无机材料学报 ›› 2026, Vol. 41 ›› Issue (5): 637-644.DOI: 10.15541/jim20250348 CSTR: 32189.14.10.15541/jim20250348
收稿日期:2025-08-31
修回日期:2025-11-09
出版日期:2025-11-26
网络出版日期:2025-11-26
通讯作者:
姜海波, 副研究员. E-mail: jianghaibo@ecust.edu.cn作者简介:李中意(2001-), 男, 硕士研究生. E-mail: 2911277205@qq.com
基金资助:
LI Zhongyi( ), LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo(
), LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo( )
)
Received:2025-08-31
Revised:2025-11-09
Published:2025-11-26
Online:2025-11-26
Contact:
JIANG Haibo, associate professor. E-mail: jianghaibo@ecust.edu.cnAbout author:LI Zhongyi (2001-), male, Master candidate. E-mail: 2911277205@qq.com
Supported by:摘要:
二氧化硫(SO2)是一种常见的大气污染物, 对环境和人体健康造成严重危害。目前, 氢氧化钙(Ca(OH)2)作为一种常用的脱硫剂, 因其制备简单、成本低廉且脱硫效果较好而被广泛应用。然而, 如何进一步提高其固硫效率仍是研究重点。本工作在氧化钙(CaO)消化过程中加入氢氧化铁(Fe(OH)3), 分步制备了钙铁脱硫剂, 并研究了其脱硫性能。结果表明, 钙铁脱硫剂的固硫效率显著提高, 与纯Ca(OH)2相比, 固硫效率最大提升了26.16%。加入Fe3+改变了Ca(OH)2的形貌, 使其表面更加粗糙, 增加了孔隙和裂缝, 从而提供了更多的反应活性位点。此外, 根据X射线光电子能谱测试结果, 脱硫后样品中S6+的比例从9.71%(纯Ca(OH)2)提高到33.33%(钙铁脱硫剂), 并且脱硫前钙铁脱硫剂中均是Fe3+, 脱硫后Fe2+和Fe3+分别占68.42%和31.58%, 其中35.56%的硫元素被氧化, 64.44%的硫元素被催化, 说明Fe3+的氧化以及催化作用促进了S4+向S6+转化, 进一步提高了固硫效率。本工作为提高Ca(OH)2固硫效率提供了一种有效的方法, 对选择工业烟气SO2脱除材料具有重要的借鉴意义。
中图分类号:
李中意, 刘彪, 陈茜, 李春忠, 姜海波. Fe3+增强氢氧化钙固硫效率: 氧化、催化双重机制[J]. 无机材料学报, 2026, 41(5): 637-644.
LI Zhongyi, LIU Biao, CHEN Xi, LI Chunzhong, JIANG Haibo. Enhanced Sulfur Fixation Efficiency of Calcium Hydroxide by Fe3+: Dual Mechanisms of Oxidation and Catalysis[J]. Journal of Inorganic Materials, 2026, 41(5): 637-644.
| CaO/g | Deionized water/g | H2O/CaO |
|---|---|---|
| 10 | 6 | 0.6 |
| 10 | 7 | 0.7 |
| 10 | 8 | 0.8 |
| 10 | 9 | 0.9 |
| 10 | 10 | 1.0 |
| 10 | 11 | 1.1 |
表1 纯Ca(OH)2的制备
Table 1 Preparation of pure calcium hydroxide
| CaO/g | Deionized water/g | H2O/CaO |
|---|---|---|
| 10 | 6 | 0.6 |
| 10 | 7 | 0.7 |
| 10 | 8 | 0.8 |
| 10 | 9 | 0.9 |
| 10 | 10 | 1.0 |
| 10 | 11 | 1.1 |
| CaO/g | Deionized water/g | Fe(OH)3/g | H2O/CaO |
|---|---|---|---|
| 10 | 6 | 6.25 | 0.6 |
| 10 | 7 | 6.25 | 0.7 |
| 10 | 8 | 6.25 | 0.8 |
| 10 | 9 | 6.25 | 0.9 |
| 10 | 10 | 6.25 | 1.0 |
| 10 | 11 | 6.25 | 1.1 |
表2 钙铁脱硫剂的制备
Table 2 Preparation of calcium-iron desulfurizer
| CaO/g | Deionized water/g | Fe(OH)3/g | H2O/CaO |
|---|---|---|---|
| 10 | 6 | 6.25 | 0.6 |
| 10 | 7 | 6.25 | 0.7 |
| 10 | 8 | 6.25 | 0.8 |
| 10 | 9 | 6.25 | 0.9 |
| 10 | 10 | 6.25 | 1.0 |
| 10 | 11 | 6.25 | 1.1 |
| Composition | Content/% (in mass) | Composition | Content/% (in mass) |
|---|---|---|---|
| CaO | 96.05 | SO3 | 0.16 |
| MgO | 2.1 | K2O | 0.086 |
| Al2O3 | 0.33 | Fe2O3 | 0.426 |
| SiO2 | 0.757 | SrO | 0.09 |
表3 生石灰样品物质组成分析
Table 3 Composition analysis of quicklime sample
| Composition | Content/% (in mass) | Composition | Content/% (in mass) |
|---|---|---|---|
| CaO | 96.05 | SO3 | 0.16 |
| MgO | 2.1 | K2O | 0.086 |
| Al2O3 | 0.33 | Fe2O3 | 0.426 |
| SiO2 | 0.757 | SrO | 0.09 |
| H2O/CaO | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) |
|---|---|---|
| 0.6 | 25.596 | 0.116 |
| 0.7 | 25.419 | 0.122 |
| 0.8 | 27.256 | 0.130 |
| 0.9 | 26.214 | 0.127 |
| 1.0 | 26.646 | 0.135 |
| 1.1 | 31.694 | 0.182 |
表4 钙铁脱硫剂的比表面积和孔容
Table 4 Specific surface areas and pore volumes of calcium-iron desulfurizers
| H2O/CaO | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) |
|---|---|---|
| 0.6 | 25.596 | 0.116 |
| 0.7 | 25.419 | 0.122 |
| 0.8 | 27.256 | 0.130 |
| 0.9 | 26.214 | 0.127 |
| 1.0 | 26.646 | 0.135 |
| 1.1 | 31.694 | 0.182 |
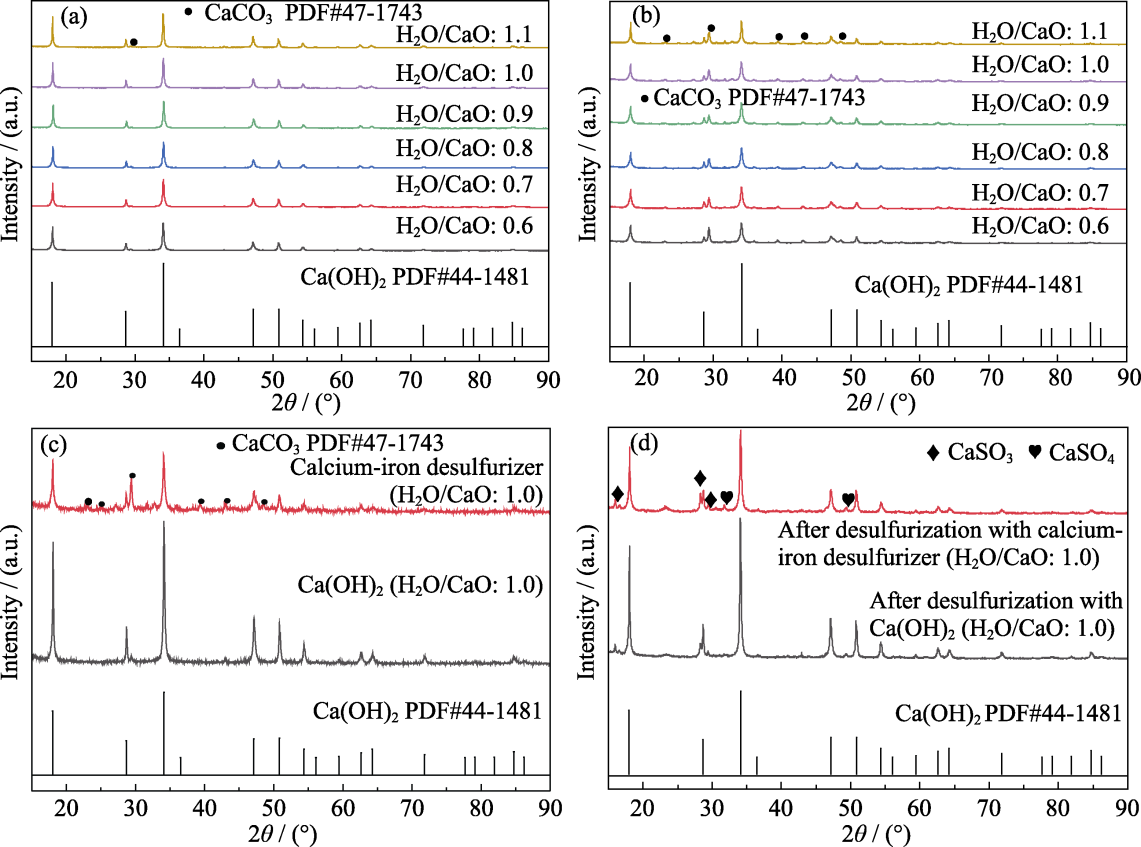
图4 脱硫剂脱硫前后的XRD图谱
Fig. 4 XRD patterns of desulfurizers before and after desulfurization (a) Pure Ca(OH)2 with water cement ratio of 0.6-1.1; (b) Calcium-iron desulfurizers with water cement ratio of 0.6-1.1; (c, d) Comparison of pure Ca(OH)2 and calcium-iron desulfurizer with water cement ratio of 1.0 before (c) and after (d) desulfurization
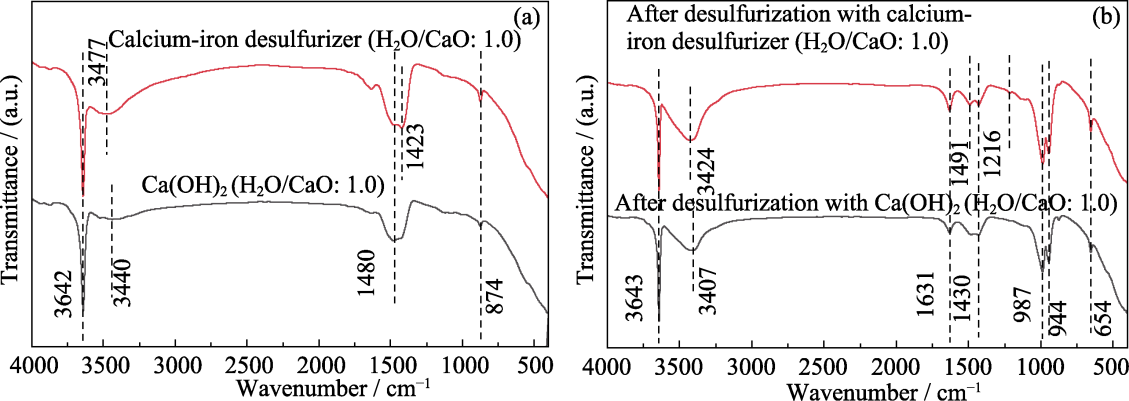
图5 水灰比1.0的纯Ca(OH)2和钙铁脱硫剂脱硫前(a)后(b)的FT-IR图谱
Fig. 5 FT-IR spectra of pure Ca(OH)2 and calcium-iron desulfurizer with water cement ratio of 1.0 before (a) and after (b) desulfurization
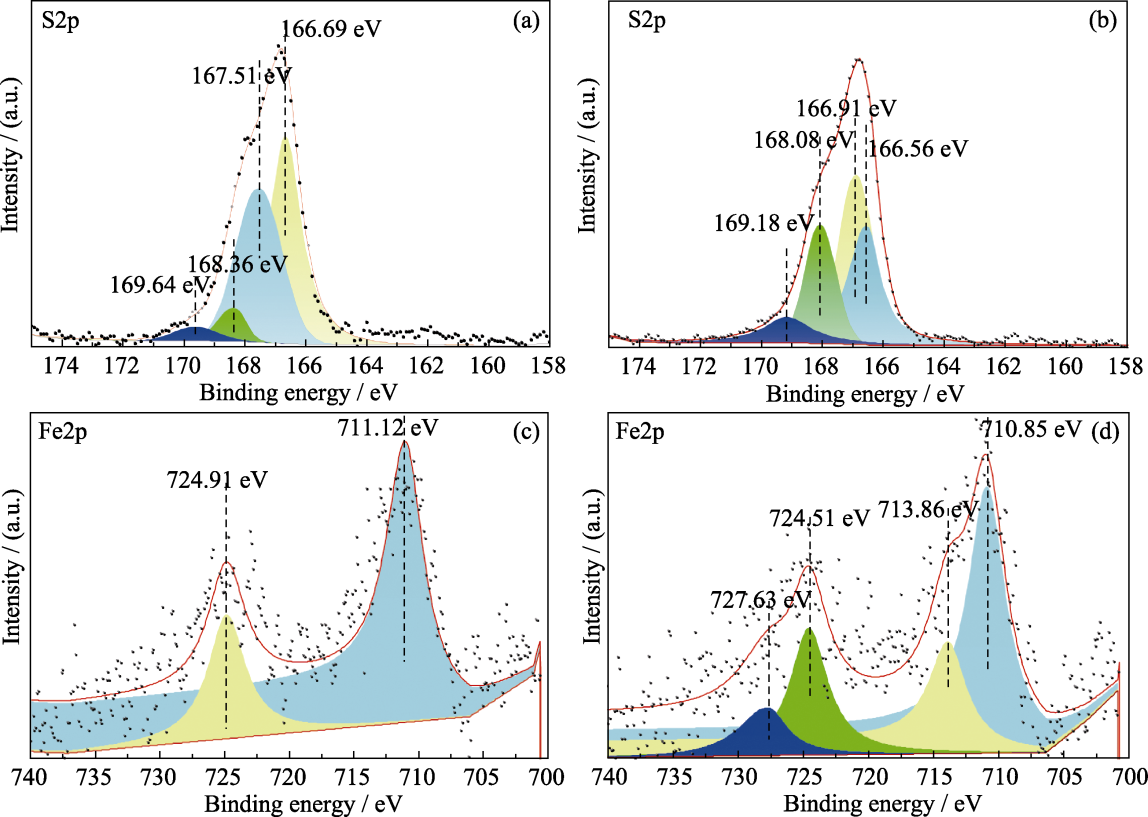
图6 水灰比1.0的纯Ca(OH)2和钙铁脱硫剂脱硫前后的XPS图谱
Fig. 6 XPS spectra of pure Ca(OH)2 and calcium-iron desulfurizer with water cement ratio of 1.0 before and after desulfurization (a) S2p of pure Ca(OH)2 after desulfurization; (b) S2p of calcium-iron desulfurizer after desulfurization; (c) Fe2p of calcium-iron desulfurizer before desulfurization; (d) Fe2p of calcium-iron desulfurizer after desulfurization
| Desulfurizer | Sulfur fixation efficiency/% |
|---|---|
| Calcium-iron desulfurizer | 17.07 |
| Yan et al.[ | 1.95 |
| Li et al.[ | 6.4 |
表5 钙铁脱硫剂与其他脱硫剂固硫效率比较
Table 5 Comparison of sulfur fixation efficiency between calcium-iron desulfurizer and other desulfurizers
| Desulfurizer | Sulfur fixation efficiency/% |
|---|---|
| Calcium-iron desulfurizer | 17.07 |
| Yan et al.[ | 1.95 |
| Li et al.[ | 6.4 |
| [1] |
LUM M M X, NG K H, LAI S Y, et al. Sulfur dioxide catalytic reduction for environmental sustainability and circular economy: a review. Process Safety and Environmental Protection, 2023, 176: 580.
DOI URL |
| [2] |
CHEN W C, XIANG Y H, LIU J, et al. Foreign investor and industrial pollution: evidence from sulfur dioxide emission. Finance Research Letters, 2022, 50: 103279.
DOI URL |
| [3] |
YANG C H, CHEN P H, YANG C S, et al. Analysis and forecasting of air pollution on nitrogen dioxide and sulfur dioxide using deep learning. IEEE Access, 2024, 12: 165236.
DOI URL |
| [4] |
ZHANG J Y. The mechanism of foreign direct investment on China’s sulfur dioxide emissions: evidence from partial least squares structural equation modeling. Journal of Environmental Management, 2025, 380: 124998.
DOI URL |
| [5] |
QIAN Y, CAO H, HUANG S M. Decoupling and decomposition analysis of industrial sulfur dioxide emissions from the industrial economy in 30 Chinese provinces. Journal of Environmental Management, 2020, 260: 110142.
DOI URL |
| [6] |
LI Y, GAO L H, ZHANG J H, et al. Synergetic utilization of microwave-assisted fly ash and carbide slag for simultaneous desulfurization and denitrification: high efficiency, low cost and catalytic mechanism. Chemical Engineering Journal, 2022, 437: 135488.
DOI URL |
| [7] |
DAI G F, ZHANG J Y, WANG X B, et al. Calcination and desulfurization characteristics of calcium carbonate in pressurized oxy-combustion. Energy, 2022, 261: 125150.
DOI URL |
| [8] |
KUMAR A. Desulfurization performance of sulfur dioxide and product characteristics in semi-batch bubble column and foam-bed contactor. Chemical Papers, 2019, 74: 2427.
DOI |
| [9] |
OSAKA Y, TSUIGUCHI T, KODAMA A, et al. Improvement of dry desulfurization performance using activated calcium carbonate by amorphous citric acid complex method for diesel gas purification. Journal of Material Cycles and Waste Management, 2020, 22: 470.
DOI |
| [10] |
SHIN H G, KIM H, NOH T, et al. Degradation behavior of high surface area calcium hydroxide sorbent for SO2 removal. International Journal of Mineral Processing, 2011, 98(3/4): 145.
DOI URL |
| [11] |
YAN D J, HE R N, WU X Y, et al. An investigation of the surface structure of surfactants modified calcium hydroxide and enhancement of dry flue gas desulfurization performance. Research on Chemical Intermediates, 2025, 51(3): 1583.
DOI |
| [12] | SHIN H G, KIM H, KIM Y N, et al. Preparation and characterization of high surface area calcium hydroxide sorbent for SO2 removal. Current Applied Physics, 2009, 9(3): S276. |
| [13] |
LIU M, DING W X, CHANG J Y, et al. Pilot study on coke oven flue gas injection desulfurization using highly active modified calcium hydroxide desulfurizer. Asia-Pacific Journal of Chemical Engineering, 2025, 20(2): e3170.
DOI URL |
| [14] | 熊爽, 严金生, 周洲, 等. 生石灰消化反应条件对氢氧化钙特性影响. 无机盐工业, 2023, 55(12): 50. |
| [15] | 刘越, 郑强, 邢佳斌, 等. 石灰石干法制备高性能氢氧化钙的工艺及应用研究. 无机盐工业, 2023, 55(10): 42. |
| [16] |
RENEDO M J, FERNÁNDEZ-FERRERAS J. Characterization and behavior of modified calcium-hydroxide-based sorbents in a dry desulfurization process. Energy & Fuels, 2016, 30(8): 6350.
DOI URL |
| [17] |
LI Y R, QI H Y, YOU C F, et al. Kinetic model of CaO/fly ash sorbent for flue gas desulphurization at moderate temperatures. Fuel, 2007, 86(5/6): 785.
DOI URL |
| [18] |
FANG D X, HUANG L P, FANG Z Y, et al. Evaluation of porous calcium silicate hydrate derived from carbide slag for removing phosphate from wastewater. Chemical Engineering Journal, 2018, 354: 1.
DOI URL |
| [19] | 李春花, 苏春华, 王超, 等. 草木灰促进CaO粉末高温烟气脱硫实验研究. 应用化工, 2010, 39(4): 521. |
| [20] |
SALVITTI C, ROSI M, PEPI F, et al. Reactivity of transition metal dioxide anions MO2- (M = Co, Ni, Cu, Zn) with sulfur dioxide in the gas phase: an experimental and theoretical study. Chemical Physics Letters, 2021, 776: 138555.
DOI URL |
| [21] | 高翔, 骆仲泱, 陈亚非, 等. 钙基吸收剂脱硫反应特性的研究. 燃烧科学与技术, 1998, 4(4): 369. |
| [22] |
SU M H, YANG L C. Fe/Mn-MOFs with monocarboxylic acid-induced defects enhances the catalytic oxidation of calcium sulfite in desulfurization ash. Separation and Purification Technology, 2024, 353: 128300.
DOI URL |
| [23] |
LATYPOVA S, ESEVA E, LEVIN I, et al. Catalytic performance of transition metal molybdates in accelerated oxidative desulfurization. Fuel, 2025, 403: 136114.
DOI URL |
| [24] |
CHEN X Y, ZHOU S R, WANG L Y, et al. Facile preparation of Fe-Beta zeolite-supported transition metal oxide catalysts and their catalytic performance for the simultaneous removal of NOx and soot. Chinese Journal of Chemical Engineering, 2024, 76: 10.
DOI URL |
| [25] |
KIM H M, JEONG C H, CHEON B S, et al. Improving the performance of a Co-CeO2 catalyst for hydrogen production via water gas shift reaction by addition of transition metal oxides. Energy & Fuels, 2024, 38(5): 4743.
DOI URL |
| [26] |
WU H D, YU B, Ni C S, et al. Enhancement of catalytic performance in Zr0.1Ce0.9O2-δ through transition metal doping and exsolution. Solid State Ionics, 2025, 425: 116868.
DOI URL |
| [27] | SUN B, WANG J G, CHEN M, et al. Boosting acetone oxidation performance over mesocrystal MxCe1-xO2 (M = Ni, Cu, Zn) solid solution within hollow spheres by tailoring transition-metal cations. International Materials Chemistry and Physics, 2023, 293: 126925. |
| [28] |
HE K J, SONG Q, YAN Z N, et al. Study on competitive absorption of SO3 and SO2 by calcium hydroxide. Fuel, 2019, 242: 355.
DOI URL |
| [29] |
MA X Y, WU H, CHANG L, et al. The influence of calcium hydroxide crystal morphology on the desulfurization of cement kiln flue gas. Journal of Materials Science, 2022, 57(39): 18287.
DOI |
| [30] |
DOS SANTOS V H J M, PONTIN D, PONZI G G D, et al. Application of Fourier transform infrared spectroscopy (FTIR) coupled with multivariate regression for calcium carbonate (CaCO3) quantification in cement. Construction and Building Materials, 2021, 313: 125413.
DOI URL |
| [31] |
YAN D J, ZHU Y P, ZHAO J X, et al. Synthesis and utilization of polyol-modified high specific surface area Ca(OH)2: an investigation. Environmental Science and Pollution Research International, 2024, 31(22): 32714.
DOI |
| [32] |
HUANG T K, QIN Y L, LI M C, et al. Preparation and characterization of deacetylated konjac glucomannan/pectin composite films crosslinked with calcium hydroxide. Journal of Polymer Research, 2022, 29(6): 238.
DOI |
| [33] |
ZHANG Y, TONG S R, GE M F, et al. The formation and growth of calcium sulfate crystals through oxidation of SO2 by O3 on size-resolved calcium carbonate. RSC Advances, 2018, 8(29): 16285.
DOI URL |
| [34] |
BÖKE H, AKKURT S, SERHAN Ö, et al. Quantification of CaCO3-CaSO3·0.5H2O-CaSO4·2H2O mixtures by FTIR analysis and its ANN model. Materials Letters, 2004, 58(5): 723.
DOI URL |
| [35] |
FU H B, WANG X, WU H B, et al. Heterogeneous uptake and oxidation of SO2 on iron oxides. The Journal of Physical Chemistry C, 2007, 111(16): 6077.
DOI URL |
| [36] |
ZHANG Y X, JIA Y. A facile solution approach for the synthesis of akaganéite (β-FeOOH) nanorods and their ion-exchange mechanism toward As(V) ions. Applied Surface Science, 2014, 290: 102.
DOI URL |
| [37] |
DING M, ROOSENDAAL S J, DE JONG B H W S, et al. XPS studies on the electronic structure of bonding between solid and solutes: adsorption of arsenate, chromate, phosphate, Pb2+, and Zn2+ ions on amorphous black ferric oxyhydroxide. Geochimica et Cosmochimica Acta, 2000, 64(7): 1209.
DOI URL |
| [38] | LI L L, CHU Y, LIU Y, et al. Template-free synthesis and photocatalytic properties of novel Fe2O3 hollow spheres. The Journal of Physical Chemistry C, 2007, 111(5): 212. |
| [1] | 陈笑晨, 王阳, 杨彬, 王敏, 阿博涵, 王蔓, 张玲霞. 配体羟基改性增强UiO-66的光热催化氧化VOCs性能[J]. 无机材料学报, 2026, 41(5): 663-672. |
| [2] | 王萌, 曹磊磊, 苟王燕, 程娅伊, 战琪, 原孟磊. CuNi双金属MOFs串联催化促进硝酸盐还原合成氨[J]. 无机材料学报, 2026, 41(5): 628-636. |
| [3] | 李娜, 魏进, 曹锐霄, 刘玉, 黄贵文, 肖红梅. 自研正仲氢转化催化剂的催化测试分析及批量制备工艺优化[J]. 无机材料学报, 2026, 41(5): 645-652. |
| [4] | 程澳芃, 王跃文, 许文涛, 刘全伟, 张海涛, 周有福. 吸附-沉淀自组装结合放电等离子烧结法制备石墨烯增强氧化铝复合陶瓷[J]. 无机材料学报, 2026, 41(4): 536-544. |
| [5] | 王禹贺, 罗颐秀, 郭会明, 张广珩, 张思岩, 孙鲁超, 王杰民, 王京阳. 高熵稀土氧化物热障涂层材料弹性及热物性的第一性原理研究[J]. 无机材料学报, 2026, 41(4): 445-454. |
| [6] | 朱开煌, 杨世杰, 李欣格, 宋贯卿, 史淦升, 王焱, 任小孟, 陆遥, 徐新宏, 孙静. 基于UiO-66骨架的氧化石墨烯改性金属有机框架凝胶的制备及其对甲苯的高效吸附性能[J]. 无机材料学报, 2026, 41(4): 519-526. |
| [7] | 蒋圣楠, 郑重, 何唯一, 刘涛, 潘秀红, 陈锟, 郭辉, 高攀, 刘春俊, 刘学超. 硼镓共掺氧化锌透明电极的制备及性能优化[J]. 无机材料学报, 2026, 41(4): 479-485. |
| [8] | 薛新燕, 张晓卫, 鲁恒, 李世杰, 张卫珂, 曾少华. 钴-镁协同纳米洋葱碳催化剂的制备及低温催化氨分解制氢性能[J]. 无机材料学报, 2026, 41(4): 500-508. |
| [9] | 张梦婕, 李智博, 黄瑞楠, 吕向菲, 王伟. 堇青石/硼酸铝晶须/Co0.8FexCe0.2-xCr2O4催化剂的制备及其碳烟过滤-催化燃烧性能[J]. 无机材料学报, 2026, 41(4): 509-518. |
| [10] | 田洪旺, 罗龙飞, 胡成龙, 闫猛, 庞生洋, 李建, 汤素芳. C/CA表面陶瓷-树脂涂层的简易制备与中温抗氧化性能[J]. 无机材料学报, 2026, 41(3): 401-408. |
| [11] | 韦连金, 齐志杰, 汪信, 朱俊武, 付永胜. 纳米金刚石改性及其在电催化氧还原反应中的应用[J]. 无机材料学报, 2026, 41(3): 273-288. |
| [12] | 黄应贺, 黄仁兴, 石宇星, 雷一杰, 于涛, 王诚, 顾军. 利用介孔碳的限域效应提升氧还原反应中Pt催化剂的耐久性[J]. 无机材料学报, 2026, 41(3): 289-294. |
| [13] | 戚芳, 刘辉, 吴郑敏, 陆毅, 吴雯雯, 王震. 1100 ℃水氧环境下Mini-SiCf/BN/SiC的基体/界面/纤维一体化氧化机制[J]. 无机材料学报, 2026, 41(3): 340-348. |
| [14] | 郑晨, 王湘宁, 苑贺楠, 杨嘉伟, 李传建, 王华栋. 氧化铝纤维增强二氧化硅复合材料力学性能失效研究[J]. 无机材料学报, 2026, 41(3): 331-339. |
| [15] | 李浩, 齐源, 高相东, 张星星, 王金敏. 溶胶凝胶水热法制备耐高温、隔热增强钙掺杂二氧化硅气凝胶[J]. 无机材料学报, 2026, 41(2): 262-272. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||