026, 41(3): 273-288. |
| [4] | 刘占一, 李勉, 欧阳晓平, 柴之芳, 黄庆. 干法后处理熔盐中Sr/Cs去除方法的研究进展[J]. 无机材料学报, 2026, 41(2): 150-158. |
| [5] | 孙炼, 张磊磊, 薛泽旭, 吴坤, 陈晔, 李志远, 王鲁凯, 王尊刚. 面向辐射探测应用的零维金属卤化物闪烁体研究进展[J]. 无机材料学报, 2026, 41(2): 159-176. |
| [6] | 任先培, 李超, 胡启威, 向晖, 彭跃红. 金属/过渡金属化合物莫特-肖特基析氢催化剂研究进展[J]. 无机材料学报, 2026, 41(2): 137-149. |
| [7] | 范雨竹, 王媛, 王林燕, 向美玲, 鄢雨婷, 黎本慧, 李敏, 文志东, 王海超, 陈永福, 邱会东, 赵波, 周成裕. 氧化石墨烯基吸附材料去除水体中Pb(II): 制备、性能及机理[J]. 无机材料学报, 2026, 41(1): 12-26. |
| [8] | 徐锦涛, 高攀, 何唯一, 蒋圣楠, 潘秀红, 汤美波, 陈锟, 刘学超. 3C-SiC晶体制备研究进展[J]. 无机材料学报, 2026, 41(1): 1-11. |
| [9] | 余升阳, 苏海军, 姜浩, 余明辉, 姚佳彤, 杨培鑫. 激光增材制造超高温氧化物陶瓷孔隙缺陷形成及抑制研究进展[J]. 无机材料学报, 2025, 40(9): 944-956. |
| [10] | 文伸豪, 彭德招, 林喆与, 郭霞, 黄培鑫, 章志珍. 基于LLZTO电解质的固态锂金属电池负极界面调控[J]. 无机材料学报, 2025, 40(9): 1013-1021. |
| [11] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [12] | 肖晓琳, 王玉祥, 谷佩洋, 朱圳荣, 孙勇. 二维无机材料调控病损皮肤组织再生的研究进展[J]. 无机材料学报, 2025, 40(8): 860-870. |
| [13] | 马景阁, 吴成铁. 无机生物材料用于毛囊和毛发再生的研究[J]. 无机材料学报, 2025, 40(8): 901-910. |
| [14] | 张洪健, 赵梓壹, 吴成铁. 无机生物材料调控神经细胞功能及神经化组织再生的研究进展[J]. 无机材料学报, 2025, 40(8): 849-859. |
| [15] | 艾敏慧, 雷波. 微纳米生物活性玻璃: 功能化设计与血管化皮肤再生[J]. 无机材料学报, 2025, 40(8): 921-932. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||