
Journal of Inorganic Materials ›› 2026, Vol. 41 ›› Issue (5): 663-672.DOI: 10.15541/jim20250157
Special Issue: 【能源环境】金属有机框架材料MOF(202512)
• RESEARCH LETTER • Previous Articles Next Articles
CHEN Xiaochen1,2( ), WANG Yang2, YANG Bin1, WANG Min2, A Bohan2, WANG Man2, ZHANG Lingxia1,2(
), WANG Yang2, YANG Bin1, WANG Min2, A Bohan2, WANG Man2, ZHANG Lingxia1,2( )
)
Received:2025-04-13
Revised:2025-06-26
Published:2025-07-31
Online:2025-07-31
Contact:
ZHANG Lingxia, professor. E-mail: zhlingxia@mail.sic.ac.cnAbout author:CHEN Xiaochen (1999-), male, Master candidate. E-mail: chenxiaochen22@mails.ucas.ac.cn
Supported by:CLC Number:
CHEN Xiaochen, WANG Yang, YANG Bin, WANG Min, A Bohan, WANG Man, ZHANG Lingxia. Ligand-hydroxylated UiO-66 for Enhanced Photothermally Catalytic VOCs Oxidation[J]. Journal of Inorganic Materials, 2026, 41(5): 663-672.
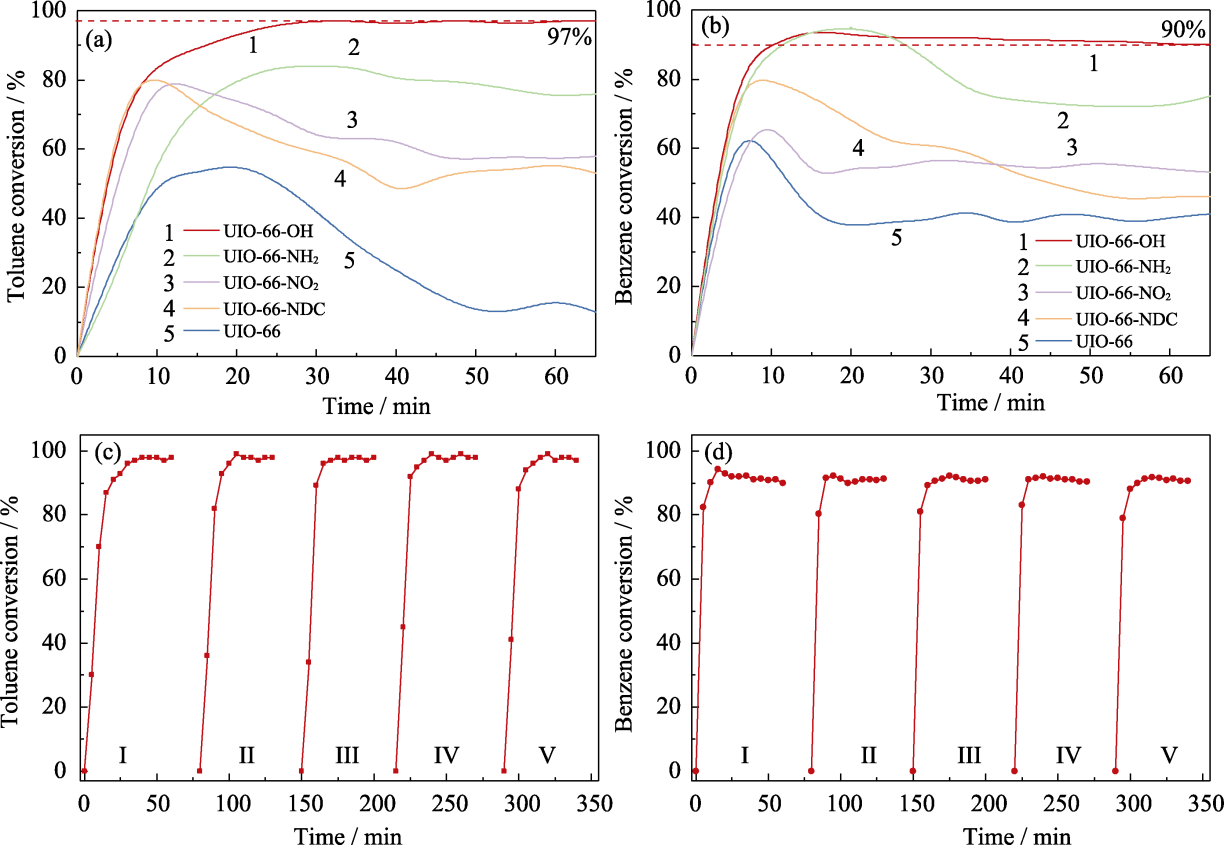
Fig. 2 (a, b) Photothermal catalytic performance of UiO-66-X and UiO-66 for oxidation of (a) toluene and (b) benzene, and (c, d) cycle stability of UiO-66-OH for photothermal catalytic oxidation of (c) toluene and (d) benzene
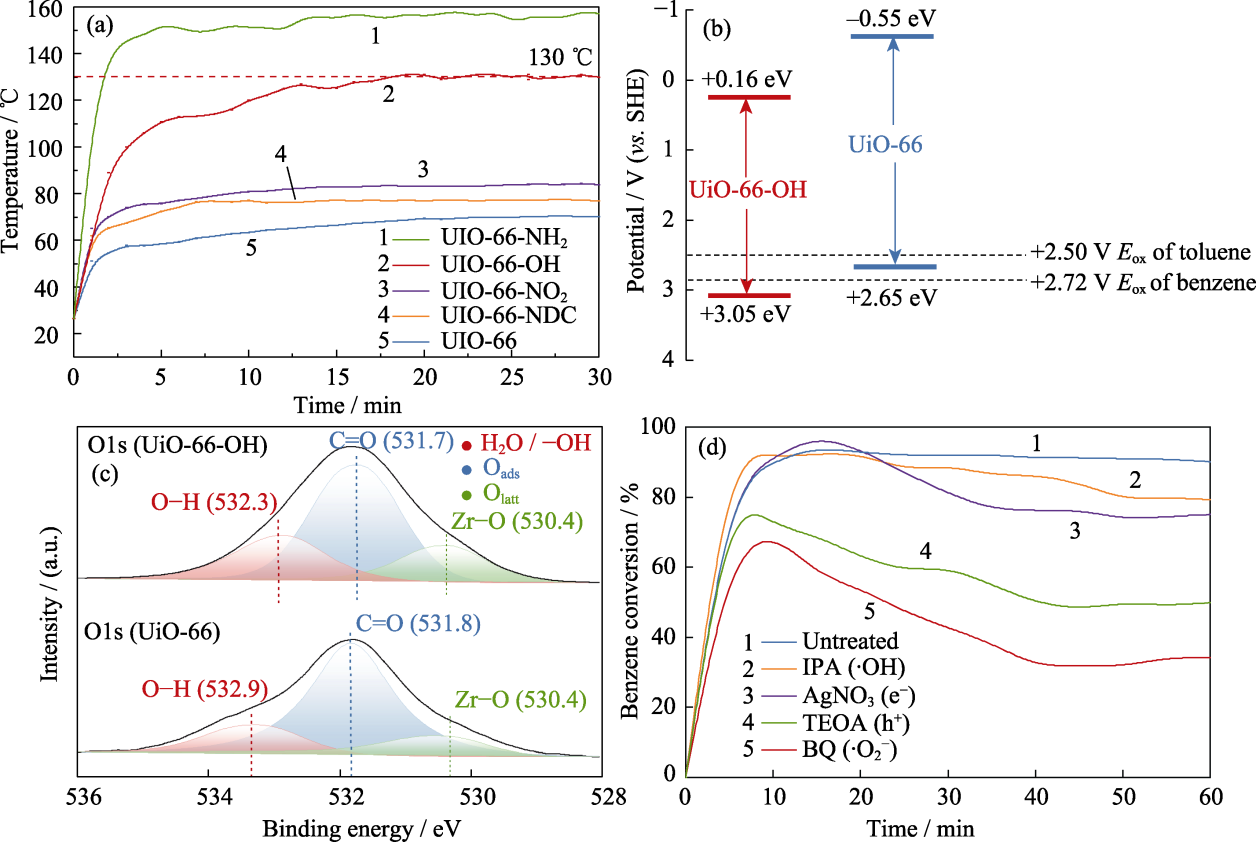
Fig. 3 (a) Surface temperature of the catalysts, (b) schematic band structure, (c) O1s XPS spectra, and (d) active species trapping experiments of UiO-66-OH
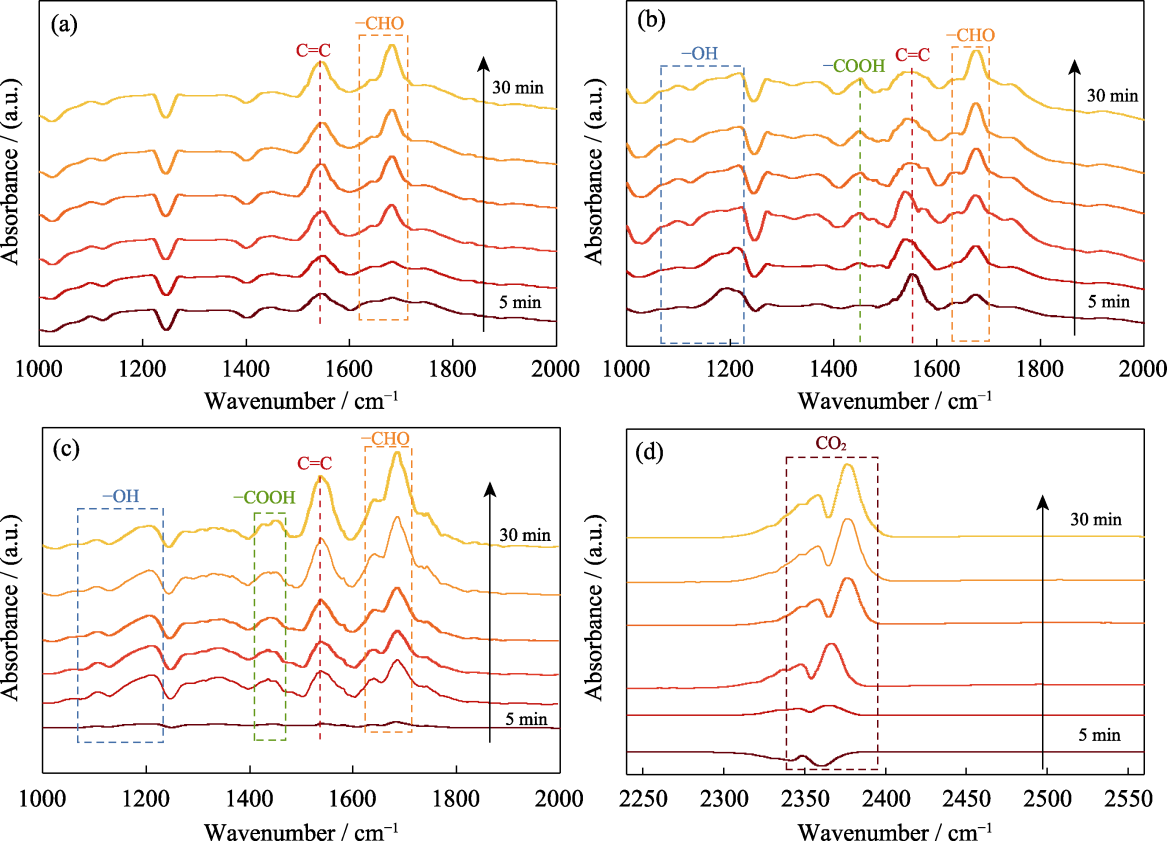
Fig. 4 In-situ FT-IR spectra of UiO-66-OH for oxidation of toluene under different successive conditions (a) In dark without O2; (b) In dark with O2; (c, d) With O2 and irradiation
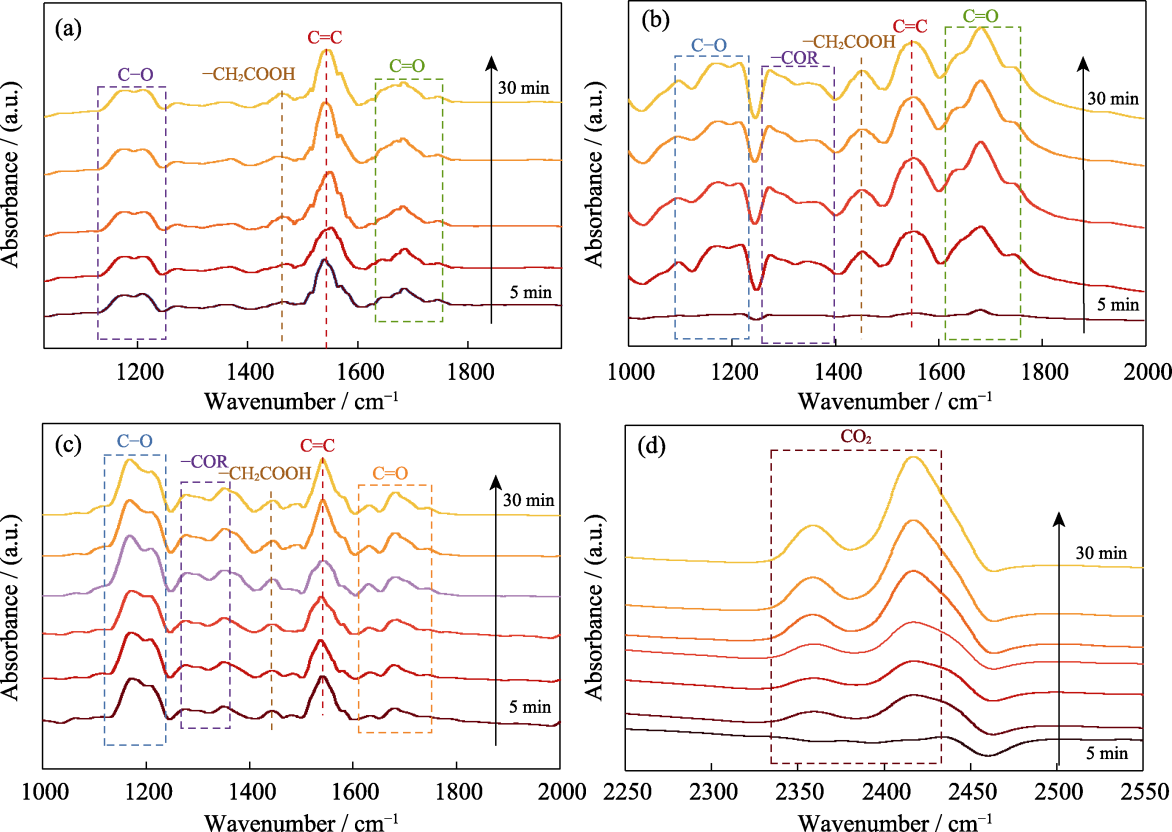
Fig. 5 In-situ FT-IR spectra of UiO-66-OH for oxidation of benzene under different successive conditions (a) In dark without O2; (b) In dark with O2; (c, d) With O2 and irradiation
| Sample | Temperature/℃ | Time/h |
|---|---|---|
| UiO-66 | 120 | 24 |
| UiO-66-NDC | 120 | 24 |
| UiO-66-NO2 | 120 | 24 |
| UiO-66-NH2 | 120 | 12 |
| UiO-66-OH | 80 | 12 |
Table S1 Hydrothermal conditions for the synthesis of UiO-66 and UiO-66-X catalysts
| Sample | Temperature/℃ | Time/h |
|---|---|---|
| UiO-66 | 120 | 24 |
| UiO-66-NDC | 120 | 24 |
| UiO-66-NO2 | 120 | 24 |
| UiO-66-NH2 | 120 | 12 |
| UiO-66-OH | 80 | 12 |
| Catalyst | VOC | Concentration/(mg∙L-1) | Catalyst amount/mg | Light intensity/(mW∙cm-2) | Conversion/% |
|---|---|---|---|---|---|
| Pt/TiO2[ | Benzene | 0.96 | 100 | 300 | 84.5 |
| Pt/g-C3N4[ | Benzene | 0.96 | 150 | 500 | 95 |
| This work | Benzene | 0.064 | 100 | 300 | 90 |
| MnOx-TiO2[ | Toluene | 0.02 | 500 | - | 72 |
| TiO2-UiO-66-NH2[ | Toluene | 0.094 | 100 | 50 | 73 |
| Pt/MnOx[ | Toluene | 0.75 | 100 | 200 | 90 |
| This work | Toluene | 0.075 | 100 | 300 | 97 |
Table S2 Performance comparison of relevant catalysts on benzene and toluene oxidation
| Catalyst | VOC | Concentration/(mg∙L-1) | Catalyst amount/mg | Light intensity/(mW∙cm-2) | Conversion/% |
|---|---|---|---|---|---|
| Pt/TiO2[ | Benzene | 0.96 | 100 | 300 | 84.5 |
| Pt/g-C3N4[ | Benzene | 0.96 | 150 | 500 | 95 |
| This work | Benzene | 0.064 | 100 | 300 | 90 |
| MnOx-TiO2[ | Toluene | 0.02 | 500 | - | 72 |
| TiO2-UiO-66-NH2[ | Toluene | 0.094 | 100 | 50 | 73 |
| Pt/MnOx[ | Toluene | 0.75 | 100 | 200 | 90 |
| This work | Toluene | 0.075 | 100 | 300 | 97 |
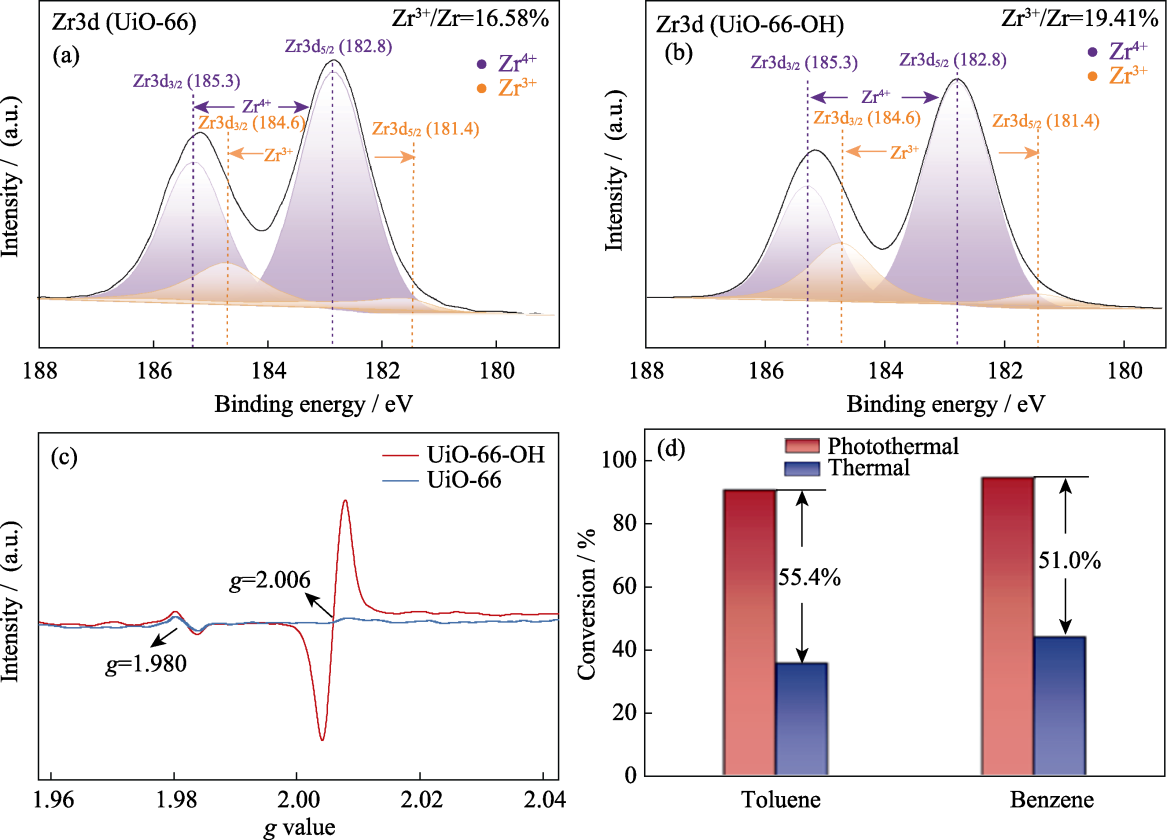
Fig. S6 (a, b) Zr3d XPS spectra of (a) UiO-66 and (b) UiO-66-OH, (c) EPR spectra of UiO-66 and UiO-66-OH, and(d) comparison of toluene and benzene conversion by photothermal catalysis and thermal catalysis
| [1] |
HE C, CHENG J, ZHANG X, et al. Recent advances in the catalytic oxidation of volatile organic compounds: a review based on pollutant sorts and sources. Chemical Reviews, 2019, 119(7): 4471.
DOI PMID |
| [2] |
MARINO E, CARUSO M, CAMPAGNA D, et al. Impact of air quality on lung health: myth or reality? Therapeutic Advances in Chronic Disease, 2015, 6(5): 286.
DOI PMID |
| [3] | HUANG H B, XU Y, FENG Q Y, et al. Low temperature catalytic oxidation of volatile organic compounds: a review. Catalysis Science & Technology, 2015, 5(5): 2649. |
| [4] |
ELIMIAN E A, ZHANG M, SUN Y, et al. Harnessing solar energy towards synergistic photothermal catalytic oxidation of volatile organic compounds. Solar RRL, 2023, 7(14): 2300238.
DOI URL |
| [5] | YANG Y, ZHAO S H, CUI L F, et al. Recent advancement and future challenges of photothermal catalysis for VOCs elimination: from catalyst design to applications. Green Energy & Environment, 2023, 8(3): 654. |
| [6] |
MA X L, WANG W L, SUN C G, et al. Adsorption performance and kinetic study of hierarchical porous Fe-based MOFs for toluene removal. Science of the Total Environment, 2021, 793: 148622.
DOI URL |
| [7] |
CHEN R F, YAO Z X, HAN N, et al. Insights into the adsorption of VOCs on a cobalt-adeninate metal-organic framework (bio-MOF-11). ACS Omega, 2020, 5(25): 15402.
DOI PMID |
| [8] |
LV S W, LIU J M, LI C Y, et al. Two novel MOFs@COFs hybrid-based photocatalytic platforms coupling with sulfate radical-involved advanced oxidation processes for enhanced degradation of bisphenol A. Chemosphere, 2020, 243: 125378.
DOI URL |
| [9] |
ZHANG J H, HU Y, QIN J X, et al. TiO2-UiO-66-NH2 nanocomposites as efficient photocatalysts for the oxidation of VOCs. Chemical Engineering Journal, 2020, 385: 123814.
DOI URL |
| [10] |
WU X P, GAGLIARDI L, TRUHLAR D G. Cerium metal-organic framework for photocatalysis. Journal of the American Chemical Society, 2018, 140(25): 7904.
DOI URL |
| [11] |
HEU R, ATEIA M, YOSHIMURA C. Photocatalytic nanofiltration membrane using Zr-MOF/GO nanocomposite with high-flux and anti-fouling properties. Catalysts, 2020, 10(6): 711.
DOI URL |
| [12] |
KIM H G, CHOI K, LEE K, et al. Controlling the structural robustness of zirconium-based metal organic frameworks for efficient adsorption on tetracycline antibiotics. Water, 2021, 13(13): 1869.
DOI URL |
| [13] |
NAIK T S S K, SINGH S, PAVITHRA N, et al. Advanced experimental techniques for the sensitive detection of a toxic bisphenol A using UiO-66-NDC/GO-based electrochemical sensor. Chemosphere, 2023, 311: 137104.
DOI URL |
| [14] |
MA Y L, LI A R, WANG Z H, et al. Preparation of UiO-66-type adsorbents for the separation of SO2 from flue gas. Adsorption, 2024, 30(3): 377.
DOI |
| [15] |
LUCATERO E, BASHIRI R, SO M C. Synthesis, characterization, and evaluation of metal-organic frameworks for oxidative desulfurization: an integrated experiment. Journal of Chemical Education, 2024, 101(8): 3428.
DOI URL |
| [16] |
YOU X Q, LI Y, MO H R, et al. Theoretical studies on Lennard-Jones parameters of benzene and polycyclic aromatic hydrocarbons. Faraday Discussions, 2022, 238: 103.
DOI PMID |
| [17] |
ZHAO Y, WANG Y, WANG X Y, et al. Recent progress of photothermal therapy based on conjugated nanomaterials in combating microbial infections. Nanomaterials, 2023, 13(15): 2269.
DOI URL |
| [18] |
MOGHADDAM Z S, KAYKHAII M, KHAJEH M, et al. Synthesis of UiO-66-OH zirconium metal-organic framework and its application for selective extraction and trace determination of thorium in water samples by spectrophotometry. Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2018, 194: 76.
DOI URL |
| [19] |
NGUYEN V H, PHAM A L H, NGUYEN V H, et al. Facile synthesis of bismuth(III) based metal-organic framework with difference ligands using microwave irradiation method. Chemical Engineering Research and Design, 2022, 177: 321.
DOI URL |
| [20] |
WRIGHT S, BARKLIE R C. EPR characterization of defects in monoclinic powders of ZrO2 and HfO2. Materials Science in Semiconductor Processing, 2006, 9(6): 892.
DOI URL |
| [21] | ZHANG Y T, CHENG C, ZHOU Z H, et al. Surface hydroxylation during water splitting promotes the photoactivity of BiVO4(010) surface by suppressing polaron-mediated charge recombination. 2023, 14(40): 9096. |
| [22] |
WU H, CHUA Y S, KRUNGLEVICIUTE V, et al. Unusual and highly tunable missing-linker defects in zirconium metal-organic framework UiO-66 and their important effects on gas adsorption. Journal of the American Chemical Society, 2013, 135(28): 10525.
DOI PMID |
| [23] |
BAKRADZE G, JEURGENS L P H, MITTEMEIJER E J. Valence-band and chemical-state analyses of Zr and O in thermally grown thin zirconium-oxide films: an XPS study. The Journal of Physical Chemistry C, 2011, 115(40): 19841.
DOI URL |
| [24] |
CHI M Y, SUN X N, SUJAN A, et al. A quantitative XPS examination of UV induced surface modification of TiO2 sorbents for the increased saturation capacity of sulfur heterocycles. Fuel, 2019, 238: 454.
DOI URL |
| [25] |
JIANG Y, NING H Y, TIAN C G, et al. Single-crystal TiO2 nanorods assembly for efficient and stable cocatalyst-free photocatalytic hydrogen evolution. Applied Catalysis B: Environmental, 2018, 229: 1.
DOI URL |
| [26] |
LIU L Z, HUANG H W, CHEN F, et al. Cooperation of oxygen vacancies and 2D ultrathin structure promoting CO2 photoreduction performance of Bi4Ti3O12. Science Bulletin, 2020, 65(11): 934.
DOI URL |
| [27] |
IBRAHIM D A H, HAIKAL D R R, ELDIN R S, et al. The role of free-radical pathway in catalytic dye degradation by hydrogen peroxide on the Zr-based UiO-66-NH2 MOF. ChemistrySelect, 2021, 6(42): 11675.
DOI URL |
| [28] | MERKEL P B, LUO P, DINNOCENZO J P, et al. Accurate oxidation potentials of benzene and biphenyl derivatives via electron- transfer equilibria and transient kinetics. 2009, 74(15): 5163. |
| [29] |
LIN H X, LONG J L, GU Q, et al. In situ IR study of surface hydroxyl species of dehydrated TiO2: towards understanding pivotal surface processes of TiO2 photocatalytic oxidation of toluene. Physical Chemistry Chemical Physics, 2012, 14(26): 9468.
DOI URL |
| [30] | WANG F Z, LI W J, GU S N, et al. Fabrication of FeWO4@ZnWO4/ZnO heterojunction photocatalyst: synergistic effect of ZnWO4/ZnO and FeWO4@ZnWO4/ZnO heterojunction structure on the enhancement of visible-light photocatalytic activity. ACS Sustainable Chemistry & Engineering, 2016, 4(12): 6288. |
| [31] |
MO S P, LI J, LIAO R Q, et al. Unraveling the decisive role of surface CeO2 nanoparticles in the Pt-CeO2/MnO2 hetero-catalysts for boosting toluene oxidation: synergistic effect of surface decorated and intrinsic O-vacancies. Chemical Engineering Journal, 2021, 418: 129399.
DOI URL |
| [32] |
MO S P, ZHANG Q, LI J Q, et al. Highly efficient mesoporous MnO2 catalysts for the total toluene oxidation: oxygen-vacancy defect engineering and involved intermediates using in situ DRIFTS. Applied Catalysis B: Environmental, 2020, 264: 118464.
DOI URL |
| [33] | CHEN L C, CHEN P, WANG H, et al. Surface lattice oxygen activation on Sr2Sb2O7 enhances the photocatalytic mineralization of toluene: from reactant activation, intermediate conversion to product desorption. ACS Applied Materials & Interfaces, 2021, 13(4): 5153. |
| [1] | ZHU Kaihuang, YANG Shijie, LI Xinge, SONG Guanqing, SHI Gansheng, WANG Yan, REN Xiaomeng, LU Yao, XU Xinhong, SUN Jing. Graphene Oxide Modified UiO-66 Based Metal Organic Framework Gel: Preparation and Efficient Toluene Adsorption Performance [J]. Journal of Inorganic Materials, 2026, 41(4): 519-526. |
| [2] | LIU Jiangping, GUAN Xin, TANG Zhenjie, ZHU Wenjie, LUO Yongming. Research Progress on Catalytic Oxidation of Nitrogen-containing Volatile Organic Compounds [J]. Journal of Inorganic Materials, 2025, 40(9): 933-943. |
| [3] | ZHU Wenjie, TANG Lu, LU Jichang, LIU Jiangping, LUO Yongming. Research Progress on Catalytic Oxidation of Volatile Organic Compounds by Perovskite Oxides [J]. Journal of Inorganic Materials, 2025, 40(7): 735-746. |
| [4] | JIANG Zongyu, HUANG Honghua, QING Jiang, WANG Hongning, YAO Chao, CHEN Ruoyu. Aluminum Ion Doped MIL-101(Cr): Preparation and VOCs Adsorption Performance [J]. Journal of Inorganic Materials, 2025, 40(7): 747-753. |
| [5] | QU Mujing, ZHANG Shulan, ZHU Mengmeng, DING Haojie, DUAN Jiaxin, DAI Henglong, ZHOU Guohong, LI Huili. CsPbBr3@MIL-53 Nanocomposite Phosphors: Synthesis, Properties and Applications in White LEDs [J]. Journal of Inorganic Materials, 2024, 39(9): 1035-1043. |
| [6] | MA Xiaosen, ZHANG Lichen, LIU Yanchao, WANG Quanhua, ZHENG Jiajun, LI Ruifeng. 13X@SiO2: Synthesis and Toluene Adsorption [J]. Journal of Inorganic Materials, 2023, 38(5): 537-543. |
| [7] | JIANG Runlu, WU Xin, GUO Haocheng, ZHENG Qi, WANG Lianjun, JIANG Wan. UiO-67 Based Conductive Composites: Preparation and Thermoelectric Performance [J]. Journal of Inorganic Materials, 2023, 38(11): 1338-1344. |
| [8] | SUN Chen, ZHAO Kunfeng, YI Zhiguo. Research Progress in Catalytic Total Oxidation of Methane [J]. Journal of Inorganic Materials, 2023, 38(11): 1245-1256. |
| [9] | WANG Xiao, ZHU Zhijie, WU Zhiyi, ZHANG Chengcheng, CHEN Zhijie, XIAO Mengqi, LI Chaoran, HE Le. Preparation and Photothermal Catalytic Application of Powder-form Cobalt Plasmonic Superstructures [J]. Journal of Inorganic Materials, 2022, 37(1): 22-28. |
| [10] | LIANG Fengqing, WEN Zhaoyin. MOF/Poly(Ethylene Oxide) Composite Polymer Electrolyte for Solid-state Lithium Battery [J]. Journal of Inorganic Materials, 2021, 36(3): 332-336. |
| [11] | WANG Yuwei, CHEN Jiajie, TIAN Zhengfang, ZHU Min, ZHU Yufang. Potassium Ferrate-loaded Porphyrin-based (VI) Metal-organic Frameworks for Combined Photodymanic and Chemodynamic Tumor Therapy [J]. Journal of Inorganic Materials, 2021, 36(12): 1305-1315. |
| [12] | ZHANG Xincong,GUO Ke,PENG Lianlian,WU Jieyu,ZHANG Fumin,ZHU Weidong,FU Yanghe. Degradation of Dye Wastewater over NH2-UiO-66: Piezoelectrically Induced Mechano-Catalytic Effect [J]. Journal of Inorganic Materials, 2020, 35(9): 1023-1028. |
| [13] | LI Si-Han, ZHANG Chao, WU Chen-Liang, ZHANG He-Feng, YAN Xin-Huan. Pd/CeO2/γ-Al2O3 Catalyst with Low Loading for Catalytic Oxidation of VOCs [J]. Journal of Inorganic Materials, 2019, 34(8): 827-833. |
| [14] | Shi-Qiang LUO, Chun-Man ZHENG, Wei-Wei SUN, Wei XIE, Jian-Huang KE, Shuang-Ke LIU, Xiao-Bin HONG, Yu-Jie LI, Jing XU. Controllable Preparation of Co-NC Nanoporous Carbon Derived from ZIF-67 for Advanced Lithium-sulfur Batteries [J]. Journal of Inorganic Materials, 2019, 34(5): 502-508. |
| [15] | GONG Yun, LIU Yan, GU Ping, ZHOU Xiao-Xia. Synthesis of Nano Manganese Oxide with Assistance of Ultrasonic for Removal of Low Concentration NO [J]. Journal of Inorganic Materials, 2019, 34(2): 186-192. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||