无机材料学报 ›› 2026, Vol. 41 ›› Issue (5): 645-652.DOI: 10.15541/jim20250373
李娜1( ), 魏进1, 曹锐霄1,2, 刘玉1, 黄贵文1, 肖红梅1(
), 魏进1, 曹锐霄1,2, 刘玉1, 黄贵文1, 肖红梅1( )
)
收稿日期:2025-09-26
修回日期:2025-11-26
出版日期:2026-01-06
网络出版日期:2026-01-06
通讯作者:
肖红梅, 研究员. E-mail: hmxiao@mail.ipc.ac.cn作者简介:李 娜(1987-), 女, 博士. E-mail: lina110@mail.ipc.ac.cn
基金资助:
LI Na1( ), WEI Jin1, CAO Ruixiao1,2, LIU Yu1, HUANG Guiwen1, XIAO Hongmei1(
), WEI Jin1, CAO Ruixiao1,2, LIU Yu1, HUANG Guiwen1, XIAO Hongmei1( )
)
Received:2025-09-26
Revised:2025-11-26
Published:2026-01-06
Online:2026-01-06
Contact:
XIAO Hongmei, professor. E-mail: hmxiao@mail.ipc.ac.cnAbout author:LI Na (1987-), female, PhD. E-mail: lina110@mail.ipc.ac.cn
Supported by:摘要:
正仲氢转化催化剂是大型氢液化工程中的关键材料之一。近年来, 研究大多聚焦于提升现有体系的低温催化活性, 而准确测量仲氢含量则是研究的基础。目前仅有少数研究以及相关行业规范对仲氢分析的测试精度与可靠性进行报道。除此之外, 批量生产的相关工艺研究报道仍是少之又少, 而在此基础上通过工艺优化指导批量催化剂的生产对于打破进口依赖尤为重要。本研究从正仲氢转化催化剂的催化测试分析和批量制备工艺优化两方面进行探讨。首先证明了所研制的测试平台获得的实验结果稳定性好、测量精度高, 在此基础上通过催化性能和机械强度的综合对比, 确定了产率最大化的最优批量生产工艺组合, 并通过低温活化再激活、粒径优化、二次洗涤等操作, 对批量制备工艺进行优化, 获得了催化活性更优的催化剂产品。结果表明, 自研催化剂2#样品(直接粉碎经0.8 mm筛网筛分、二次洗涤并低温活化)在空速为1.2 L/(min·mL)条件下的催化性能比进口催化剂高约3.4%, 相应的转化率和反应速率常数k分别比进口催化剂高约7.42%和25.78%。本研究证明了通过批量化制备亦能获得性能优异的正仲氢转化催化剂, 为国产化替代提供了重要的应用支撑。
中图分类号:
李娜, 魏进, 曹锐霄, 刘玉, 黄贵文, 肖红梅. 自研正仲氢转化催化剂的催化测试分析及批量制备工艺优化[J]. 无机材料学报, 2026, 41(5): 645-652.
LI Na, WEI Jin, CAO Ruixiao, LIU Yu, HUANG Guiwen, XIAO Hongmei. Self-developed Ortho-para Hydrogen Conversion Catalyst: Catalytic Testing and Optimization of Batch Preparation Process[J]. Journal of Inorganic Materials, 2026, 41(5): 645-652.
| Sample | Aperture of crushing sieve/mm | Particle size/mm | Granulation and post-processing technology | Secondary activation process |
|---|---|---|---|---|
| Commercial catalyst | / | 0.315-0.600 (30-50 mesh ) | / | / |
| 1# | 1.0 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | With low-temperature activation |
| 2# | 0.8 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | With low-temperature activation |
| 3# | 1.0 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | With low-temperature activation |
| 4# | 0.8 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | With low-temperature activation |
| 5# | 0.8 | 0.315-0.900 (20-50 mesh) | Without washing | / |
| 6# | 0.8 | 0.315-0.600 (30-50 mesh) | Primary crushing, washing | Without low-temperature activation |
| 7# | 0.8 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | Without low-temperature activation |
| 8# | 1.0 | 0.315-0.600 (30-50 mesh) | Secondary granulation, washing | Without low-temperature activation |
| 9# | 1.0 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | Without low-temperature activation |
表1 自研催化剂制备参数
Table 1 Preparation parameters of self-developed catalysts
| Sample | Aperture of crushing sieve/mm | Particle size/mm | Granulation and post-processing technology | Secondary activation process |
|---|---|---|---|---|
| Commercial catalyst | / | 0.315-0.600 (30-50 mesh ) | / | / |
| 1# | 1.0 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | With low-temperature activation |
| 2# | 0.8 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | With low-temperature activation |
| 3# | 1.0 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | With low-temperature activation |
| 4# | 0.8 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | With low-temperature activation |
| 5# | 0.8 | 0.315-0.900 (20-50 mesh) | Without washing | / |
| 6# | 0.8 | 0.315-0.600 (30-50 mesh) | Primary crushing, washing | Without low-temperature activation |
| 7# | 0.8 | 0.315-0.900 (20-50 mesh) | Primary crushing, washing | Without low-temperature activation |
| 8# | 1.0 | 0.315-0.600 (30-50 mesh) | Secondary granulation, washing | Without low-temperature activation |
| 9# | 1.0 | 0.315-0.900 (20-50 mesh) | Secondary granulation, washing | Without low-temperature activation |
| Parameter | Control sample | Test sample |
|---|---|---|
| Inner tube diameter | φ12.1 mm | φ8.9 mm |
| Working pressure | 0.1-0.2 MPa | 0.1-0.2 MPa |
| Hydrogen flow rate | 0.3-0.5 L/min | 0.4-1.6 L/min |
| Test section length | 65 mm | 32 mm |
| Catalyst packing volume | 4-5 mL | 1 mL |
表2 实验工况参数
Table 2 Experimental operating parameters
| Parameter | Control sample | Test sample |
|---|---|---|
| Inner tube diameter | φ12.1 mm | φ8.9 mm |
| Working pressure | 0.1-0.2 MPa | 0.1-0.2 MPa |
| Hydrogen flow rate | 0.3-0.5 L/min | 0.4-1.6 L/min |
| Test section length | 65 mm | 32 mm |
| Catalyst packing volume | 4-5 mL | 1 mL |
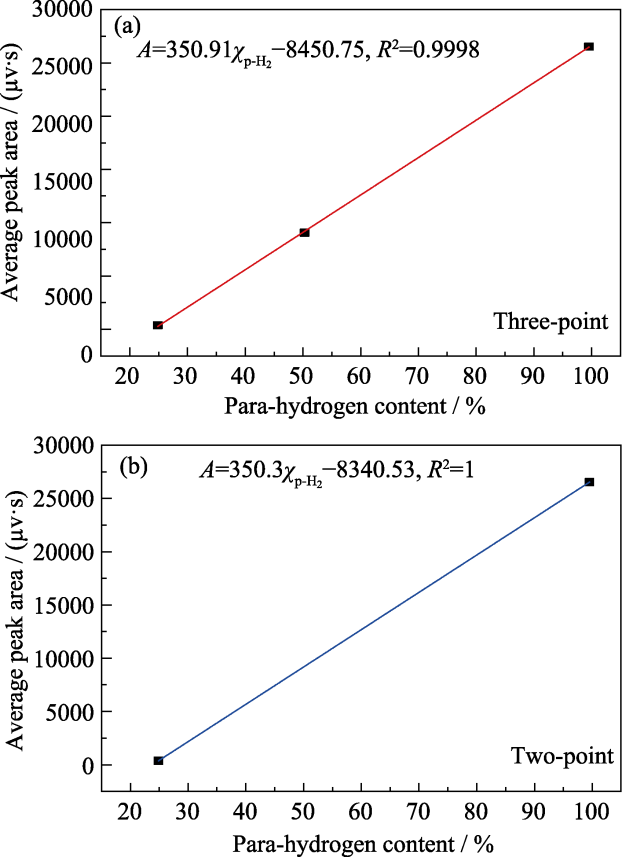
图3 (a)三点法和(b)两点法标准曲线及拟合的线性回归方程
Fig. 3 Standard curves and fitted linear regression equations using (a) the three-point method and (b) the two-point method
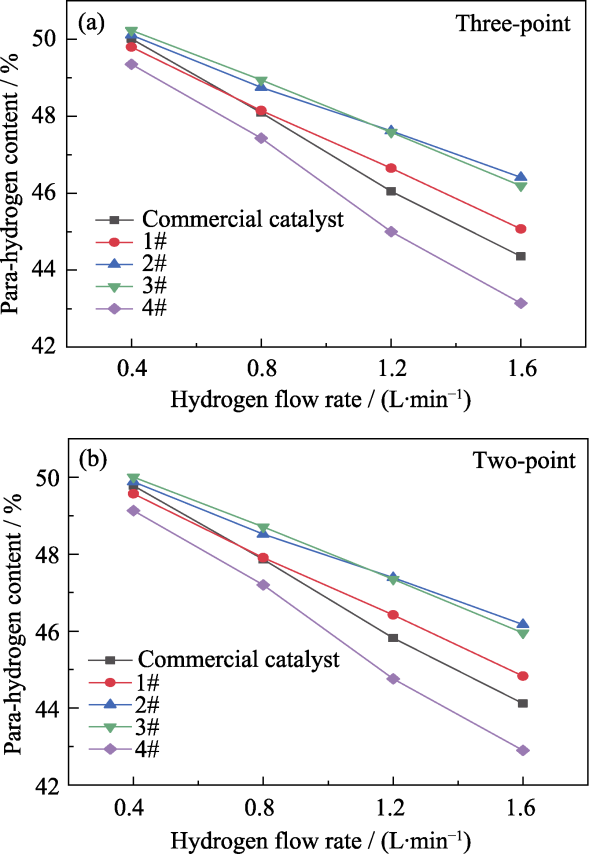
图4 (a)三点法和(b)两点法计算不同样品在77 K时的催化转化仲氢含量
Fig. 4 Para-hydrogen contents of different samples at 77 K calculated by (a) the three-point method and (b) the two-point method
| Hydrogen flow rate/ (L·min-1) | Calculation parameter | Commercial catalyst | 1# | 2# | 3# | 4# |
|---|---|---|---|---|---|---|
| 0.4 | k value/(mol·(L·s)-1) | 0.6697 | 0.5881 | 0.7330 | 0.8825 | 0.4920 |
| Catalytic conversion rate/% | 98.86 | 98.03 | 99.25 | 99.72 | 96.26 | |
| 0.8 | k value/(mol·(L·s)-1) | 0.7313 | 0.7396 | 0.8375 | 0.8767 | 0.6531 |
| Catalytic conversion rate/% | 91.31 | 91.54 | 93.90 | 94.65 | 88.71 | |
| 1.2 | k value/(mol·(L·s)-1) | 0.8033 | 0.8716 | 1.0104 | 1.0037 | 0.7041 |
| Catalytic conversion rate/% | 83.28 | 85.64 | 89.46 | 89.30 | 79.15 | |
| 1.6 | k value/(mol·(L·s)-1) | 0.8706 | 0.9468 | 1.1240 | 1.0911 | 0.7587 |
| Catalytic conversion rate/% | 76.63 | 79.43 | 84.70 | 83.83 | 71.83 |
表3 不同样品的反应速率常数k和催化转化率(三点法)
Table 3 Reaction rate constant (k) and catalytic conversion rates of various samples by the three-point method
| Hydrogen flow rate/ (L·min-1) | Calculation parameter | Commercial catalyst | 1# | 2# | 3# | 4# |
|---|---|---|---|---|---|---|
| 0.4 | k value/(mol·(L·s)-1) | 0.6697 | 0.5881 | 0.7330 | 0.8825 | 0.4920 |
| Catalytic conversion rate/% | 98.86 | 98.03 | 99.25 | 99.72 | 96.26 | |
| 0.8 | k value/(mol·(L·s)-1) | 0.7313 | 0.7396 | 0.8375 | 0.8767 | 0.6531 |
| Catalytic conversion rate/% | 91.31 | 91.54 | 93.90 | 94.65 | 88.71 | |
| 1.2 | k value/(mol·(L·s)-1) | 0.8033 | 0.8716 | 1.0104 | 1.0037 | 0.7041 |
| Catalytic conversion rate/% | 83.28 | 85.64 | 89.46 | 89.30 | 79.15 | |
| 1.6 | k value/(mol·(L·s)-1) | 0.8706 | 0.9468 | 1.1240 | 1.0911 | 0.7587 |
| Catalytic conversion rate/% | 76.63 | 79.43 | 84.70 | 83.83 | 71.83 |

图8 (a, b)一次粉碎和(c, d)二次造粒后(a, c)未洗和(b, d)已洗样品的SEM形貌对比图
Fig. 8 SEM morphology comparison of the prepared samples (a, c) before and (b, d) after washing following (a, b) primary crushing and (c, d) secondary granulation
| Peak area | Hydrogen flow rate | Average value | RSD/% | Equilibrium para-hydrogen content/% | ||
|---|---|---|---|---|---|---|
| 0.3 L/min | 0.4 L/min | 0.5 L/min | ||||
| Peak area at 298 K/(μV·s) | 378.5 | 356.6 | 252.1 | 328.1 | 16.97 | 24.88 |
| 371.1 | 342.9 | 244.9 | ||||
| 375.1 | 329.7 | 284.3 | ||||
| Average peak area/(μV·s) | 374.9 | 343.0 | 266.6 | |||
| Corrected | 374.9 | 0.99 | ||||
| Peak area at 77 K/(μV·s) | 9131.7 | 9021.9 | 8919.2 | 9056.3 | 0.89 | 50.30 |
| 9161.6 | 9030.5 | 9020.0 | ||||
| 9131.6 | 9092.6 | 8995.6 | ||||
| Average peak area/(μV·s) | 9141.6 | 9048.3 | 8979.2 | |||
| Peak area at 21 K/(μV·s) | 26481.2 | 26501.7 | 26622.3 | 26531.7 | 0.37 | 99.55 |
| 26477.7 | 26486.8 | 26602.1 | ||||
| 26461.9 | 26444.0 | 26708.0 | ||||
| Average peak area/(μV·s) | 26473.6 | 26477.5 | 26644.1 | |||
表S1 不同温度下的标定数值
Table S1 Calibration data at different temperatures
| Peak area | Hydrogen flow rate | Average value | RSD/% | Equilibrium para-hydrogen content/% | ||
|---|---|---|---|---|---|---|
| 0.3 L/min | 0.4 L/min | 0.5 L/min | ||||
| Peak area at 298 K/(μV·s) | 378.5 | 356.6 | 252.1 | 328.1 | 16.97 | 24.88 |
| 371.1 | 342.9 | 244.9 | ||||
| 375.1 | 329.7 | 284.3 | ||||
| Average peak area/(μV·s) | 374.9 | 343.0 | 266.6 | |||
| Corrected | 374.9 | 0.99 | ||||
| Peak area at 77 K/(μV·s) | 9131.7 | 9021.9 | 8919.2 | 9056.3 | 0.89 | 50.30 |
| 9161.6 | 9030.5 | 9020.0 | ||||
| 9131.6 | 9092.6 | 8995.6 | ||||
| Average peak area/(μV·s) | 9141.6 | 9048.3 | 8979.2 | |||
| Peak area at 21 K/(μV·s) | 26481.2 | 26501.7 | 26622.3 | 26531.7 | 0.37 | 99.55 |
| 26477.7 | 26486.8 | 26602.1 | ||||
| 26461.9 | 26444.0 | 26708.0 | ||||
| Average peak area/(μV·s) | 26473.6 | 26477.5 | 26644.1 | |||
| Sample No. | Test data | Hydrogen flow rate | |||
|---|---|---|---|---|---|
| 0.4 L/min | 0.8 L/min | 1.2 L/min | 1.6 L/min | ||
| Commercial catalyst | Average peak area/(μV·s) | 9096.6 | 8425.2 | 7709.0 | 7116.4 |
| RSD/% | 0.21 | 0.63 | 0.31 | 0.31 | |
| 1# | Average peak area/(μV·s) | 9025.0 | 8444.0 | 7920.3 | 7365.0 |
| RSD/% | 0.38 | 0.27 | 0.18 | 0.24 | |
| 2# | Average peak area/(μV·s) | 9134.1 | 8656.6 | 8261.8 | 7833.5 |
| RSD/% | 0.25 | 0.17 | 0.38 | 0.33 | |
| 3# | Average peak area/(μV·s) | 9174.6 | 8721.3 | 8244.6 | 7756.4 |
| RSD/% | 0.27 | 0.10 | 0.33 | 0.12 | |
| 4# | Average peak area/(μV·s) | 8868.2 | 8192.3 | 7339.9 | 6687.0 |
| RSD/% | 0.12 | 0.13 | 0.42 | 0.19 | |
表S2 77 K时不同样品的催化测试数值
Table S2 Catalytic test data for various samples at 77 K
| Sample No. | Test data | Hydrogen flow rate | |||
|---|---|---|---|---|---|
| 0.4 L/min | 0.8 L/min | 1.2 L/min | 1.6 L/min | ||
| Commercial catalyst | Average peak area/(μV·s) | 9096.6 | 8425.2 | 7709.0 | 7116.4 |
| RSD/% | 0.21 | 0.63 | 0.31 | 0.31 | |
| 1# | Average peak area/(μV·s) | 9025.0 | 8444.0 | 7920.3 | 7365.0 |
| RSD/% | 0.38 | 0.27 | 0.18 | 0.24 | |
| 2# | Average peak area/(μV·s) | 9134.1 | 8656.6 | 8261.8 | 7833.5 |
| RSD/% | 0.25 | 0.17 | 0.38 | 0.33 | |
| 3# | Average peak area/(μV·s) | 9174.6 | 8721.3 | 8244.6 | 7756.4 |
| RSD/% | 0.27 | 0.10 | 0.33 | 0.12 | |
| 4# | Average peak area/(μV·s) | 8868.2 | 8192.3 | 7339.9 | 6687.0 |
| RSD/% | 0.12 | 0.13 | 0.42 | 0.19 | |
| Hydrogen flow rate/(L·min-1) | Para-hydrogen content/% | Calibration method | ||||
|---|---|---|---|---|---|---|
| Commercial catalyst | 1# | 2# | 3# | 4# | ||
| 0.4 | 50.01 | 49.80 | 50.11 | 50.23 | 49.35 | Three-point method |
| 0.8 | 48.09 | 48.15 | 48.75 | 48.94 | 47.43 | |
| 1.2 | 46.05 | 46.65 | 47.62 | 47.58 | 45.00 | |
| 1.6 | 44.36 | 45.07 | 46.41 | 46.19 | 43.14 | |
| 0.4 | 49.78 | 49.57 | 49.88 | 50.00 | 49.13 | Two-point method |
| 0.8 | 47.86 | 47.91 | 48.52 | 48.71 | 47.20 | |
| 1.2 | 45.82 | 46.42 | 47.39 | 47.35 | 44.76 | |
| 1.6 | 44.12 | 44.83 | 46.17 | 45.95 | 42.90 | |
表S3 分别利用三点法和两点法计算77 K时不同样品的仲氢含量
Table S3 Para-hydrogen content of various samples at 77 K calculated by three-point and two-point methods
| Hydrogen flow rate/(L·min-1) | Para-hydrogen content/% | Calibration method | ||||
|---|---|---|---|---|---|---|
| Commercial catalyst | 1# | 2# | 3# | 4# | ||
| 0.4 | 50.01 | 49.80 | 50.11 | 50.23 | 49.35 | Three-point method |
| 0.8 | 48.09 | 48.15 | 48.75 | 48.94 | 47.43 | |
| 1.2 | 46.05 | 46.65 | 47.62 | 47.58 | 45.00 | |
| 1.6 | 44.36 | 45.07 | 46.41 | 46.19 | 43.14 | |
| 0.4 | 49.78 | 49.57 | 49.88 | 50.00 | 49.13 | Two-point method |
| 0.8 | 47.86 | 47.91 | 48.52 | 48.71 | 47.20 | |
| 1.2 | 45.82 | 46.42 | 47.39 | 47.35 | 44.76 | |
| 1.6 | 44.12 | 44.83 | 46.17 | 45.95 | 42.90 | |
| [1] |
XU P, WEN J, LI K, et al. Review of the continuous catalytic ortho-para hydrogen conversion technology for hydrogen liquefaction. International Journal of Hydrogen Energy, 2024, 62: 473.
DOI URL |
| [2] | 唐璐, 邱利民, 姚蕾, 等. 氢液化系统的研究进展与展望. 制冷学报, 2011, 32(6): 1. |
| [3] |
YIN L, JU Y L. Review on the design and optimization of hydrogen liquefaction processes. Frontiers in Energy, 2020, 14(3): 530.
DOI |
| [4] |
ZHUZHGOV A V, KRIVORUCHKO O P, ISUPOVA L A, et al. Low-temperature conversion of ortho-hydrogen into liquid para- hydrogen: process and catalysts. Review. Catalysis in Industry, 2018, 10(1): 9.
DOI |
| [5] |
LARSEN A H, SIMON F E, SWENSON C A. The rate of evaporation of liquid hydrogen due to the ortho-para hydrogen conversion. Review of Scientific Instruments, 1948, 19(4): 266.
DOI URL |
| [6] |
ILISCA E. Ortho-para conversion of hydrogen molecules physisorbed on surfaces. Progress in Surface Science, 1992, 41(3): 217.
DOI URL |
| [7] |
XU Y F, BI Y J, JU Y L. The thermodynamic analysis on the catalytical ortho-para hydrogen conversion during the hydrogen liquefaction process. International Journal of Hydrogen Energy, 2024, 54: 1329.
DOI URL |
| [8] |
DAS T, NAH I W, CHOI J G, et al. Synthesis of iron oxide catalysts using various methods for the spin conversion of hydrogen. Reaction Kinetics, Mechanisms and Catalysis, 2016, 118(2): 669.
DOI URL |
| [9] |
SULLIVAN N S, ZHOU D, EDWARDS C M. Precise and efficient in situ ortho-para-hydrogen converter. Cryogenics, 1990, 30(8): 734.
DOI URL |
| [10] |
KIM J H, KARNG S W, OH I H, et al. Ortho-para hydrogen conversion characteristics of amorphous and mesoporous Cr2O3 powders at a temperature of 77 K. International Journal of Hydrogen Energy, 2015, 40(41): 14147.
DOI URL |
| [11] |
WAKAO N, SMITH J M, SELWOOD P W. The low-temperature orthohydrogen conversion over supported oxides and metals. Journal of Catalysis, 1962, 1(1): 62.
DOI URL |
| [12] | 花亦怀, 李秋英, 程昊, 等. 正仲氢催化转化机理研究综述. 制冷学报, 2025, 46(5): 12. |
| [13] |
TURRO N J, CHEN J Y, SARTORI E, et al. The spin chemistry and magnetic resonance of H2@C60. From the Pauli principle to trapping a long lived nuclear excited spin state inside a buckyball. Accounts of Chemical Research, 2010, 43(2): 335.
DOI PMID |
| [14] |
BOEVA O, ANTONOV A, ZHAVORONKOVA K. Influence of the nature of IB group metals on catalytic activity in reactions of homomolecular hydrogen exchange on Cu, Ag, Au nanoparticles. Catalysis Communications, 2021, 148: 106173.
DOI URL |
| [15] |
BOEVA O A, ODINTZOV A A, SOLOVOV R D, et al. Low-temperature ortho-para hydrogen conversion catalyzed by gold nanoparticles: particle size does not affect the rate. International Journal of Hydrogen Energy, 2017, 42(36): 22897.
DOI URL |
| [16] |
CHEN Y, YANG L J, LI X B. Preparation and study of high efficiency M-BTC ortho-para hydrogen conversion catalysts. International Journal of Hydrogen Energy, 2024, 81: 10.
DOI URL |
| [17] |
POLYUKHOV D M, KUDRIAVYKH N A, GROMILOV S A, et al. Efficient MOF-catalyzed ortho-para hydrogen conversion for practical liquefaction and energy storage. ACS Energy Letters, 2022, 7(12): 4336.
DOI URL |
| [18] |
WANG J Y, YUE C Z, ZHAO J, et al. Doping-induced structural transformations in maghemite for enhanced ortho-para hydrogen conversion. Catalysis Today, 2025, 452: 115243.
DOI URL |
| [19] |
ZHAO Z Z, LI A Q, CHEN Y S, et al. Insights into the facet dependent conversion of ortho- to para-hydrogen over α-Fe2O3 nanocrystals. International Journal of Hydrogen Energy, 2025, 123: 281.
DOI URL |
| [20] |
YUE C Z, WANG J Y, WANG S F, et al. Identification of structural factors in iron oxide triggering ortho-para hydrogen conversion. The Journal of Physical Chemistry C, 2024, 128(30): 12355.
DOI URL |
| [21] |
CHEN Y S, ZHUO H Y, SHEN Z, et al. Catalytic mechanism studies of ortho-para H2 conversion over iron oxide catalysts. EcoEnergy, 2025, 3(3): e70004.
DOI URL |
| [22] | 陈志强, 汪丽, 丁明伟, 等. 封装型α-Fe2O3@SiO2催化剂的制备及其催化正仲氢转化性能评价. 低碳化学与化工, 2024, 49(9): 106. |
| [23] | 范质, 吴俊哲, 杨昌乐, 等. 正仲氢转化催化剂性能实验研究. 低温工程, 2025(1): 65. |
| [24] |
XU H, WANG J W, HAN Y S, et al. Effect of unpaired electron number elements (Al, Cr, Mn) doping in Fe2O3 on ortho to para hydrogen conversion at 77 K. Journal of Energy Storage, 2023, 74: 109512.
DOI URL |
| [25] |
XU H, BI S H, XUE M Z, et al. Amorphous cobalt iron oxide nanoparticles with high magnetization intensity for spin conversion of hydrogen at 77 K. International Journal of Hydrogen Energy, 2023, 48(81): 31643.
DOI URL |
| [26] |
XUE M Z, XU H, SHEN J, et al. A high specific surface area and amorphous cobalt oxide@molecular sieve supported catalyst for ortho-to para-hydrogen conversion. International Journal of Hydrogen Energy, 2025, 103: 341.
DOI URL |
| [27] |
LI N, CAO R X, WEI J, et al. Performance and influencing factors of iron-based catalyst for ortho to para hydrogen conversion. Journal of Inorganic Materials, 2025, 40(1): 47.
DOI URL |
| [28] | WEITZEL D H, BLAKE J H, KONECNIK M. Boston, Flow conversion kinetics of ortho and parahydrogen. Advances in Cryogenic Engineering, Boston, MA, 1960. |
| [1] | 薛新燕, 张晓卫, 鲁恒, 李世杰, 张卫珂, 曾少华. 钴-镁协同纳米洋葱碳催化剂的制备及低温催化氨分解制氢性能[J]. 无机材料学报, 2026, 41(4): 500-508. |
| [2] | 黄应贺, 黄仁兴, 石宇星, 雷一杰, 于涛, 王诚, 顾军. 利用介孔碳的限域效应提升氧还原反应中Pt催化剂的耐久性[J]. 无机材料学报, 2026, 41(3): 289-294. |
| [3] | 任先培, 李超, 胡启威, 向晖, 彭跃红. 金属/过渡金属化合物莫特-肖特基析氢催化剂研究进展[J]. 无机材料学报, 2026, 41(2): 137-149. |
| [4] | 邬博宇, 张深根, 张生杨, 刘波, 张柏林. CeO2对MnOx催化剂低温脱硝性能的影响及其机理研究[J]. 无机材料学报, 2026, 41(1): 87-95. |
| [5] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [6] | 郭子玉, 朱云洲, 王力, 陈健, 李红, 黄政仁. Zn2+催化剂对酚醛树脂/乙二醇制备多孔碳微观孔结构的影响[J]. 无机材料学报, 2025, 40(5): 466-472. |
| [7] | 李建军, 陈芳明, 张梨梨, 王磊, 张丽亭, 陈慧雯, 薛长国, 徐良骥. CoFe2O4/MgAl-LDH催化剂活化过氧一硫酸盐促进抗生素降解[J]. 无机材料学报, 2025, 40(4): 440-448. |
| [8] | 信震宇, 郭瑞华, 乌仁托亚, 王艳, 安胜利, 张国芳, 关丽丽. Pt-Fe/GO纳米催化剂的制备及其电催化乙醇氧化性能研究[J]. 无机材料学报, 2025, 40(4): 379-387. |
| [9] | 孙树娟, 郑南南, 潘昊坤, 马猛, 陈俊, 黄秀兵. 单原子催化剂制备方法的研究进展[J]. 无机材料学报, 2025, 40(2): 113-127. |
| [10] | 李薛茹, 马哲杰, 郭宇杰, 李平. 载体特性对Pt/C催化剂上离聚物覆盖度及氧还原性能的影响[J]. 无机材料学报, 2025, 40(12): 1395-1404. |
| [11] | 赵丽娟, 谭哲, 张晓光, 蒋国赛, 陶然, 潘德安. 废加氢催化剂颗粒分级数值模拟研究[J]. 无机材料学报, 2025, 40(12): 1387-1394. |
| [12] | 刘盼盼, 姚鹏, 刘栩孜, 屈丽, 曾路, 宋兆华, 焦毅, 王健礼, 陈耀强. MnOx/CeO2-ZrO2复合氧化物的构筑及其在碳烟氧化中的应用[J]. 无机材料学报, 2025, 40(11): 1300-1308. |
| [13] | 李娜, 曹锐霄, 魏进, 周晗, 肖红梅. 铁基正仲氢转化催化剂的影响因素[J]. 无机材料学报, 2025, 40(1): 47-52. |
| [14] | 连敏丽, 苏佳欣, 黄鸿杨, 嵇玉寅, 邓海帆, 张彤, 陈崇启, 李达林. Ni-Mg-Al类水滑石衍生镍基催化剂的制备及其氨分解性能[J]. 无机材料学报, 2025, 40(1): 53-60. |
| [15] | 刘磊, 郭瑞华, 王丽, 王艳, 张国芳, 关丽丽. Pt3Co高指数晶面氧还原过程的密度泛函理论研究[J]. 无机材料学报, 2025, 40(1): 39-46. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||