无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 500-508.DOI: 10.15541/jim20250211 CSTR: 32189.14.10.15541/jim20250211
薛新燕1,2( ), 张晓卫1,2, 鲁恒1,2, 李世杰1,2, 张卫珂1,2(
), 张晓卫1,2, 鲁恒1,2, 李世杰1,2, 张卫珂1,2( ), 曾少华2,3
), 曾少华2,3
收稿日期:2025-05-15
修回日期:2025-09-23
出版日期:2026-04-20
网络出版日期:2025-10-17
通讯作者:
张卫珂, 副教授. E-mail: zhangweike@tyut.edu.cn作者简介:薛新燕(2000-), 女, 硕士研究生. E-mail: 17336334425@163.com
基金资助:
XUE Xinyan1,2( ), ZHANG Xiaowei1,2, LU Heng1,2, LI Shijie1,2, ZHANG Weike1,2(
), ZHANG Xiaowei1,2, LU Heng1,2, LI Shijie1,2, ZHANG Weike1,2( ), CHAN Siewhwa2,3
), CHAN Siewhwa2,3
Received:2025-05-15
Revised:2025-09-23
Published:2026-04-20
Online:2025-10-17
Contact:
ZHANG Weike, associate professor. E-mail: zhangweike@tyut.edu.cnAbout author:XUE Xinyan (2000-), female, Master candidate. E-mail: 17336334425@163.com
Supported by:摘要:
氨分解制氢作为一种极具前景的氢气制备方法, 其关键在于开发具有高活性、高选择性和低成本的中低温催化剂。本研究以甲烷850 ℃裂解制氢副产物纳米洋葱碳(CNOs)为载体, 采用均匀沉积沉淀法负载活性金属钴(Co), 并引入碱金属氧化镁(MgO)作助剂, 成功制备了高性能氨分解催化剂。通过系统研究酸洗及钾(K)活化处理对CNOs载体形貌的调控作用, 深入探究了其对催化剂性能的影响机制。多种表征与化学吸附实验证实, 催化剂碱性强度与其氨分解性能呈正相关。引入CNOs显著提升了催化剂的电子导电性, 并促进了Co2MgO4纳米颗粒在载体上的均匀分散。这种均匀分散增加了碱性活性位点的暴露度, 从而增强了催化剂表面对氨分子的吸附能力。酸洗处理在CNOs表面引入了更多含氧官能团, 这些官能团可作为锚定位点, 与Co2+或Mg2+形成强化学键(配位键或离子键), 进而稳定Co2MgO4颗粒。这种强相互作用提高了金属氧化物的还原难度, 导致其还原温度升高。催化性能测试表明, CNOs、MgO、K和Co的协同作用显著优化了催化剂的结构特性、金属粒径及催化性能。在一系列合成催化剂中, Co2Mg/K-CNO’展现出最优异的氨分解催化活性, 在12000 mL·gcat-1·h-1、550 ℃条件下氨转化率为99.6%。
中图分类号:
薛新燕, 张晓卫, 鲁恒, 李世杰, 张卫珂, 曾少华. 钴-镁协同纳米洋葱碳催化剂的制备及低温催化氨分解制氢性能[J]. 无机材料学报, 2026, 41(4): 500-508.
XUE Xinyan, ZHANG Xiaowei, LU Heng, LI Shijie, ZHANG Weike, CHAN Siewhwa. Co-Mg Synergistic Carbon Nano Onions Catalyst: Preparation and Low-temperature Catalytic Hydrogen Production from Ammonia Decomposition[J]. Journal of Inorganic Materials, 2026, 41(4): 500-508.
| Catalyst | BET specific surface area/ (m2·g-1) | Micropore volume/ (cm3·g-1) | Micropore area/ (m2·g-1) | Average pore size/nm | Co content/% (in mass) |
|---|---|---|---|---|---|
| Co2Mg | 12.7434 | 0.002212 | 6.0318 | 14.6403 | 50 |
| Co2Mg/CNO | 23.4634 | 0.002876 | 6.0278 | 19.4730 | 29 |
| Co2Mg/CNO’ | 23.4880 | 0.003788 | 8.6815 | 20.0063 | 29 |
| Co2Mg/K-CNO | 28.5727 | 0.002307 | 4.8487 | 18.2552 | 29 |
| Co2Mg/K-CNO’ | 34.8266 | 0.002539 | 8.6815 | 17.6383 | 29 |
表1 不同催化剂的比表面积和孔结构参数
Table 1 Specific surface area and pore structure parameters of different catalysts
| Catalyst | BET specific surface area/ (m2·g-1) | Micropore volume/ (cm3·g-1) | Micropore area/ (m2·g-1) | Average pore size/nm | Co content/% (in mass) |
|---|---|---|---|---|---|
| Co2Mg | 12.7434 | 0.002212 | 6.0318 | 14.6403 | 50 |
| Co2Mg/CNO | 23.4634 | 0.002876 | 6.0278 | 19.4730 | 29 |
| Co2Mg/CNO’ | 23.4880 | 0.003788 | 8.6815 | 20.0063 | 29 |
| Co2Mg/K-CNO | 28.5727 | 0.002307 | 4.8487 | 18.2552 | 29 |
| Co2Mg/K-CNO’ | 34.8266 | 0.002539 | 8.6815 | 17.6383 | 29 |
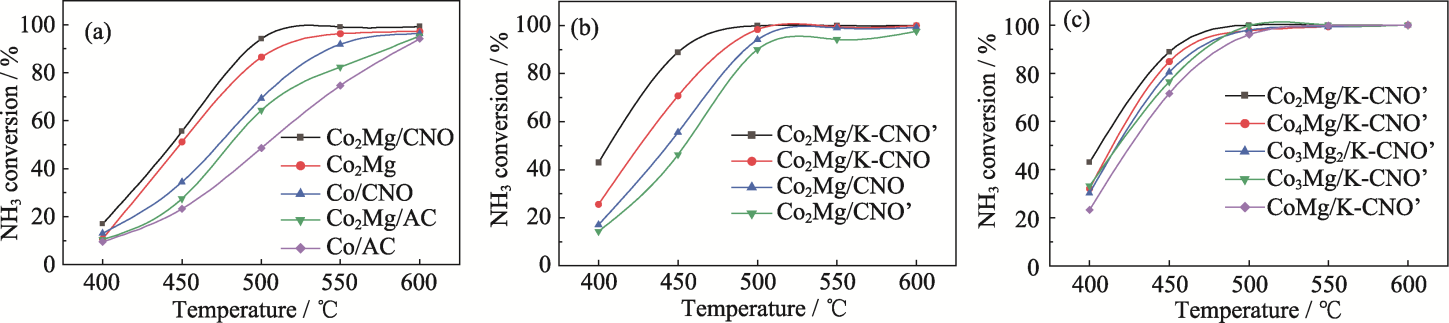
图7 催化剂氨分解活性对比
Fig. 7 Comparison of catalytic activity for ammonia decomposition (a) Co-Mg synergy; (b) K-activated carriers; (c) Different ratios of Co-Mg
| Catalyst | Temperature/℃ | GHSV/(mL·gcat-1·h-1) | NH3 conversion/% | Ref. |
|---|---|---|---|---|
| Co2Mg/K-CNO’ | 550 | 12000 | 99.6 | This work |
| 20CoNaTi-NT | 550 | 6000 | 41 | [ |
| 35%Co/SiC-700 | 550 | 30000 | 78.3 | [ |
| CoOx@C-700A | 500 | 15000 | 55 | [ |
| 90CoAl | 500 | 30000 | 37.8 | [ |
| Co/CNTs | 500 | 6000 | 61 | [ |
| Co-Mo/SiO2-Cap | 500 | 6000 | 20 | [ |
| Co/CeO2 | 500 | 6000 | 62 | [ |
| 26Co-CAT(A) | 500 | 6000 | 58 | [ |
| Fe/CNTs | 500 | 6000 | 15 | [ |
| Co/MWCNTs | 500 | 6000 | 74.6 | [ |
表2 Co基催化剂氨分解性能
Table 2 Catalytic performance of Co-based catalysts for ammonia decomposition
| Catalyst | Temperature/℃ | GHSV/(mL·gcat-1·h-1) | NH3 conversion/% | Ref. |
|---|---|---|---|---|
| Co2Mg/K-CNO’ | 550 | 12000 | 99.6 | This work |
| 20CoNaTi-NT | 550 | 6000 | 41 | [ |
| 35%Co/SiC-700 | 550 | 30000 | 78.3 | [ |
| CoOx@C-700A | 500 | 15000 | 55 | [ |
| 90CoAl | 500 | 30000 | 37.8 | [ |
| Co/CNTs | 500 | 6000 | 61 | [ |
| Co-Mo/SiO2-Cap | 500 | 6000 | 20 | [ |
| Co/CeO2 | 500 | 6000 | 62 | [ |
| 26Co-CAT(A) | 500 | 6000 | 58 | [ |
| Fe/CNTs | 500 | 6000 | 15 | [ |
| Co/MWCNTs | 500 | 6000 | 74.6 | [ |
| [1] |
SUN Y H, SHEN C Q, LAI Q W, et al. Tailoring magnesium based materials for hydrogen storage through synthesis: current state of the art. Energy Storage Materials, 2018, 10: 168.
DOI URL |
| [2] |
SIKIRU S, OLADOSU T L, AMOSA T I, et al. Hydrogen- powered horizons: transformative technologies in clean energy generation, distribution, and storage for sustainable innovation. International Journal of Hydrogen Energy, 2024, 56: 1152.
DOI URL |
| [3] | AL-GHUSSAIN L. Global warming: review on driving forces and mitigation. Environmental Progress & Sustainable Energy, 2019, 38(1): 13. |
| [4] |
LE P A, TRUNG V D, NGUYEN P L, et al. The current status of hydrogen energy: an overview. RSC Advances, 2023, 13(40): 28262.
DOI URL |
| [5] |
PASHCHENKO D. Green hydrogen as a power plant fuel: what is energy efficiency from production to utilization? Renewable Energy, 2024, 223: 120033.
DOI URL |
| [6] |
FELDERHOFF M, WEIDENTHALER C, VON HELMOLT R, et al. Hydrogen storage: the remaining scientific and technological challenges. Physical Chemistry Chemical Physics, 2007, 9(21): 2643.
PMID |
| [7] |
METKEMEIJER R, ACHARD P. Comparison of ammonia and methanol applied indirectly in a hydrogen fuel cell. International Journal of Hydrogen Energy, 1994, 19(6): 535.
DOI URL |
| [8] |
LAMB K E, DOLAN M D, KENNEDY D F. Ammonia for hydrogen storage; a review of catalytic ammonia decomposition and hydrogen separation and purification. International Journal of Hydrogen Energy, 2019, 44(7): 3580.
DOI URL |
| [9] |
MUKHERJEE S, DEVAGUPTAPU S V, SVIRIPA A, et al. Low-temperature ammonia decomposition catalysts for hydrogen generation. Applied Catalysis B: Environmental, 2018, 226: 162.
DOI URL |
| [10] | LIANG D T, FENG C, XU L, et al. Promotion effects of different methods in COx-free hydrogen production from ammonia decomposition. Catalysis Science & Technology, 2023, 13(12): 3614. |
| [11] |
LIU Y, LI X, ZHANG D Q, et al. A general route to prepare low-ruthenium-content bimetallic electrocatalysts for pH-universal hydrogen evolution reaction by using carbon quantum dots. Angewandte Chemie International Edition, 2020, 59(4): 1718.
DOI URL |
| [12] | ZHANG J, COMOTTI M, SCHÜTH F, et al. Commercial Fe- or Co-containing carbon nanotubes as catalysts for NH3 decomposition. Chemical Communications, 2007(19): 1916. |
| [13] |
YIN S F, XU B Q, NG C F, et al. Nano Ru/CNTs: a highly active and stable catalyst for the generation of COx-free hydrogen in ammonia decomposition. Applied Catalysis B: Environmental, 2004, 48(4): 237.
DOI URL |
| [14] |
ZHANG X W, LU H, LI S J, et al. NiTiO3 enhanced Ni/TiO2 strong metal support interaction catalyst towards effective methane catalytic decomposition for hydrogen production with COx-free. Journal of Industrial and Engineering Chemistry, 2025, 147: 482.
DOI URL |
| [15] |
SCHAETZEL P, FAVRE É, THOMAS S, et al. A simple mechanistic multilayer model for the rigorous description of Brunauer- Emmett-Teller type isotherms. Industrial & Engineering Chemistry Research, 2021, 60(34): 12545.
DOI URL |
| [16] |
CHEN B F, LI F B, HUANG Z J, et al. Carbon-coated Cu-Co bimetallic nanoparticles as selective and recyclable catalysts for production of biofuel 2,5-dimethylfuran. Applied Catalysis B: Environmental, 2017, 200: 192.
DOI URL |
| [17] |
NIELSEN D, GAO Q, JANSSENS T V W, et al. Cu-speciation in dehydrated CHA zeolites studied by H2-TPR and in situ EPR. The Journal of Physical Chemistry C, 2023, 127(27): 12995.
DOI URL |
| [18] | YING Q J, WANG X Q, LIU Y, et al. Rational design of a novel core-shell Cu-ZSM-5@Ru/S-1 tandem catalyst for the catalytic combustion of dichloromethane. ACS ES&T Engineering, 2023, 3(7): 1013. |
| [19] | TABASSUM H, MUKHERJEE S, CHEN J J, et al. Hydrogen generation via ammonia decomposition on highly efficient and stable Ru-free catalysts: approaching complete conversion at 450 ℃. Energy & Environmental Science, 2022, 15(10): 4190. |
| [20] |
PINZÓN M, SÁNCHEZ-SÁNCHEZ A, SÁNCHEZ P, et al. Ammonia as a carrier for hydrogen production by using lanthanum based perovskites. Energy Conversion and Management, 2021, 246: 114681.
DOI URL |
| [21] |
GUO X H, LI Y, SHI R J, et al. Co/MgO catalysts for hydrogenolysis of glycerol to 1,2-propanediol. Applied Catalysis A: General, 2009, 371(1/2): 108.
DOI URL |
| [22] |
LARA-GARCÍA H A, MENDOZA-NIETO J A, PFEIFFER H, et al. COx-free hydrogen production from ammonia on novel cobalt catalysts supported on 1D titanate nanotubes. International Journal of Hydrogen Energy, 2019, 44(57): 30062.
DOI URL |
| [23] |
XU R J, YIN F X, ZHANG J, et al. Preparation of Co/SiC catalyst and its catalytic activity for ammonia decomposition to produce hydrogen. Catalysis Today, 2024, 437: 114774.
DOI URL |
| [24] | LI L, JIANG R Y, CHU W, et al. Cobalt nanoparticles embedded in a porous carbon matrix as an efficient catalyst for ammonia decomposition. Catalysis Science & Technology, 2017, 7(6): 1363. |
| [25] |
ZHANG Z S, FU X P, WANG W W, et al. Promoted porous Co3O4-Al2O3 catalysts for ammonia decomposition. Science China Chemistry, 2018, 61(11): 1389.
DOI |
| [26] |
LI Y P, WEN J, ALI A M, et al. Size structure-catalytic performance correlation of supported Ni/MCF-17 catalysts for COx-free hydrogen production. Chemical Communications, 2018, 54(49): 6364.
DOI URL |
| [27] |
SPATOLISANO E, PELLEGRINI L A, DE ANGELIS A R, et al. Ammonia as a carbon-free energy carrier: NH3 cracking to H2. Industrial & Engineering Chemistry Research, 2023, 62(28): 10813.
DOI URL |
| [28] |
GOU H P, LI W B, YANG Y F, et al. Porous skeleton-stabilized Co/N-C coated separator for boosting lithium-ion batteries stability and safety. Journal of Power Sources, 2021, 499: 229933.
DOI URL |
| [29] |
ALAMOUDI O M, KHAN W U, HANTOKO D, et al. Catalytic activity of Co/γ-Al2O3 catalysts for decomposition of ammonia to produce hydrogen. Fuel, 2024, 372: 132230.
DOI URL |
| [30] | ZHANG H, ALHAMED Y A, CHU W, et al. Controlling co-support interaction in Co/MWCNTs catalysts and catalytic performance for hydrogen production via NH3 decomposition. Applied Catalysis A: General, 2013, 464: 156. |
| [1] | 黄应贺, 黄仁兴, 石宇星, 雷一杰, 于涛, 王诚, 顾军. 利用介孔碳的限域效应提升氧还原反应中Pt催化剂的耐久性[J]. 无机材料学报, 2026, 41(3): 289-294. |
| [2] | 任先培, 李超, 胡启威, 向晖, 彭跃红. 金属/过渡金属化合物莫特-肖特基析氢催化剂研究进展[J]. 无机材料学报, 2026, 41(2): 137-149. |
| [3] | 邬博宇, 张深根, 张生杨, 刘波, 张柏林. CeO2对MnOx催化剂低温脱硝性能的影响及其机理研究[J]. 无机材料学报, 2026, 41(1): 87-95. |
| [4] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [5] | 郭子玉, 朱云洲, 王力, 陈健, 李红, 黄政仁. Zn2+催化剂对酚醛树脂/乙二醇制备多孔碳微观孔结构的影响[J]. 无机材料学报, 2025, 40(5): 466-472. |
| [6] | 李建军, 陈芳明, 张梨梨, 王磊, 张丽亭, 陈慧雯, 薛长国, 徐良骥. CoFe2O4/MgAl-LDH催化剂活化过氧一硫酸盐促进抗生素降解[J]. 无机材料学报, 2025, 40(4): 440-448. |
| [7] | 信震宇, 郭瑞华, 乌仁托亚, 王艳, 安胜利, 张国芳, 关丽丽. Pt-Fe/GO纳米催化剂的制备及其电催化乙醇氧化性能研究[J]. 无机材料学报, 2025, 40(4): 379-387. |
| [8] | 孙树娟, 郑南南, 潘昊坤, 马猛, 陈俊, 黄秀兵. 单原子催化剂制备方法的研究进展[J]. 无机材料学报, 2025, 40(2): 113-127. |
| [9] | 李薛茹, 马哲杰, 郭宇杰, 李平. 载体特性对Pt/C催化剂上离聚物覆盖度及氧还原性能的影响[J]. 无机材料学报, 2025, 40(12): 1395-1404. |
| [10] | 赵丽娟, 谭哲, 张晓光, 蒋国赛, 陶然, 潘德安. 废加氢催化剂颗粒分级数值模拟研究[J]. 无机材料学报, 2025, 40(12): 1387-1394. |
| [11] | 刘盼盼, 姚鹏, 刘栩孜, 屈丽, 曾路, 宋兆华, 焦毅, 王健礼, 陈耀强. MnOx/CeO2-ZrO2复合氧化物的构筑及其在碳烟氧化中的应用[J]. 无机材料学报, 2025, 40(11): 1300-1308. |
| [12] | 唐阳, 刘立敏, 周晓亮, 张搏, 蒋星洲, 贾浩义, 罗延麟庆. 质子陶瓷膜反应器的制备及低温氨分解性能研究[J]. 无机材料学报, 2025, 40(11): 1277-1284. |
| [13] | 李娜, 曹锐霄, 魏进, 周晗, 肖红梅. 铁基正仲氢转化催化剂的影响因素[J]. 无机材料学报, 2025, 40(1): 47-52. |
| [14] | 连敏丽, 苏佳欣, 黄鸿杨, 嵇玉寅, 邓海帆, 张彤, 陈崇启, 李达林. Ni-Mg-Al类水滑石衍生镍基催化剂的制备及其氨分解性能[J]. 无机材料学报, 2025, 40(1): 53-60. |
| [15] | 刘磊, 郭瑞华, 王丽, 王艳, 张国芳, 关丽丽. Pt3Co高指数晶面氧还原过程的密度泛函理论研究[J]. 无机材料学报, 2025, 40(1): 39-46. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||