
Journal of Inorganic Materials ›› 2026, Vol. 41 ›› Issue (5): 637-644.DOI: 10.15541/jim20250348
• RESEARCH ARTICLE • Previous Articles Next Articles
LI Zhongyi( ), LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo(
), LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo( )
)
Received:2025-08-31
Revised:2025-11-09
Published:2025-11-26
Online:2025-11-26
Contact:
JIANG Haibo, associate professor. E-mail: jianghaibo@ecust.edu.cnAbout author:LI Zhongyi (2001-), male, Master candidate. E-mail: 2911277205@qq.com
Supported by:CLC Number:
LI Zhongyi, LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo. Enhanced Sulfur Fixation Efficiency of Calcium Hydroxide by Fe3+: Dual Mechanisms of Oxidation and Catalysis[J]. Journal of Inorganic Materials, 2026, 41(5): 637-644.
| CaO/g | Deionized water/g | H2O/CaO |
|---|---|---|
| 10 | 6 | 0.6 |
| 10 | 7 | 0.7 |
| 10 | 8 | 0.8 |
| 10 | 9 | 0.9 |
| 10 | 10 | 1.0 |
| 10 | 11 | 1.1 |
Table 1 Preparation of pure calcium hydroxide
| CaO/g | Deionized water/g | H2O/CaO |
|---|---|---|
| 10 | 6 | 0.6 |
| 10 | 7 | 0.7 |
| 10 | 8 | 0.8 |
| 10 | 9 | 0.9 |
| 10 | 10 | 1.0 |
| 10 | 11 | 1.1 |
| CaO/g | Deionized water/g | Fe(OH)3/g | H2O/CaO |
|---|---|---|---|
| 10 | 6 | 6.25 | 0.6 |
| 10 | 7 | 6.25 | 0.7 |
| 10 | 8 | 6.25 | 0.8 |
| 10 | 9 | 6.25 | 0.9 |
| 10 | 10 | 6.25 | 1.0 |
| 10 | 11 | 6.25 | 1.1 |
Table 2 Preparation of calcium-iron desulfurizer
| CaO/g | Deionized water/g | Fe(OH)3/g | H2O/CaO |
|---|---|---|---|
| 10 | 6 | 6.25 | 0.6 |
| 10 | 7 | 6.25 | 0.7 |
| 10 | 8 | 6.25 | 0.8 |
| 10 | 9 | 6.25 | 0.9 |
| 10 | 10 | 6.25 | 1.0 |
| 10 | 11 | 6.25 | 1.1 |
| Composition | Content/% (in mass) | Composition | Content/% (in mass) |
|---|---|---|---|
| CaO | 96.05 | SO3 | 0.16 |
| MgO | 2.1 | K2O | 0.086 |
| Al2O3 | 0.33 | Fe2O3 | 0.426 |
| SiO2 | 0.757 | SrO | 0.09 |
Table 3 Composition analysis of quicklime sample
| Composition | Content/% (in mass) | Composition | Content/% (in mass) |
|---|---|---|---|
| CaO | 96.05 | SO3 | 0.16 |
| MgO | 2.1 | K2O | 0.086 |
| Al2O3 | 0.33 | Fe2O3 | 0.426 |
| SiO2 | 0.757 | SrO | 0.09 |
| H2O/CaO | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) |
|---|---|---|
| 0.6 | 25.596 | 0.116 |
| 0.7 | 25.419 | 0.122 |
| 0.8 | 27.256 | 0.130 |
| 0.9 | 26.214 | 0.127 |
| 1.0 | 26.646 | 0.135 |
| 1.1 | 31.694 | 0.182 |
Table 4 Specific surface areas and pore volumes of calcium-iron desulfurizers
| H2O/CaO | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) |
|---|---|---|
| 0.6 | 25.596 | 0.116 |
| 0.7 | 25.419 | 0.122 |
| 0.8 | 27.256 | 0.130 |
| 0.9 | 26.214 | 0.127 |
| 1.0 | 26.646 | 0.135 |
| 1.1 | 31.694 | 0.182 |
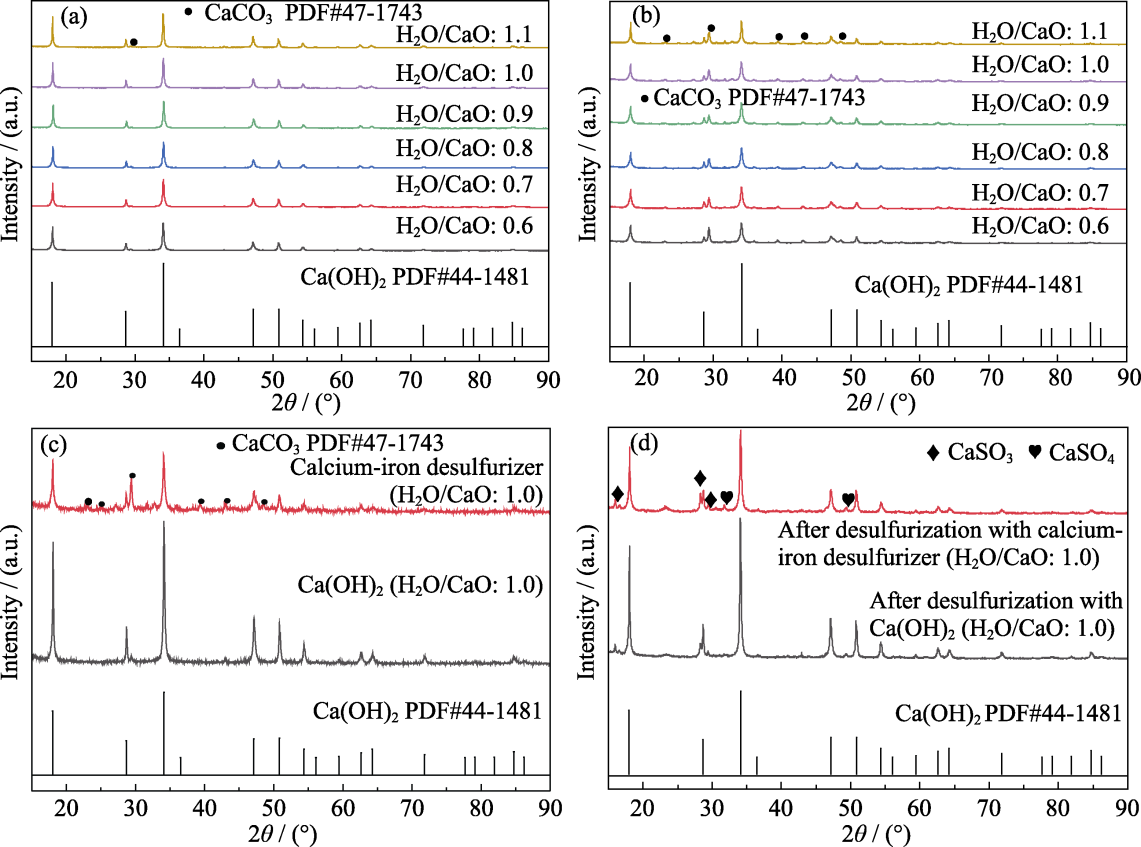
Fig. 4 XRD patterns of desulfurizers before and after desulfurization (a) Pure Ca(OH)2 with water cement ratio of 0.6-1.1; (b) Calcium-iron desulfurizers with water cement ratio of 0.6-1.1; (c, d) Comparison of pure Ca(OH)2 and calcium-iron desulfurizer with water cement ratio of 1.0 before (c) and after (d) desulfurization
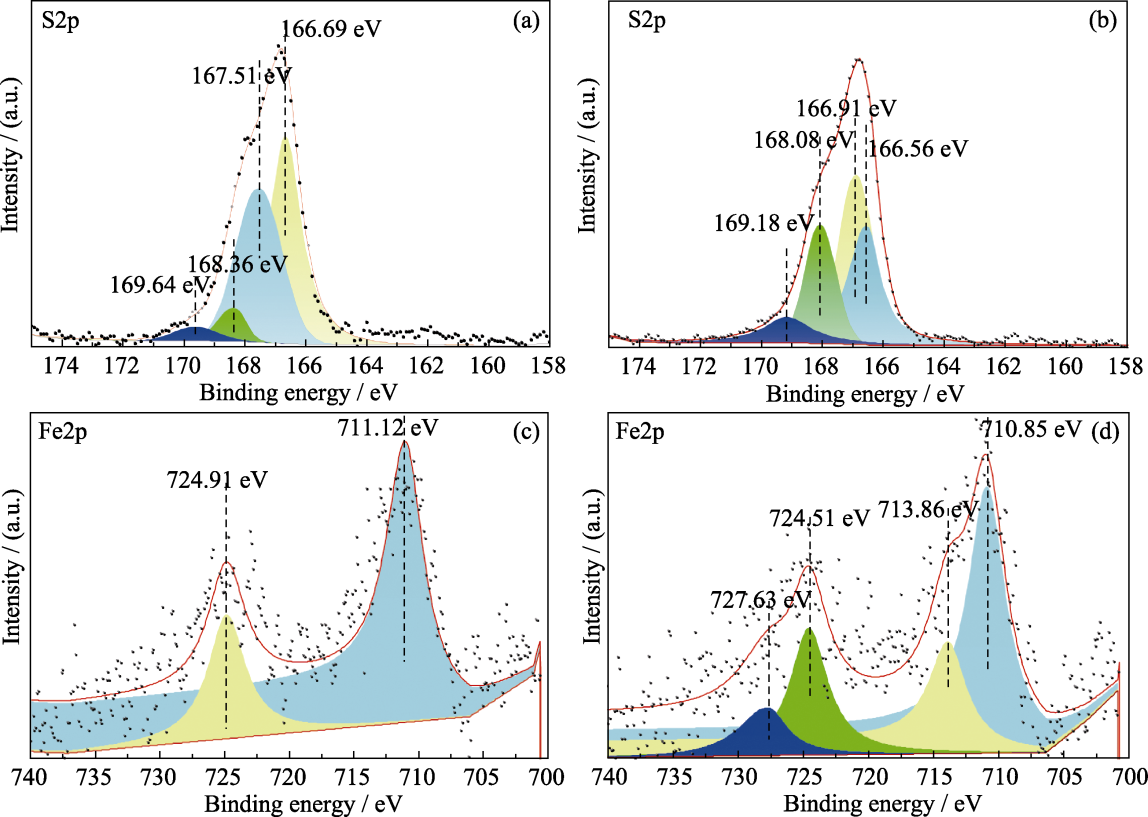
Fig. 6 XPS spectra of pure Ca(OH)2 and calcium-iron desulfurizer with water cement ratio of 1.0 before and after desulfurization (a) S2p of pure Ca(OH)2 after desulfurization; (b) S2p of calcium-iron desulfurizer after desulfurization; (c) Fe2p of calcium-iron desulfurizer before desulfurization; (d) Fe2p of calcium-iron desulfurizer after desulfurization
| Desulfurizer | Sulfur fixation efficiency/% |
|---|---|
| Calcium-iron desulfurizer | 17.07 |
| Yan et al.[ | 1.95 |
| Li et al.[ | 6.4 |
Table 5 Comparison of sulfur fixation efficiency between calcium-iron desulfurizer and other desulfurizers
| Desulfurizer | Sulfur fixation efficiency/% |
|---|---|
| Calcium-iron desulfurizer | 17.07 |
| Yan et al.[ | 1.95 |
| Li et al.[ | 6.4 |
| [1] |
LUM M M X, NG K H, LAI S Y, et al. Sulfur dioxide catalytic reduction for environmental sustainability and circular economy: a review. Process Safety and Environmental Protection, 2023, 176: 580.
DOI URL |
| [2] |
CHEN W C, XIANG Y H, LIU J, et al. Foreign investor and industrial pollution: evidence from sulfur dioxide emission. Finance Research Letters, 2022, 50: 103279.
DOI URL |
| [3] |
YANG C H, CHEN P H, YANG C S, et al. Analysis and forecasting of air pollution on nitrogen dioxide and sulfur dioxide using deep learning. IEEE Access, 2024, 12: 165236.
DOI URL |
| [4] |
ZHANG J Y. The mechanism of foreign direct investment on China’s sulfur dioxide emissions: evidence from partial least squares structural equation modeling. Journal of Environmental Management, 2025, 380: 124998.
DOI URL |
| [5] |
QIAN Y, CAO H, HUANG S M. Decoupling and decomposition analysis of industrial sulfur dioxide emissions from the industrial economy in 30 Chinese provinces. Journal of Environmental Management, 2020, 260: 110142.
DOI URL |
| [6] |
LI Y, GAO L H, ZHANG J H, et al. Synergetic utilization of microwave-assisted fly ash and carbide slag for simultaneous desulfurization and denitrification: high efficiency, low cost and catalytic mechanism. Chemical Engineering Journal, 2022, 437: 135488.
DOI URL |
| [7] |
DAI G F, ZHANG J Y, WANG X B, et al. Calcination and desulfurization characteristics of calcium carbonate in pressurized oxy-combustion. Energy, 2022, 261: 125150.
DOI URL |
| [8] |
KUMAR A. Desulfurization performance of sulfur dioxide and product characteristics in semi-batch bubble column and foam-bed contactor. Chemical Papers, 2019, 74: 2427.
DOI |
| [9] |
OSAKA Y, TSUIGUCHI T, KODAMA A, et al. Improvement of dry desulfurization performance using activated calcium carbonate by amorphous citric acid complex method for diesel gas purification. Journal of Material Cycles and Waste Management, 2020, 22: 470.
DOI |
| [10] |
SHIN H G, KIM H, NOH T, et al. Degradation behavior of high surface area calcium hydroxide sorbent for SO2 removal. International Journal of Mineral Processing, 2011, 98(3/4): 145.
DOI URL |
| [11] |
YAN D J, HE R N, WU X Y, et al. An investigation of the surface structure of surfactants modified calcium hydroxide and enhancement of dry flue gas desulfurization performance. Research on Chemical Intermediates, 2025, 51(3): 1583.
DOI |
| [12] | SHIN H G, KIM H, KIM Y N, et al. Preparation and characterization of high surface area calcium hydroxide sorbent for SO2 removal. Current Applied Physics, 2009, 9(3): S276. |
| [13] |
LIU M, DING W X, CHANG J Y, et al. Pilot study on coke oven flue gas injection desulfurization using highly active modified calcium hydroxide desulfurizer. Asia-Pacific Journal of Chemical Engineering, 2025, 20(2): e3170.
DOI URL |
| [14] | 熊爽, 严金生, 周洲, 等. 生石灰消化反应条件对氢氧化钙特性影响. 无机盐工业, 2023, 55(12): 50. |
| [15] | 刘越, 郑强, 邢佳斌, 等. 石灰石干法制备高性能氢氧化钙的工艺及应用研究. 无机盐工业, 2023, 55(10): 42. |
| [16] |
RENEDO M J, FERNÁNDEZ-FERRERAS J. Characterization and behavior of modified calcium-hydroxide-based sorbents in a dry desulfurization process. Energy & Fuels, 2016, 30(8): 6350.
DOI URL |
| [17] |
LI Y R, QI H Y, YOU C F, et al. Kinetic model of CaO/fly ash sorbent for flue gas desulphurization at moderate temperatures. Fuel, 2007, 86(5/6): 785.
DOI URL |
| [18] |
FANG D X, HUANG L P, FANG Z Y, et al. Evaluation of porous calcium silicate hydrate derived from carbide slag for removing phosphate from wastewater. Chemical Engineering Journal, 2018, 354: 1.
DOI URL |
| [19] | 李春花, 苏春华, 王超, 等. 草木灰促进CaO粉末高温烟气脱硫实验研究. 应用化工, 2010, 39(4): 521. |
| [20] |
SALVITTI C, ROSI M, PEPI F, et al. Reactivity of transition metal dioxide anions MO2- (M = Co, Ni, Cu, Zn) with sulfur dioxide in the gas phase: an experimental and theoretical study. Chemical Physics Letters, 2021, 776: 138555.
DOI URL |
| [21] | 高翔, 骆仲泱, 陈亚非, 等. 钙基吸收剂脱硫反应特性的研究. 燃烧科学与技术, 1998, 4(4): 369. |
| [22] |
SU M H, YANG L C. Fe/Mn-MOFs with monocarboxylic acid-induced defects enhances the catalytic oxidation of calcium sulfite in desulfurization ash. Separation and Purification Technology, 2024, 353: 128300.
DOI URL |
| [23] |
LATYPOVA S, ESEVA E, LEVIN I, et al. Catalytic performance of transition metal molybdates in accelerated oxidative desulfurization. Fuel, 2025, 403: 136114.
DOI URL |
| [24] |
CHEN X Y, ZHOU S R, WANG L Y, et al. Facile preparation of Fe-Beta zeolite-supported transition metal oxide catalysts and their catalytic performance for the simultaneous removal of NOx and soot. Chinese Journal of Chemical Engineering, 2024, 76: 10.
DOI URL |
| [25] |
KIM H M, JEONG C H, CHEON B S, et al. Improving the performance of a Co-CeO2 catalyst for hydrogen production via water gas shift reaction by addition of transition metal oxides. Energy & Fuels, 2024, 38(5): 4743.
DOI URL |
| [26] |
WU H D, YU B, Ni C S, et al. Enhancement of catalytic performance in Zr0.1Ce0.9O2-δ through transition metal doping and exsolution. Solid State Ionics, 2025, 425: 116868.
DOI URL |
| [27] | SUN B, WANG J G, CHEN M, et al. Boosting acetone oxidation performance over mesocrystal MxCe1-xO2 (M = Ni, Cu, Zn) solid solution within hollow spheres by tailoring transition-metal cations. International Materials Chemistry and Physics, 2023, 293: 126925. |
| [28] |
HE K J, SONG Q, YAN Z N, et al. Study on competitive absorption of SO3 and SO2 by calcium hydroxide. Fuel, 2019, 242: 355.
DOI URL |
| [29] |
MA X Y, WU H, CHANG L, et al. The influence of calcium hydroxide crystal morphology on the desulfurization of cement kiln flue gas. Journal of Materials Science, 2022, 57(39): 18287.
DOI |
| [30] |
DOS SANTOS V H J M, PONTIN D, PONZI G G D, et al. Application of Fourier transform infrared spectroscopy (FTIR) coupled with multivariate regression for calcium carbonate (CaCO3) quantification in cement. Construction and Building Materials, 2021, 313: 125413.
DOI URL |
| [31] |
YAN D J, ZHU Y P, ZHAO J X, et al. Synthesis and utilization of polyol-modified high specific surface area Ca(OH)2: an investigation. Environmental Science and Pollution Research International, 2024, 31(22): 32714.
DOI |
| [32] |
HUANG T K, QIN Y L, LI M C, et al. Preparation and characterization of deacetylated konjac glucomannan/pectin composite films crosslinked with calcium hydroxide. Journal of Polymer Research, 2022, 29(6): 238.
DOI |
| [33] |
ZHANG Y, TONG S R, GE M F, et al. The formation and growth of calcium sulfate crystals through oxidation of SO2 by O3 on size-resolved calcium carbonate. RSC Advances, 2018, 8(29): 16285.
DOI URL |
| [34] |
BÖKE H, AKKURT S, SERHAN Ö, et al. Quantification of CaCO3-CaSO3·0.5H2O-CaSO4·2H2O mixtures by FTIR analysis and its ANN model. Materials Letters, 2004, 58(5): 723.
DOI URL |
| [35] |
FU H B, WANG X, WU H B, et al. Heterogeneous uptake and oxidation of SO2 on iron oxides. The Journal of Physical Chemistry C, 2007, 111(16): 6077.
DOI URL |
| [36] |
ZHANG Y X, JIA Y. A facile solution approach for the synthesis of akaganéite (β-FeOOH) nanorods and their ion-exchange mechanism toward As(V) ions. Applied Surface Science, 2014, 290: 102.
DOI URL |
| [37] |
DING M, ROOSENDAAL S J, DE JONG B H W S, et al. XPS studies on the electronic structure of bonding between solid and solutes: adsorption of arsenate, chromate, phosphate, Pb2+, and Zn2+ ions on amorphous black ferric oxyhydroxide. Geochimica et Cosmochimica Acta, 2000, 64(7): 1209.
DOI URL |
| [38] | LI L L, CHU Y, LIU Y, et al. Template-free synthesis and photocatalytic properties of novel Fe2O3 hollow spheres. The Journal of Physical Chemistry C, 2007, 111(5): 212. |
| [1] | CHEN Xiaochen, WANG Yang, YANG Bin, WANG Min, A Bohan, WANG Man, ZHANG Lingxia. Ligand-hydroxylated UiO-66 for Enhanced Photothermally Catalytic VOCs Oxidation [J]. Journal of Inorganic Materials, 2026, 41(5): 663-672. |
| [2] | WANG Meng, CAO Leilei, GOU Wangyan, CHENG Yayi, ZHAN Qi, YUAN Menglei. Tandem Catalysis of CuNi Bimetallic MOFs Boosting Nitrate Reduction for Ammonia Production [J]. Journal of Inorganic Materials, 2026, 41(5): 628-636. |
| [3] | ZHANG Mengjie, LI Zhibo, HUANG Ruinan, LÜ Xiangfei, WANG Wei. Cordierite/Aluminum Borate Whiskers/Co0.8FexCe0.2-xCr2O4 Catalysts: Preparation and Filtration Catalytic Performance for Carbon Soot [J]. Journal of Inorganic Materials, 2026, 41(4): 509-518. |
| [4] | TIAN Hongwang, LUO Longfei, HU Chenglong, YAN Meng, PANG Shengyang, LI Jian, TANG Sufang. Facile Fabrication of Ceramic-resin Coatings on C/CA Composites for Oxidation Protection at Medium Temperatures [J]. Journal of Inorganic Materials, 2026, 41(3): 401-408. |
| [5] | WEI Lianjin, QI Zhijie, WANG Xin, ZHU Junwu, FU Yongsheng. Modification of Nanodiamond and Its Application in Electrocatalytic Oxygen Reduction Reaction [J]. Journal of Inorganic Materials, 2026, 41(3): 273-288. |
| [6] | QI Fang, LIU Hui, WU Zhengmin, LU Yi, WU Wenwen, WANG Zhen. Matrix/Interface/Fiber Integrated Oxidation Mechanism of Mini-SiCf/BN/SiC in Water-oxygen Environment at 1100 ℃ [J]. Journal of Inorganic Materials, 2026, 41(3): 340-348. |
| [7] | MA Xinchao, ZHI Qing, LI Wei, CHEN Mao, WANG Hailong, ZHANG Rui, ZHANG Fan, FAN Bingbing. High-temperature Oxidation Mechanism and Electromagnetic Wave Absorption Properties of Fe2AlB2 [J]. Journal of Inorganic Materials, 2026, 41(1): 45-54. |
| [8] | LIU Jiangping, GUAN Xin, TANG Zhenjie, ZHU Wenjie, LUO Yongming. Research Progress on Catalytic Oxidation of Nitrogen-containing Volatile Organic Compounds [J]. Journal of Inorganic Materials, 2025, 40(9): 933-943. |
| [9] | ZHU Wenjie, TANG Lu, LU Jichang, LIU Jiangping, LUO Yongming. Research Progress on Catalytic Oxidation of Volatile Organic Compounds by Perovskite Oxides [J]. Journal of Inorganic Materials, 2025, 40(7): 735-746. |
| [10] | FAN Xiaoxuan, ZHENG Yonggui, XU Lirong, YAO Zimin, CAO Shuo, WANG Kexin, WANG Jiwei. Organic Pollutant Fenton Degradation Driven by Self-activated Afterglow from Oxygen-vacancy-rich LiYScGeO4: Bi3+ Long Afterglow Phosphor [J]. Journal of Inorganic Materials, 2025, 40(5): 481-488. |
| [11] | LI Jianjun, CHEN Fangming, ZHANG Lili, WANG Lei, ZHANG Liting, CHEN Huiwen, XUE Changguo, XU Liangji. Peroxymonosulfate Activation by CoFe2O4/MgAl-LDH Catalyst for the Boosted Degradation of Antibiotic [J]. Journal of Inorganic Materials, 2025, 40(4): 440-448. |
| [12] | JIA Xianghua, ZHANG Huixia, LIU Yanfeng, ZUO Guihong. Cu2O/Cu Hollow Spherical Heterojunction Photocatalysts Prepared by Wet Chemical Approach [J]. Journal of Inorganic Materials, 2025, 40(4): 397-404. |
| [13] | MU Shuang, MA Qin, ZHANG Yu, SHEN Xu, YANG Jinshan, DONG Shaoming. Oxidation Behavior of Yb2Si2O7 Modified SiC/SiC Mini-composites [J]. Journal of Inorganic Materials, 2025, 40(3): 323-328. |
| [14] | LI Wei, XU Zhiming, GOU Yanzi, YIN Senhu, YU Yiping, WANG Song. Preparation and Performance of Sintered SiC Fiber-bonded Ceramics [J]. Journal of Inorganic Materials, 2025, 40(2): 177-183. |
| [15] | LI Xiaoxuan, FU Qiangang, WEN Zihao, YANG Jinshan, NI Dewei, ZHANG Jie, CHENG Yuan, LIU Yuxuan, CHU Yanhui, CAI Feiyan, WANG Jingyang, ZHANG Xinghong. Research Progress on Ultra-high Temperature Ceramic Structural Materials for Extreme Environments [J]. Journal of Inorganic Materials, 2025, 40(10): 1045-1078. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||