无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 445-454.DOI: 10.15541/jim20250272 CSTR: 32189.14.jim20250272
王禹贺1,2( ), 罗颐秀1(
), 罗颐秀1( ), 郭会明3, 张广珩4, 张思岩4, 孙鲁超1, 王杰民1, 王京阳1(
), 郭会明3, 张广珩4, 张思岩4, 孙鲁超1, 王杰民1, 王京阳1( )
)
收稿日期:2025-06-27
修回日期:2025-08-22
出版日期:2026-04-20
网络出版日期:2025-08-26
通讯作者:
罗颐秀, 项目研究员. E-mail: yxluo13s@imr.ac.cn;作者简介:王禹贺(2001-), 男, 硕士研究生. E-mail: yhwang23s@imr.ac.cn
基金资助:
WANG Yuhe1,2( ), LUO Yixiu1(
), LUO Yixiu1( ), GUO Huiming3, ZHANG Guangheng4, ZHANG Siyan4, SUN Luchao1, WANG Jiemin1, WANG Jingyang1(
), GUO Huiming3, ZHANG Guangheng4, ZHANG Siyan4, SUN Luchao1, WANG Jiemin1, WANG Jingyang1( )
)
Received:2025-06-27
Revised:2025-08-22
Published:2026-04-20
Online:2025-08-26
Contact:
LUO Yixiu, professor. E-mail: yxluo13s@imr.ac.cn;About author:WANG Yuhe (2001-), male, Master candidate. E-mail: yhwang23s@imr.ac.cn
Supported by:摘要:
应用于高推重比航空发动机热端部件的连续纤维增强碳化硅陶瓷基复合材料需采用热/环境障复合涂层(T/EBCs)进行防护。为了得到兼具低热导率、适配的热膨胀系数及良好的高温相稳定性的新型稀土氧化物热障涂层材料, 高熵化设计概念为其成分设计和性能调控提供了新思路和新契机。本研究针对复杂高熵陶瓷体系的结构建模和性能预测难题, 提出了一种基于特殊准随机结构(SQS)的新型高熵建模策略。该方法在保证计算精度的同时, 实现了对复杂结构陶瓷性能的快速预测。随后, 结合第一性原理计算方法, 预测并比较了四种高熵稀土氧化物材料的晶体结构、弹性性质及热物理性质, 重点揭示了不同稀土成分及Hf掺杂对材料低热导率性能的调控作用及原子尺度根源。本研究为航空发动机热端部件用T/EBCs材料的理论模拟和选材设计提供了科学思路与基础数据。
中图分类号:
王禹贺, 罗颐秀, 郭会明, 张广珩, 张思岩, 孙鲁超, 王杰民, 王京阳. 高熵稀土氧化物热障涂层材料弹性及热物性的第一性原理研究[J]. 无机材料学报, 2026, 41(4): 445-454.
WANG Yuhe, LUO Yixiu, GUO Huiming, ZHANG Guangheng, ZHANG Siyan, SUN Luchao, WANG Jiemin, WANG Jingyang. First-principles Investigation of Elastic and Thermophysical Properties of High-entropy Rare-earth Oxide Thermal Barrier Coating Materials[J]. Journal of Inorganic Materials, 2026, 41(4): 445-454.
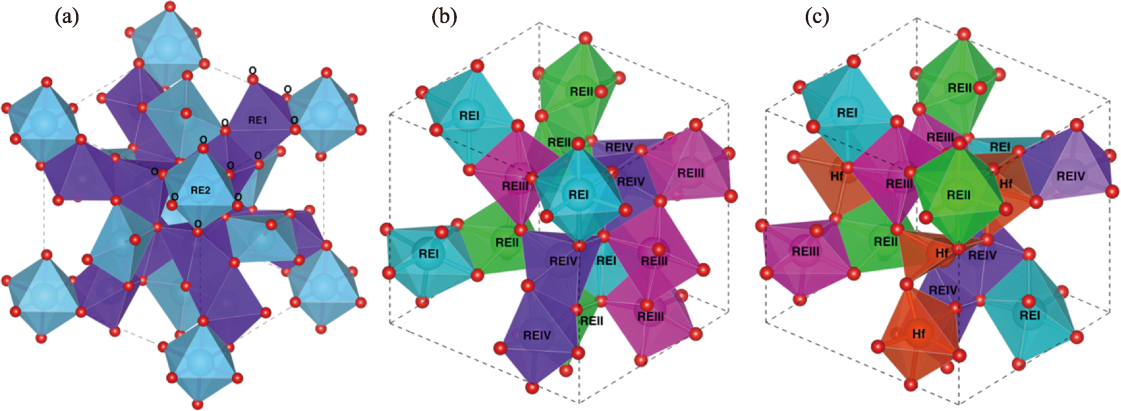
图1 高熵稀土氧化物材料的晶体结构
Fig. 1 Crystal structures of high-entropy rare-earth oxide materials (a) C-RE2O3; (b) C-(REI1/4REII1/4REIII1/4REIV1/4)2O3; (c) C-(REI3/16REII3/16REIII3/16REIV3/16Hf1/4)2O3
| Compound | Method | a/Å | b/Å | c/Å | α/(°) | β/(°) | γ/(°) | ρ/(g•cm-3) |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | Calc | 9.13 | 9.14 | 9.15 | 109.49 | 109.52 | 109.46 | 7.81 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | Calc | 9.05 | 9.04 | 9.04 | 109.43 | 109.51 | 109.46 | 9.10 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | Calc | 9.01 | 9.06 | 9.01 | 109.34 | 109.37 | 109.55 | 8.43 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | Calc | 8.92 | 9.00 | 8.98 | 109.53 | 109.74 | 109.47 | 9.46 |
表1 四种高熵稀土氧化物材料的计算晶格参数
Table 1 Calculated lattice parameters of four high-entropy rare-earth oxide materials
| Compound | Method | a/Å | b/Å | c/Å | α/(°) | β/(°) | γ/(°) | ρ/(g•cm-3) |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | Calc | 9.13 | 9.14 | 9.15 | 109.49 | 109.52 | 109.46 | 7.81 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | Calc | 9.05 | 9.04 | 9.04 | 109.43 | 109.51 | 109.46 | 9.10 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | Calc | 9.01 | 9.06 | 9.01 | 109.34 | 109.37 | 109.55 | 8.43 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | Calc | 8.92 | 9.00 | 8.98 | 109.53 | 109.74 | 109.47 | 9.46 |
| Compound | c11 | c12 | c13 | c22 | c23 | c33 | c44 | c55 | c66 |
|---|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 244.79 | 95.97 | 95.90 | 240.43 | 101.79 | 239.03 | 69.14 | 62.94 | 63.10 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 257.22 | 100.81 | 99.30 | 251.14 | 107.11 | 250.74 | 72.87 | 66.14 | 66.16 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 250.73 | 98.29 | 98.37 | 248.15 | 97.99 | 241.45 | 70.74 | 69.71 | 69.93 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 259.08 | 102.63 | 100.97 | 250.68 | 97.74 | 251.63 | 77.04 | 71.63 | 75.61 |
表2 四种高熵稀土氧化物材料的二阶弹性常数(GPa)
Table 2 Calculated second-order elastic coefficients of four high-entropy rare-earth oxide materials (GPa)
| Compound | c11 | c12 | c13 | c22 | c23 | c33 | c44 | c55 | c66 |
|---|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 244.79 | 95.97 | 95.90 | 240.43 | 101.79 | 239.03 | 69.14 | 62.94 | 63.10 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 257.22 | 100.81 | 99.30 | 251.14 | 107.11 | 250.74 | 72.87 | 66.14 | 66.16 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 250.73 | 98.29 | 98.37 | 248.15 | 97.99 | 241.45 | 70.74 | 69.71 | 69.93 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 259.08 | 102.63 | 100.97 | 250.68 | 97.74 | 251.63 | 77.04 | 71.63 | 75.61 |
| Compound | B/GPa | G/GPa | E/GPa | Emax/GPa | Emin/GPa | Emax/Emin | G/B | ν/GPa |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 146 | 67 | 175 | 192 | 151 | 1.2753 | 0.4606 | 0.3004 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 153 | 71 | 183 | 201 | 159 | 1.2639 | 0.4624 | 0.2997 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 148 | 72 | 185 | 197 | 174 | 1.1323 | 0.4852 | 0.2912 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 151 | 75 | 194 | 209 | 180 | 1.1605 | 0.4975 | 0.2866 |
表3 四种高熵稀土氧化物材料的弹性模量
Table 3 Calculated elastic modulus of four high-entropy rare-earth oxide materials
| Compound | B/GPa | G/GPa | E/GPa | Emax/GPa | Emin/GPa | Emax/Emin | G/B | ν/GPa |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 146 | 67 | 175 | 192 | 151 | 1.2753 | 0.4606 | 0.3004 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 153 | 71 | 183 | 201 | 159 | 1.2639 | 0.4624 | 0.2997 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 148 | 72 | 185 | 197 | 174 | 1.1323 | 0.4852 | 0.2912 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 151 | 75 | 194 | 209 | 180 | 1.1605 | 0.4975 | 0.2866 |
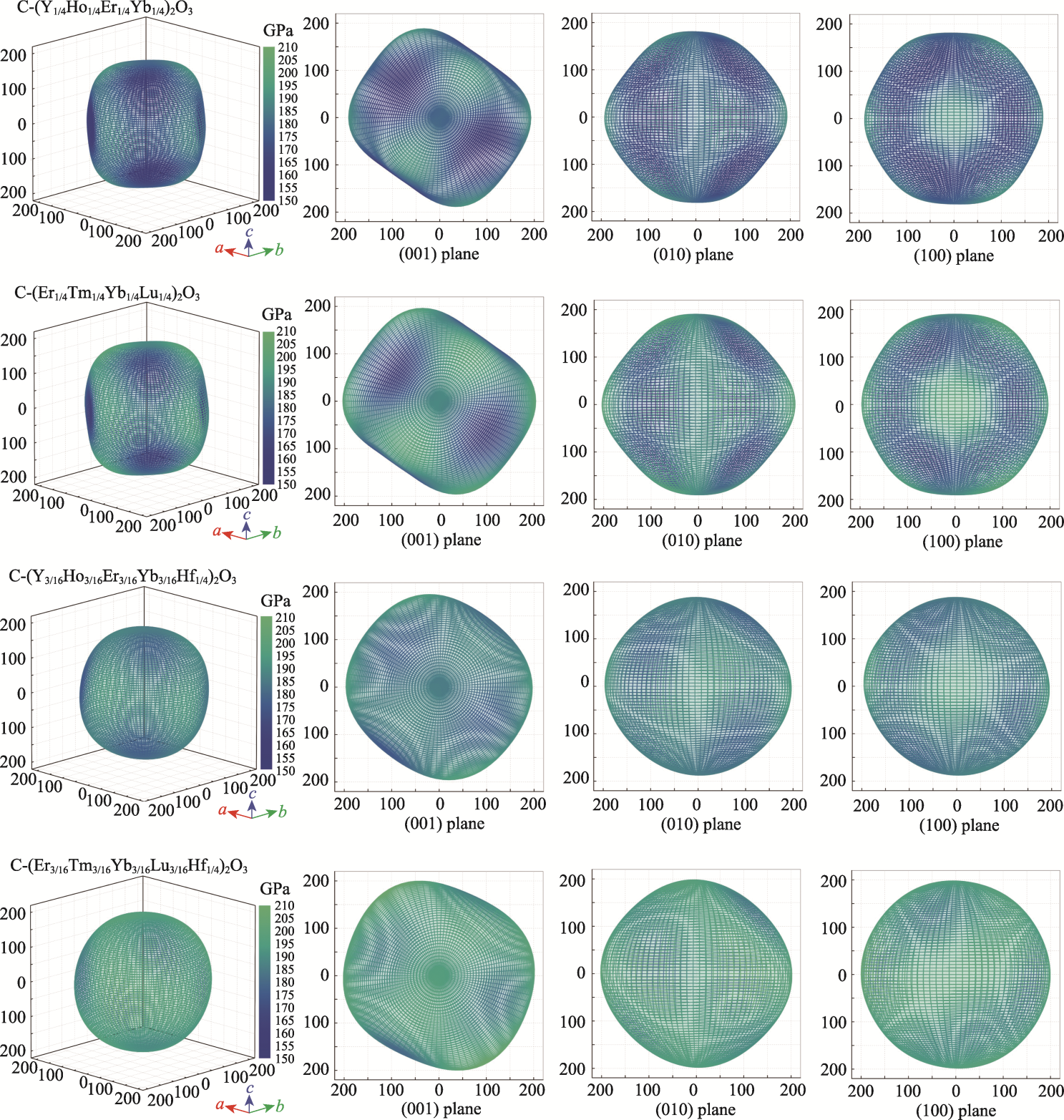
图3 四种高熵稀土氧化物杨氏模量的3D形貌图和(001)、(010)、(100)投影图
Fig. 3 3D topography, (001), (010) and (100) projective pictures of Young's modulus of four high-entropy rare-earth oxides
| Compound | vs/(m·s-1) | vl/(m·s-1) | vm/(m·s-1) | ΘD/K | γ | αL/(×10-6, K-1) | kmin/(W·m-1·K-1) |
|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 2932 | 5489 | 3276 | 398 | 1.7749 | 11.4574 | 0.7567 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 2785 | 5207 | 3111 | 382 | 1.7707 | 11.2533 | 0.7334 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 2915 | 5371 | 3253 | 400 | 1.7194 | 11.3375 | 0.7690 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 2823 | 5162 | 3148 | 390 | 1.6928 | 11.1577 | 0.7566 |
表4 四种高熵稀土氧化物材料的热物性参数
Table 4 Thermophysical parameters of four high-entropy rare-earth oxide materials
| Compound | vs/(m·s-1) | vl/(m·s-1) | vm/(m·s-1) | ΘD/K | γ | αL/(×10-6, K-1) | kmin/(W·m-1·K-1) |
|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 2932 | 5489 | 3276 | 398 | 1.7749 | 11.4574 | 0.7567 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 2785 | 5207 | 3111 | 382 | 1.7707 | 11.2533 | 0.7334 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 2915 | 5371 | 3253 | 400 | 1.7194 | 11.3375 | 0.7690 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 2823 | 5162 | 3148 | 390 | 1.6928 | 11.1577 | 0.7566 |
| Parameter | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf | Diff(1-3)/% | Diff(2-4)/% | Diff(1-2)/% | Diff(3-4)/% |
|---|---|---|---|---|---|---|---|---|
| kT | 898.98 | 892.94 | 1006.36 | 1050.75 | 12.07 | 17.70 | -0.68 | 4.32 |
| 1.44×10-12 | 1.62×10-12 | 1.49×10-12 | 1.62×10-12 | 3.15 | -0.01 | 11.90 | 8.48 | |
| ΘD3 | 6.31×107 | 5.58×107 | 6.40×107 | 5.95×107 | 1.36 | 6.69 | -11.69 | -7.05 |
| A(γ)/γ2 | 9.85×106 | 9.90×106 | 1.06×107 | 1.09×107 | 7.19 | 10.33 | 0.52 | 3.47 |
表5 四种高熵稀土氧化物材料的k~αT-1定量化关系及差异根源
Table 5 Quantitative relationship of k~αT-1 and origin of differences in four high-entropy rare-earth oxide materials
| Parameter | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf | Diff(1-3)/% | Diff(2-4)/% | Diff(1-2)/% | Diff(3-4)/% |
|---|---|---|---|---|---|---|---|---|
| kT | 898.98 | 892.94 | 1006.36 | 1050.75 | 12.07 | 17.70 | -0.68 | 4.32 |
| 1.44×10-12 | 1.62×10-12 | 1.49×10-12 | 1.62×10-12 | 3.15 | -0.01 | 11.90 | 8.48 | |
| ΘD3 | 6.31×107 | 5.58×107 | 6.40×107 | 5.95×107 | 1.36 | 6.69 | -11.69 | -7.05 |
| A(γ)/γ2 | 9.85×106 | 9.90×106 | 1.06×107 | 1.09×107 | 7.19 | 10.33 | 0.52 | 3.47 |
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XREⅠ=XREⅡ=XREⅢ= XREIV=1/4 |
表S1 C-(REⅠ1/4REⅡ1/4REⅢ1/4REⅣ1/4)2O3的亚晶格位点数和成分组成
Table S1 Number of sites on sublattice and composition for C-(REⅠ1/4REⅡ1/4REⅢ1/4REⅣ1/4)2O3
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XREⅠ=XREⅡ=XREⅢ= XREIV=1/4 |
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=0 XREⅡ=XREⅢ=XREⅣ= XHf =1/4 | XREⅠ=XHf=1/4 XREⅡ=XREⅢ=XREⅣ= 1/6 | |
| No.2 | NS | 4 | 12 |
| XS | XREⅡ=0 XREⅠ=XREⅢ=XREⅣ= XHf=1/4 | XREⅡ=XHf=1/4 XREⅠ=XREⅢ=XREⅣ= 1/6 | |
| No.3 | NS | 4 | 12 |
| XS | XREⅢ=0 XREⅠ=XREⅡ=XREⅣ= XHf=1/4 | XREⅢ=XHf=1/4 XREⅠ=XREⅡ=XREⅣ= 1/6 | |
| No.4 | NS | 4 | 12 |
| XS | XREⅣ=0 XREⅠ=XREⅡ=XREⅢ= XHf=1/4 | XREⅣ=XHf=1/4 XREⅠ=XREⅡ=XREⅢ= 1/6 | |
| No.5 | NS | 4 | 12 |
| XS | XHf=0 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XHf=1/3 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/6 |
表S2 C-(REⅠ3/16REⅡ3/16REⅢ3/16REⅣ3/16Hf1/4)2O3的亚晶格位点数和成分组成
Table S2 Number of sites on sublattice and composition for C-(REⅠ3/16REⅡ3/16REⅢ3/16REⅣ3/16Hf1/4)2O3
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=0 XREⅡ=XREⅢ=XREⅣ= XHf =1/4 | XREⅠ=XHf=1/4 XREⅡ=XREⅢ=XREⅣ= 1/6 | |
| No.2 | NS | 4 | 12 |
| XS | XREⅡ=0 XREⅠ=XREⅢ=XREⅣ= XHf=1/4 | XREⅡ=XHf=1/4 XREⅠ=XREⅢ=XREⅣ= 1/6 | |
| No.3 | NS | 4 | 12 |
| XS | XREⅢ=0 XREⅠ=XREⅡ=XREⅣ= XHf=1/4 | XREⅢ=XHf=1/4 XREⅠ=XREⅡ=XREⅣ= 1/6 | |
| No.4 | NS | 4 | 12 |
| XS | XREⅣ=0 XREⅠ=XREⅡ=XREⅢ= XHf=1/4 | XREⅣ=XHf=1/4 XREⅠ=XREⅡ=XREⅢ= 1/6 | |
| No.5 | NS | 4 | 12 |
| XS | XHf=0 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XHf=1/3 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/6 |
| Compound | Unit | Average bond length/Å | |
|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | YO6 | 2.2868 | |
| HoO6 | 2.2794 | ||
| ErO6 | 2.2700 | ||
| YbO6 | 2.2518 | ||
| REO6 | 2.2720 | ||
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | ErO6 | 2.2631 | |
| TmO6 | 2.2535 | ||
| YbO6 | 2.2446 | ||
| LuO6 | 2.2356 | ||
| REO6 | 2.2492 | ||
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | YO6 | 2.2991 | |
| HoO6 | 2.2880 | ||
| ErO6 | 2.2794 | ||
| YbO6 | 2.2451 | ||
| REO6 | 2.2779 | ||
| HfO6 | 2.1893 | ||
| XO6* | 2.2558 | ||
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | ErO6 | 2.2684 | |
| TmO6 | 2.2541 | ||
| YbO6 | 2.2437 | ||
| LuO6 | 2.2399 | ||
| REO6 | 2.2515 | ||
| HfO6 | 2.1936 | ||
| XO6* | 2.2370 | ||
表S3 四种高熵稀土氧化物材料多面体结构单元的平均键长
Table S3 Average bond length of polyhedral of four high-entropy rare-earth oxide materials
| Compound | Unit | Average bond length/Å | |
|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | YO6 | 2.2868 | |
| HoO6 | 2.2794 | ||
| ErO6 | 2.2700 | ||
| YbO6 | 2.2518 | ||
| REO6 | 2.2720 | ||
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | ErO6 | 2.2631 | |
| TmO6 | 2.2535 | ||
| YbO6 | 2.2446 | ||
| LuO6 | 2.2356 | ||
| REO6 | 2.2492 | ||
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | YO6 | 2.2991 | |
| HoO6 | 2.2880 | ||
| ErO6 | 2.2794 | ||
| YbO6 | 2.2451 | ||
| REO6 | 2.2779 | ||
| HfO6 | 2.1893 | ||
| XO6* | 2.2558 | ||
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | ErO6 | 2.2684 | |
| TmO6 | 2.2541 | ||
| YbO6 | 2.2437 | ||
| LuO6 | 2.2399 | ||
| REO6 | 2.2515 | ||
| HfO6 | 2.1936 | ||
| XO6* | 2.2370 | ||
| Material | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf |
|---|---|---|---|---|
| s11 | 0.005239 | 0.004968 | 0.005147 | 0.005011 |
| s12 | -0.001469 | -0.001396 | -0.001445 | -0.001504 |
| s13 | -0.001491 | -0.001365 | -0.001516 | -0.001426 |
| s14 | -0.000049 | -0.000006 | -0.000043 | -0.000120 |
| s15 | -0.000082 | -0.000019 | 0.000079 | 0.000086 |
| s16 | 0.000013 | 0.000002 | 0.000050 | -0.000122 |
| s22 | 0.005544 | 0.005321 | 0.005213 | 0.005182 |
| s23 | -0.001794 | -0.001739 | -0.001541 | -0.001420 |
| s24 | -0.000058 | 0.000003 | 0.000073 | -0.000020 |
| s25 | 0.000077 | -0.000031 | -0.000081 | -0.000353 |
| s26 | 0.000974 | 0.000913 | 0.000381 | 0.000603 |
| s33 | 0.005584 | 0.005303 | 0.005388 | 0.005103 |
| s34 | 0.000051 | -0.000014 | 0.000040 | 0.000062 |
| s35 | 0.000000 | 0.000049 | 0.000058 | 0.000069 |
| s36 | -0.000990 | -0.000888 | -0.000400 | -0.000397 |
| s44 | 0.014726 | 0.013938 | 0.014169 | 0.012992 |
| s45 | -0.002028 | -0.001789 | -0.000457 | -0.000304 |
| s46 | 0.000017 | -0.000002 | -0.000460 | 0.000002 |
| s55 | 0.016174 | 0.015356 | 0.014366 | 0.014004 |
| s56 | -0.000060 | 0.000077 | -0.000237 | -0.000343 |
| s66 | 0.016120 | 0.015338 | 0.014369 | 0.013311 |
表S4 四种高熵稀土氧化物材料的柔度系数
Table S4 Elastic compliance constants of four high-entropy rare-earth oxide materials
| Material | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf |
|---|---|---|---|---|
| s11 | 0.005239 | 0.004968 | 0.005147 | 0.005011 |
| s12 | -0.001469 | -0.001396 | -0.001445 | -0.001504 |
| s13 | -0.001491 | -0.001365 | -0.001516 | -0.001426 |
| s14 | -0.000049 | -0.000006 | -0.000043 | -0.000120 |
| s15 | -0.000082 | -0.000019 | 0.000079 | 0.000086 |
| s16 | 0.000013 | 0.000002 | 0.000050 | -0.000122 |
| s22 | 0.005544 | 0.005321 | 0.005213 | 0.005182 |
| s23 | -0.001794 | -0.001739 | -0.001541 | -0.001420 |
| s24 | -0.000058 | 0.000003 | 0.000073 | -0.000020 |
| s25 | 0.000077 | -0.000031 | -0.000081 | -0.000353 |
| s26 | 0.000974 | 0.000913 | 0.000381 | 0.000603 |
| s33 | 0.005584 | 0.005303 | 0.005388 | 0.005103 |
| s34 | 0.000051 | -0.000014 | 0.000040 | 0.000062 |
| s35 | 0.000000 | 0.000049 | 0.000058 | 0.000069 |
| s36 | -0.000990 | -0.000888 | -0.000400 | -0.000397 |
| s44 | 0.014726 | 0.013938 | 0.014169 | 0.012992 |
| s45 | -0.002028 | -0.001789 | -0.000457 | -0.000304 |
| s46 | 0.000017 | -0.000002 | -0.000460 | 0.000002 |
| s55 | 0.016174 | 0.015356 | 0.014366 | 0.014004 |
| s56 | -0.000060 | 0.000077 | -0.000237 | -0.000343 |
| s66 | 0.016120 | 0.015338 | 0.014369 | 0.013311 |
| [1] |
LEE K N, FOX D S, BANSAL N P. Rare earth silicate environmental barrier coatings for SiC/SiC composites and Si3N4 ceramics. Journal of the European Ceramic Society, 2005, 25(10): 1705.
DOI URL |
| [2] |
MARTIN D, BENNETT C, HUSSAIN T. A review on environmental barrier coatings: history, current state of the art and future developments. Journal of the European Ceramic Society, 2021, 41(3): 1747.
DOI URL |
| [3] |
DUAN Z, DENG L, LU K, et al. Thermal cycle and water oxygen performance of multi-layered Y3Al5O12/Yb2SiO5/Yb2Si2O7 thermal/ environmental barrier coatings. Ceramics International, 2024, 50(9): 16309.
DOI URL |
| [4] |
ZINKEVICH M. Thermodynamics of rare earth sesquioxides. Progress in Materials Science, 2007, 52(4): 597.
DOI URL |
| [5] |
ZHANG G H, ZHANG J, WANG J Y. Synthesis and characterization of ytterbium oxide: a novel CMAS-resistant environmental barrier coating material. Journal of the American Ceramic Society, 2023, 106(1): 621.
DOI URL |
| [6] |
ZHANG G H, SHI J Y, ZHANG J, et al. Investigation on crystallization behavior between (ScxYb1-x)O1.5 and CMAS: a new insight in the effect of Sc substitution. Journal of Advanced Ceramics, 2024, 13(6): 789.
DOI URL |
| [7] |
WRIGHT A J, WANG Q Y, HUANG C Y, et al. From high-entropy ceramics to compositionally-complex ceramics: a case study of fluorite oxides. Journal of the European Ceramic Society, 2020, 40(5): 2120.
DOI URL |
| [8] |
SUN L C, REN X M, DU T F, et al. High entropy engineering: new strategy for the critical property optimizations of rare earth silicates. Journal of Inorganic Materials, 2021, 36(4): 339.
DOI |
| [9] |
WANG Y D, GUO Q, ZHOU Q J, et al. Research progress in high temperature functional coatings for advanced aeroengines. Journal of Aeronautical Materials, 2024, 44(5): 48.
DOI |
| [10] |
PAN W, LIU G H, WANG X L, et al. Review of the development of thermal barrier coating materials and technologies. Thermal Spray Technology, 2025, 17(1): 1.
DOI URL |
| [11] | LUO X W, XU C H, DUAN S S, et al. Research progress of high-entropy thermal barrier coatings ceramic materials. Aeronautical Manufacturing Technology, 2022, 65(3): 82. |
| [12] |
SUN Y N, XIANG H M, DAI F Z, et al. Preparation and properties of CMAS resistant bixbyite structured high-entropy oxides RE2O3 (RE=Sm, Eu, Er, Lu, Y, and Yb): promising environmental barrier coating materials for Al2O3f/Al2O3 composites. Journal of Advanced Ceramics, 2021, 10: 596.
DOI |
| [13] |
ARDREY K D, RIDLEY M J, WANG K, et al. Opportunities for novel refractory alloy thermal/environmental barrier coatings using multicomponent rare earth oxides. Scripta Materialia, 2024, 251: 116206.
DOI URL |
| [14] |
PING X Y, MENG B, YU X H, et al. Structural, mechanical and thermal properties of cubic bixbyite-structured high-entropy oxides. Chemical Engineering Journal, 2023, 464: 142649.
DOI URL |
| [15] |
MENG H, YU R, TANG Z, et al. Formation ability descriptors for high-entropy diborides established through high-throughput experiments and machine learning. Acta Materialia, 2023, 256: 119132.
DOI URL |
| [16] |
GYORFFY B L. Coherent-potential approximation for a nonoverlap muffin-tin-potential model of random substitutional alloys. Physical Review B, 1972, 5(6): 2382.
DOI URL |
| [17] |
BELLAICHE L, VANDERBILT D. Virtual crystal approximation revisited: application to dielectric and piezoelectric properties of perovskites. Physical Review B, 2000, 61(12): 7877.
DOI URL |
| [18] |
VAN DE WALLE A, TIWARY P, DE JONG M, et al. Efficient stochastic generation of special quasirandom structures. Calphad, 2013, 42: 13.
DOI URL |
| [19] |
REN X M, TIAN Z L, ZHANG J, et al. Equiatomic quaternary (Y1/4Ho1/4Er1/4Yb1/4)2SiO5 silicate: a perspective multifunctional thermal and environmental barrier coating material. Scripta Materialia, 2019, 168: 47.
DOI URL |
| [20] |
SUN L C, LUO Y X, TIAN Z L, et al. High temperature corrosion of (Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7 environmental barrier coating material subjected to water vapor and molten calcium-magnesium- aluminosilicate (CMAS). Corrosion Science, 2020, 175: 108881.
DOI URL |
| [21] |
KRESSE G, FURTHMULLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Physical Review B, 1996, 54(16): 11169.
DOI URL |
| [22] |
BLOCHL P E. Projector augmented-wave method. Physical Review B, 1994, 50(24): 17953.
DOI PMID |
| [23] |
PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple. Physical Review Letters, 1997, 77(18): 3865.
DOI URL |
| [24] |
MONKHORST H J, PACK J D. Special points for Brillouin-zone integrations. Physical Review B, 1976, 13(12): 5188.
DOI URL |
| [25] |
LUO Y X, WANG J, SUN L C, et al. Phononic origin of the infrared dielectric properties of RE2O3 (RE=Y, Gd, Ho, Lu) compounds. Modelling and Simulation in Materials Science and Engineering, 2024, 32(5): 055009.
DOI |
| [26] | VOIG W. Lehrbuch der Kristallphysik 962. Leipzig: Teubner, 1928. |
| [27] | REUSS A. Berechnung der fließgrenze von mischkristallen auf grund der plastizitätsbedingung für einkristalle. Journal of Applied Mathematics and Mechanics, 1929, 9(1): 49. |
| [28] | HILL R. The elastic behaviour of a crystalline aggregate. Proceedings of the Physical Society. Section A, 1952, 65(5): 349. |
| [29] |
LUO Y X, WANG J M, LI J N, et al. Theoretical study on crystal structures, elastic stiffness, and intrinsic thermal conductivities of β-, γ-, and δ-Y2Si2O7. Journal of Materials Research, 2015, 30(4): 493.
DOI URL |
| [30] | NYE J F. Physical properties of crystals:their representation by tensors and matrices. Oxford: Oxford University Press, 1985. |
| [31] | KITTEL C, MCEUEN P. Introduction to solid state physics. New York: John Wiley & Sons, 2018. |
| [32] |
LUO Y X, SUN L C, WANG J M, et al. Material-genome perspective towards tunable thermal expansion of rare-earth disilicates. Journal of the European Ceramic Society, 2018, 38(10): 3547.
DOI URL |
| [33] | CLARKE D R. Materials selection guidelines for low thermal conductivity thermal barrier coatings. Surface and Coatings Technology, 2003, 163: 67. |
| [34] | MORELLI D T, SLACK G A. High lattice thermal conductivity solids. New York: Springer New York, 2006: 37-68. |
| [35] |
ANDERSON O L. A simplified method for calculating the Debye temperature from elastic constants. Journal of Physics and Chemistry of Solids, 1963, 24(7): 909.
DOI URL |
| [36] |
SANDITOV B D, TSYDYPOV S B, SANDITOV D S. Relation between the Grüneisen constant and Poisson’s ratio of vitreous systems. Acoustical Physics, 2007, 53: 594.
DOI URL |
| [37] | BRULSR J. The thermal conductivity of magnesium silicon nitride, MgSiN2, ceramics and related materials. Eindhoven: PhD in Chemical Engineering and Chemistry from Eindhoven University of Technology, 2000. |
| [38] |
LUO Y X, WANG JM J, LI Y R, et al. Giant phonon anharmonicity and anomalous pressure dependence of lattice thermal conductivity in Y2Si2O7 silicate. Scientific Reports, 2016, 6: 29801.
DOI |
| [39] |
WEI G, ZUO Y, LUO F, et al. Investigation of mechanical and thermodynamic properties of La2Zr2O7 pyrochlore. International Journal of Energy Research, 2022, 46(2): 2011.
DOI URL |
| [40] |
LUO F, LI B, GUO Z, et al. Ab initio calculation of mechanical and thermodynamic properties of Gd2Zr2O7 pyrochlore. Materials Chemistry and Physics, 2020, 243: 122565.
DOI URL |
| [41] |
CAO X Q, VASSEN R, STOVER D. Ceramic materials for thermal barrier coatings. Journal of the European Ceramic Society, 2004, 24(1): 1.
DOI URL |
| [42] |
TIAN Z L, ZHENG L Y, WANG J M, et al. Theoretical and experimental determination of the major thermo-mechanical properties of RE2SiO5 (RE=Tb, Dy, Ho, Er, Tm, Yb, Lu, and Y) for environmental and thermal barrier coating applications. Journal of the European Ceramic Society, 2016, 36(1): 189.
DOI URL |
| [43] |
TIAN Z L, ZHENG L Y, LI Z, et al. Exploration of the low thermal conductivities of γ-Y2Si2O7, β-Y2Si2O7, β-Yb2Si2O7, and β-Lu2Si2O7 as novel environmental barrier coating candidates. Journal of the European Ceramic Society, 2016, 36(11): 2813.
DOI URL |
| [1] | 郭佳芯, 陈美娟, 吴浩, 郑潇然, 闵楠, 田辉, 齐东丽, 李全军, 都时禹, 沈龙海. 高压下新型MAX相Zr3InC2的第一性原理研究[J]. 无机材料学报, 2025, 40(12): 1414-1424. |
| [2] | 吴玉豪, 彭仁赐, 程春玉, 杨丽, 周益春. HfxTa1-xC体系力学性能及熔化曲线的第一性原理研究[J]. 无机材料学报, 2024, 39(7): 761-768. |
| [3] | 靳宇翔, 宋二红, 朱永福. 3d过渡金属单原子掺杂石墨烯缺陷电催化还原CO2的第一性原理研究[J]. 无机材料学报, 2024, 39(7): 845-852. |
| [4] | 王伟华, 张磊宁, 丁峰, 代兵, 韩杰才, 朱嘉琦, 贾怡, 杨宇. 铱衬底上金刚石外延形核与生长: 第一性原理计算[J]. 无机材料学报, 2024, 39(4): 416-422. |
| [5] | 张宇晨, 陆知遥, 赫晓东, 宋广平, 朱春城, 郑永挺, 柏跃磊. 硫族MAX相硼化物的物相稳定性和性能预测[J]. 无机材料学报, 2024, 39(2): 225-232. |
| [6] | 周靖渝, 李兴宇, 赵晓琳, 王有伟, 宋二红, 刘建军. Ti和Cu掺杂β-NaMnO2正极材料:钠离子电池的倍率和循环性能[J]. 无机材料学报, 2024, 39(12): 1404-1412. |
| [7] | 陈梦杰, 王倩倩, 吴成铁, 黄健. 基于DFT的描述符预测生物陶瓷的降解性[J]. 无机材料学报, 2024, 39(10): 1175-1181. |
| [8] | 周云凯, 刁亚琪, 王明磊, 张宴会, 王利民. 聚苯胺改性Ti3C2(OH)2抗氧化性的第一性原理计算研究[J]. 无机材料学报, 2024, 39(10): 1151-1158. |
| [9] | 吴晓维, 张涵, 曾彪, 明辰, 孙宜阳. 杂化泛函HSE和PBE0计算CsPbI3缺陷性质的比较研究[J]. 无机材料学报, 2023, 38(9): 1110-1116. |
| [10] | 张守超, 陈洪雨, 刘洪飞, 杨羽, 李欣, 刘德峰. 6H-SiC中子辐照肿胀高温回复及光学特性研究[J]. 无机材料学报, 2023, 38(6): 678-686. |
| [11] | 杨颖康, 邵怡晴, 李柏良, 吕志伟, 王路路, 王亮君, 曹逊, 吴宇宁, 黄荣, 杨长. Cl掺杂对CuI薄膜发光性能增强研究[J]. 无机材料学报, 2023, 38(6): 687-692. |
| [12] | 安文然, 黄晶琪, 卢祥荣, 蒋佳宁, 邓龙辉, 曹学强. 热处理温度对LaMgAl11O19涂层热/力学性能的影响[J]. 无机材料学报, 2022, 37(9): 925-932. |
| [13] | 文志勤, 黄彬荣, 卢涛仪, 邹正光. 压力对PbTiO3结构和热物性质影响的第一性原理研究[J]. 无机材料学报, 2022, 37(7): 787-794. |
| [14] | 孙铭, 邵溥真, 孙凯, 黄建华, 张强, 修子扬, 肖海英, 武高辉. RGO/Al复合材料界面性质第一性原理研究[J]. 无机材料学报, 2022, 37(6): 651-659. |
| [15] | 肖美霞, 李苗苗, 宋二红, 宋海洋, 李钊, 毕佳颖. 表面端基卤化Ti3C2 MXene应用于锂离子电池高容量电极材料的研究[J]. 无机材料学报, 2022, 37(6): 660-668. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||