无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 469-478.DOI: 10.15541/jim20250187 CSTR: 32189.14.10.15541/jim20250187
收稿日期:2025-05-03
修回日期:2025-07-23
出版日期:2026-04-20
网络出版日期:2025-09-11
通讯作者:
张卫珂, 副教授. E-mail: zhangweike@tyut.edu.cn作者简介:马晓佳(2000-), 女, 硕士研究生. E-mail: maxiaojia0209@link.tyut.edu.cn
基金资助:
MA Xiaojia( ), GENG Xinyu, ZHANG Weike(
), GENG Xinyu, ZHANG Weike( )
)
Received:2025-05-03
Revised:2025-07-23
Published:2026-04-20
Online:2025-09-11
Contact:
ZHANG Weike, associate professor. E-mail: zhangweike@tyut.edu.cnAbout author:MA Xiaojia (2000-), female, Master candidate. E-mail: maxiaojia0209@link.tyut.edu.cn
Supported by:摘要:
硬碳具有成本低廉、来源广泛、使用寿命长等优点, 是一种极具发展前景的钠离子电池负极材料。然而, 其较低的首次库仑效率(ICE)和较差的倍率性能, 限制了实际应用。目前, 杂原子掺杂是调控无定形碳微观结构, 提高碳基材料储钠性能的有效方法。与单原子掺杂相比, 多原子掺杂产生的协同效应有利于增强材料的电化学反应活性。本研究以从马铃薯淀粉加工废液中提取的废渣为前驱体, 通过水热反应合成了碳球, 在此基础上采用尿素、四硼酸钠作为掺杂源, 利用球磨和热解的方法制备了硼氮共掺杂生物质碳球。随后, 探究了多原子掺杂对碳材料微观形貌及储钠性能的影响。结果表明: 硼、氮共掺杂提高了材料的无序度, 扩大了层间距, 同时形成了合适的、有利于生成稳定固体电解质界面膜的C=O键。在50 mA·g-1的电流密度下, 其可逆比容量为284.3 mAh·g-1, ICE为77.0%。在2 A·g-1下循环500圈后, 比容量衰减至122.5 mAh·g-1, 容量保持率为56.1%。
中图分类号:
马晓佳, 耿欣宇, 张卫珂. 硼氮共掺杂生物质碳球负极材料的制备及其储钠性能[J]. 无机材料学报, 2026, 41(4): 469-478.
MA Xiaojia, GENG Xinyu, ZHANG Weike. Boron and Nitrogen Co-doped Biomass Carbon Sphere Anode Material: Preparation and Sodium Storage Properties for Sodium-ion Batteries[J]. Journal of Inorganic Materials, 2026, 41(4): 469-478.
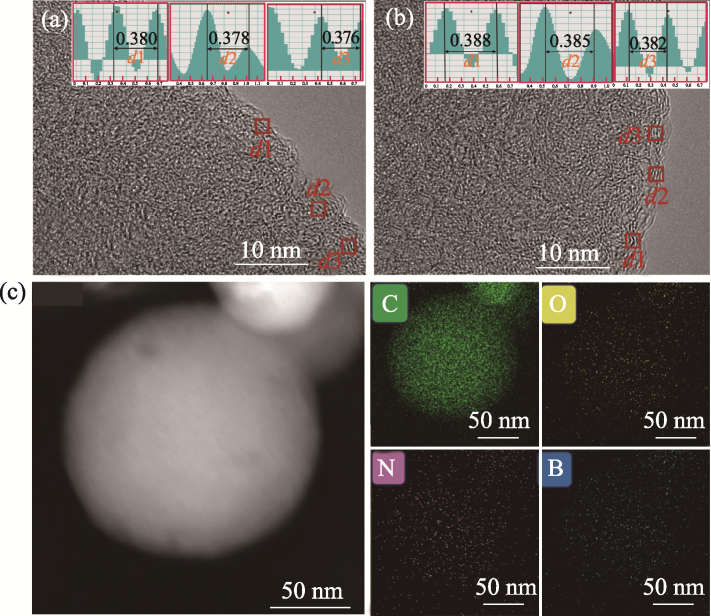
图3 (a) NHCS-900和(b) B@N@NHCS-900的HRTEM照片(插图为方框区域层间距的FFT谱图); (c) B@N@NHCS-900的STEM照片及C、O、N、B元素分布图
Fig. 3 (a, b) HRTEM images of (a) NHCS-900 and (b) B@N@NHCS-900 with inserts showing the corresponding FFT patterns for measuring the interlayer distance in square areas; (c) STEM image and C, O, N and B element mappings of B@N@NHCS-900
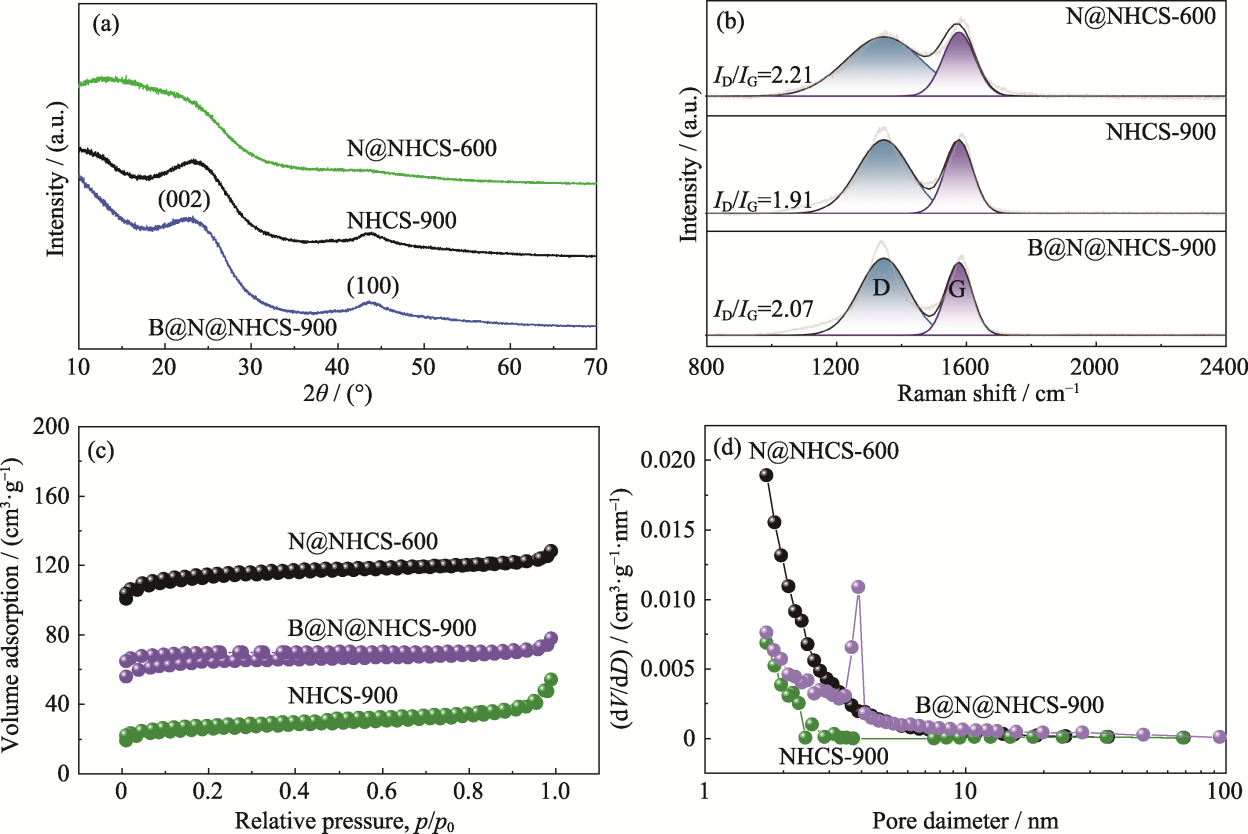
图4 N@NHCS-600、B@N@NHCS-900和NHCS-900的(a) XRD图谱、(b)拉曼谱图、(c)氮气吸脱附等温曲线和(d)孔径分布图
Fig. 4 (a) XRD patterns, (b) Raman spectra, (c) N2 adsorption-desorption isotherms and (d) pore size distributions of N@NHCS-600, B@N@NHCS-900 and NHCS-900
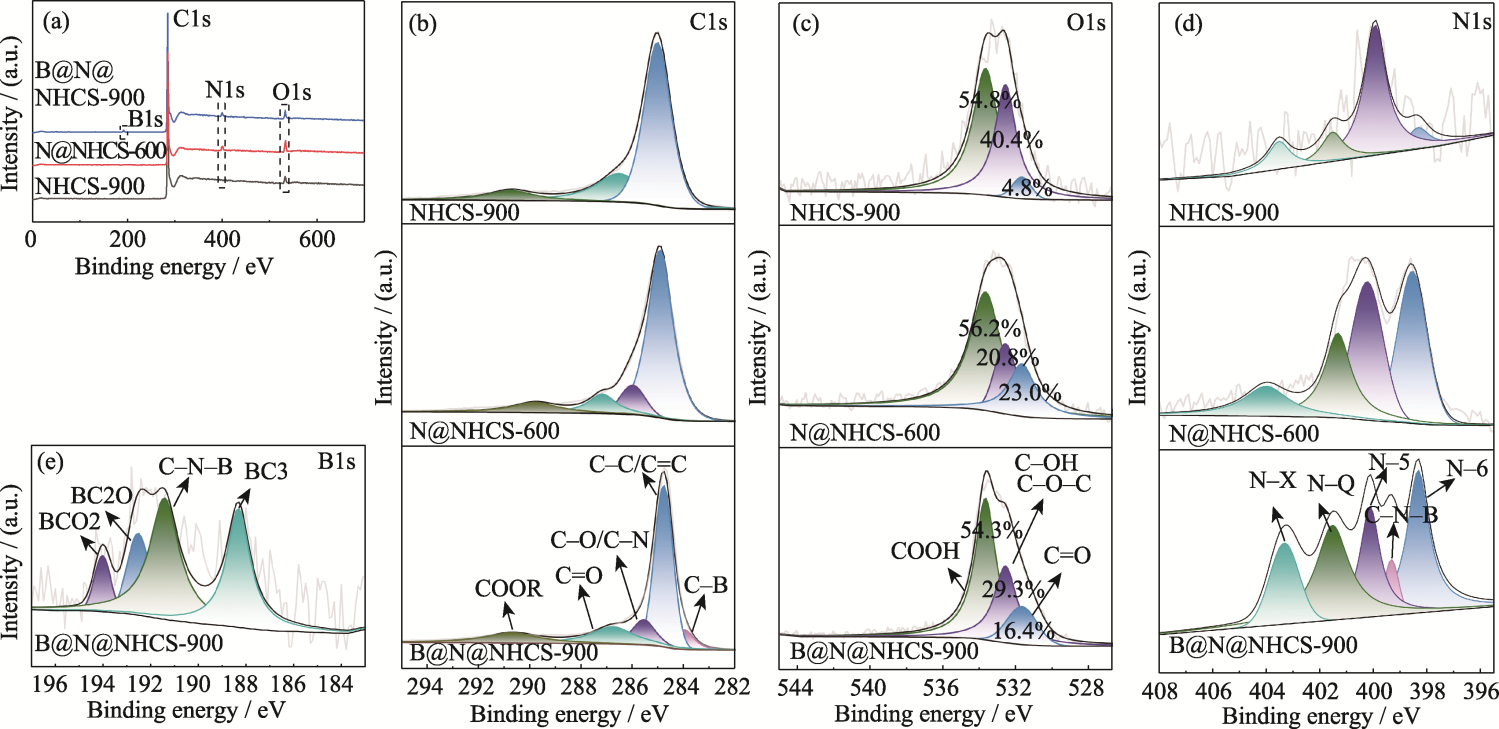
图5 N@NHCS-600、B@N@NHCS-900和NHCS-900的(a) XPS总谱图, 高分辨(b) C1s、(c) O1s和(d) N1s XPS谱图; (e) B@N@NHCS-900的高分辨B1s XPS谱图
Fig. 5 (a) XPS survey spectra, high resolution (b) C1s, (c) O1s and (d) N1s XPS spectra of N@NHCS-600, B@N@NHCS-900 and NHCS-900; (e) High resolution B1s XPS spectrum of B@N@NHCS-900
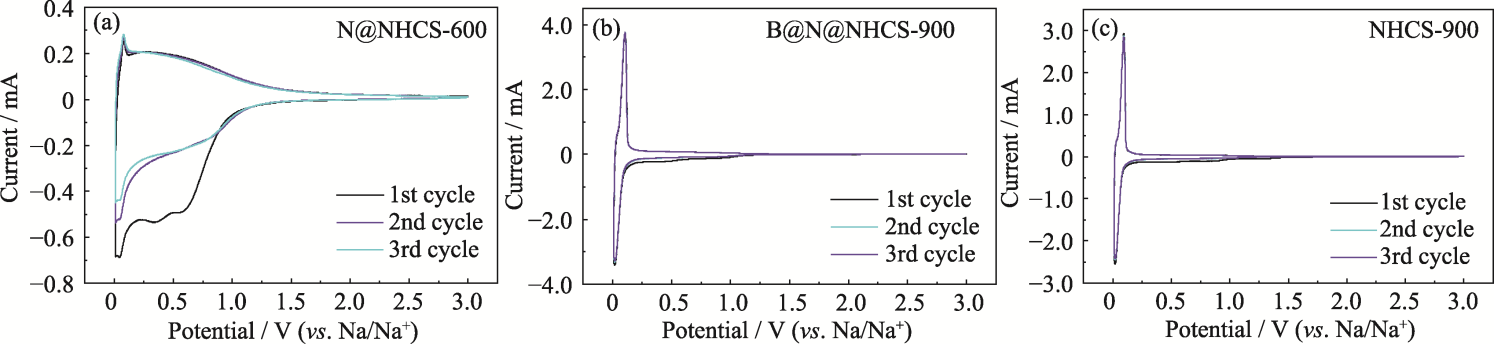
图6 (a) N@NHCS-600、(b) B@N@NHCS-900和(c) NHCS-900的CV曲线
Fig. 6 CV curves of (a) N@NHCS-600, (b) B@N@NHCS-900 and (c) NHCS-900 Colorful figures are available on website
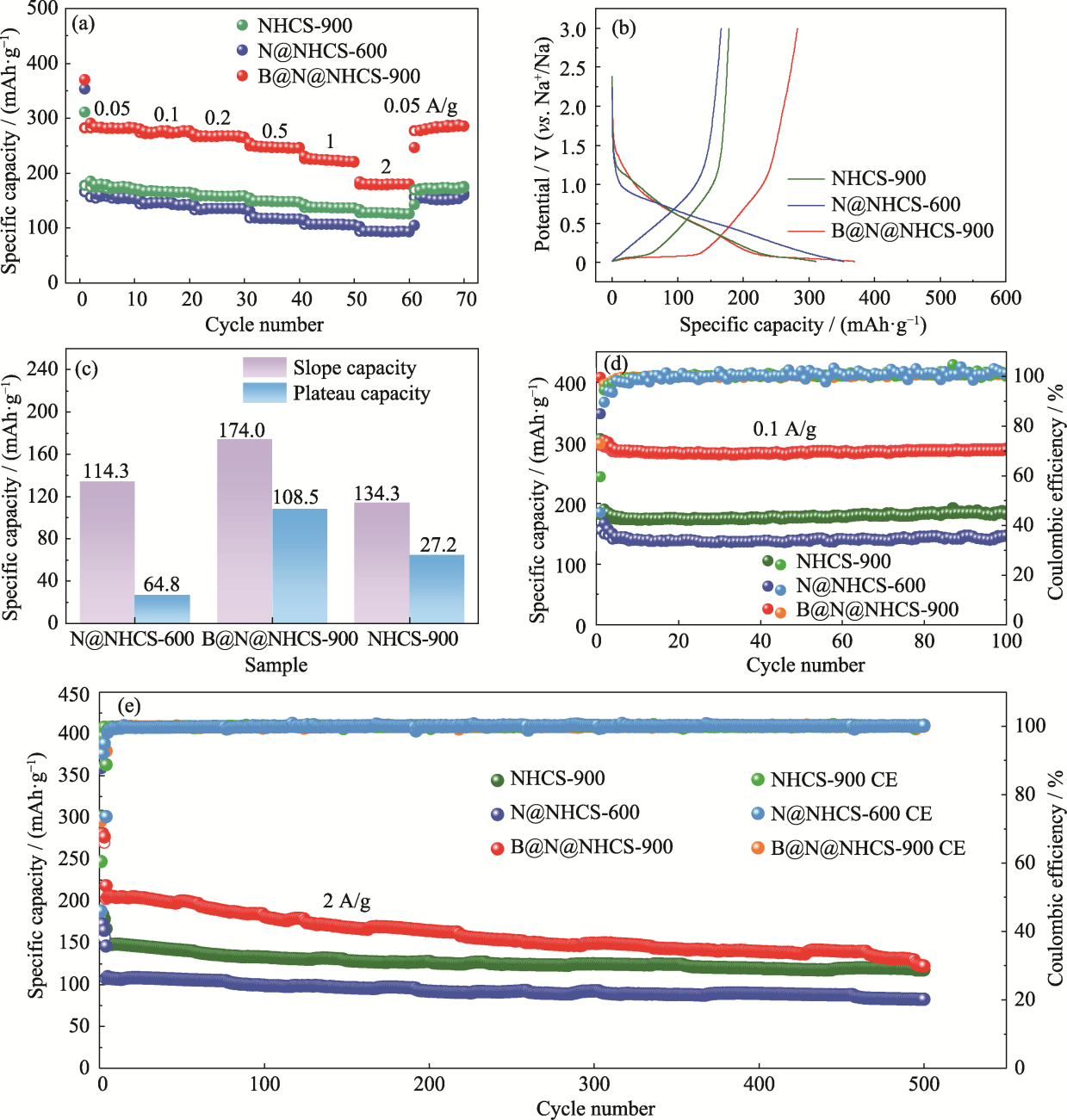
图7 N@NHCS-600、B@N@NHCS-900和NHCS-900的电化学性能
Fig. 7 Electrochemical properties of N@NHCS-600, B@N@NHCS-900 and NHCS-900 (a) Rate capability; (b) First charge-discharge curves at a current density of 0.05 A·g-1; (c) Capacity contribution of the slope/plateau region in the 2nd discharge curves; (d, e) Cycling performance at (d) 0.1 and (e) 2 A·g-1. Colorful figures are available on website
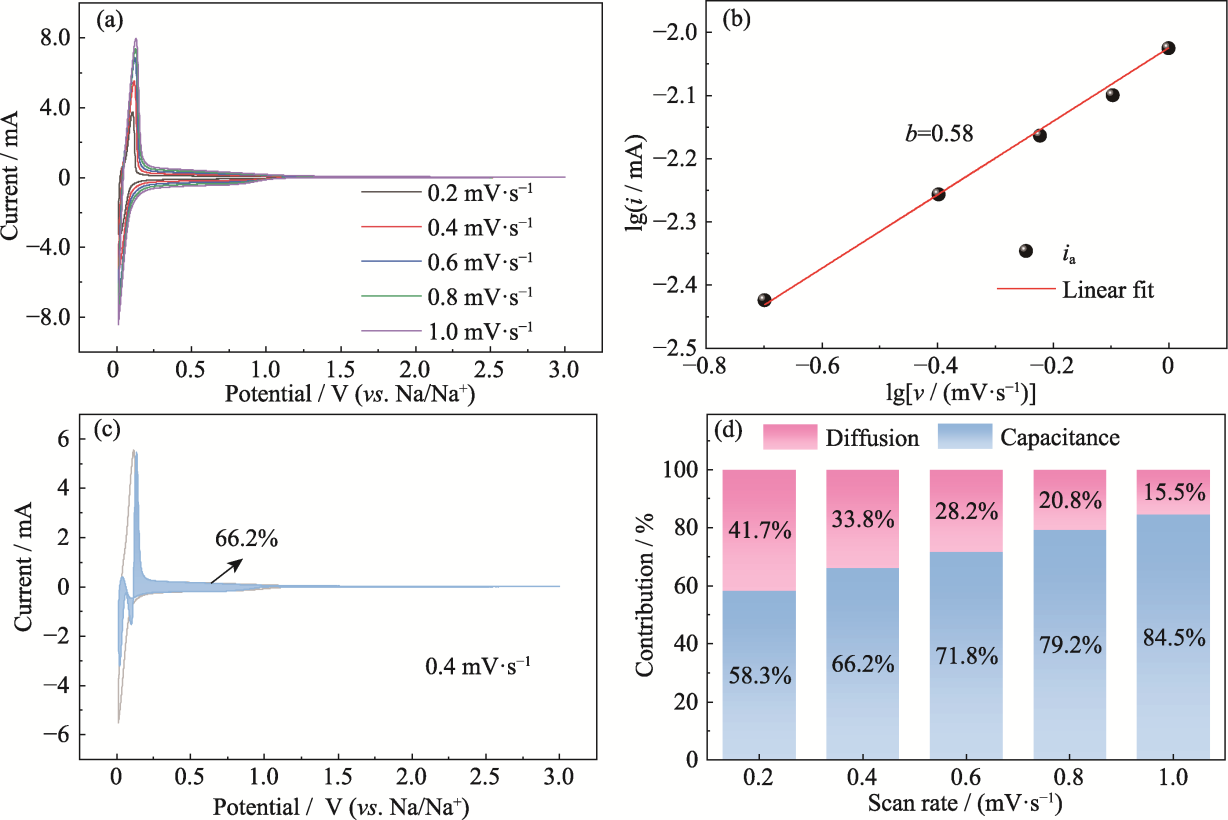
图8 B@N@NHCS-900的动力学分析
Fig. 8 Dynamic analysis of B@N@NHCS-900 (a) CV curves from 0.2 to 1.0 mV·s-1; (b) Fitting line of scan rate and peak current; (c) Capacitive contribution at 0.4 mV·s-1; (d) Ratios of diffusion and capacitance contributions at different scan rates. Colorful figures are available on website
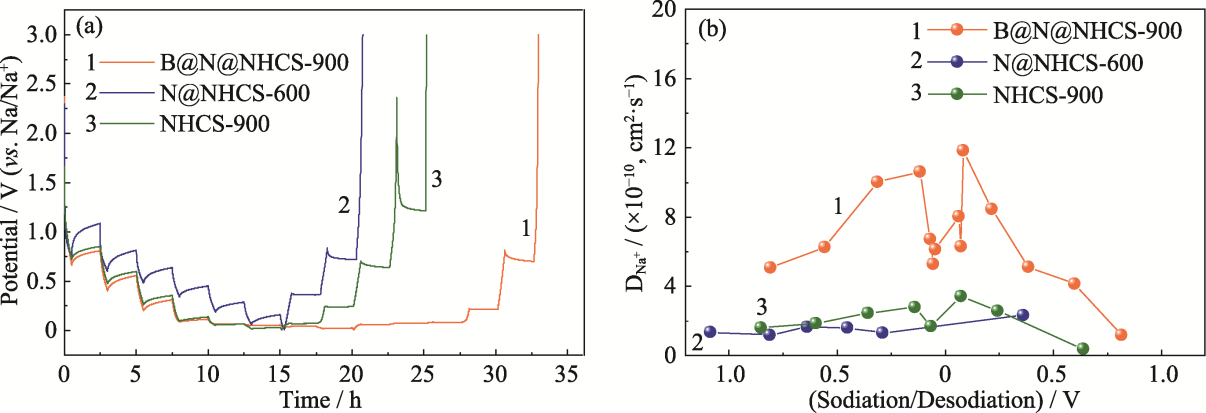
图9 N@NHCS-600、B@N@NHCS-900和NHCS-900的(a) GITT曲线和(b)扩散系数曲线
Fig. 9 (a) GITT curves and (b) corresponding diffusion coefficients of N@NHCS-600, B@N@NHCS-900 and NHCS-900
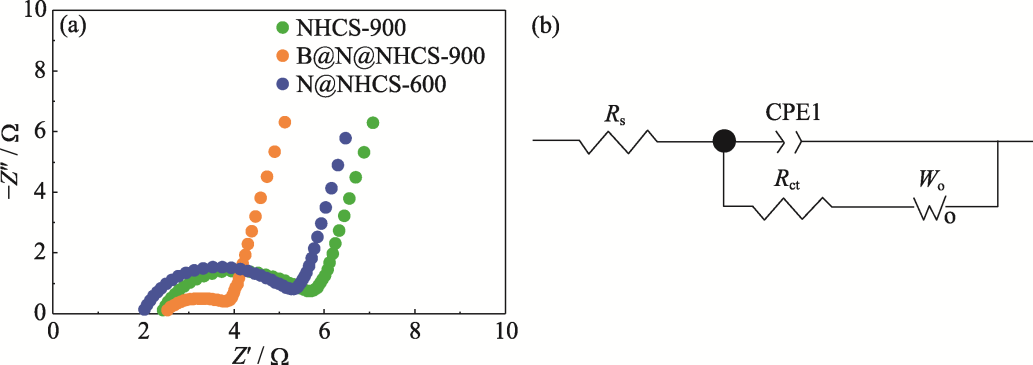
图10 N@NHCS-600、B@N@NHCS-900和NHCS-900的(a) EIS谱图和(b)等效电路图
Fig. 10 (a) EIS spectra of N@NHCS-600, B@N@NHCS-900 and NHCS-900, and (b) corresponding equivalent circuit Colorful figure is available on website
| Sample | d(002)/nm | ID/IG | SBET/(m2·g-1) | Vtotal/(cm3·g-1) | XPS content/% (in atom) | |||
|---|---|---|---|---|---|---|---|---|
| C | O | N | B | |||||
| N@NHCS-600 | - | 2.21 | 442.4 | 0.189 | 89.52 | 6.79 | 3.69 | - |
| B@N@NHCS-900 | 0.385 | 2.07 | 249.1 | 0.109 | 89.49 | 5.89 | 2.98 | 1.64 |
| NHCS-900 | 0.378 | 1.91 | 94.9 | 0.064 | 95.96 | 3.56 | 0.48 | - |
表S1 不同样品的结构参数
Table S1 Structural parameters of different samples
| Sample | d(002)/nm | ID/IG | SBET/(m2·g-1) | Vtotal/(cm3·g-1) | XPS content/% (in atom) | |||
|---|---|---|---|---|---|---|---|---|
| C | O | N | B | |||||
| N@NHCS-600 | - | 2.21 | 442.4 | 0.189 | 89.52 | 6.79 | 3.69 | - |
| B@N@NHCS-900 | 0.385 | 2.07 | 249.1 | 0.109 | 89.49 | 5.89 | 2.98 | 1.64 |
| NHCS-900 | 0.378 | 1.91 | 94.9 | 0.064 | 95.96 | 3.56 | 0.48 | - |
| Sample | Rate performance | Cycling performance | Ref. | |||
|---|---|---|---|---|---|---|
| Capacity/(mAh·g-1) | Current density/(A·g-1) | Capacity/(mAh·g-1) | Current density /(A·g-1) | Cycle number | ||
| BHCS-1200 | 234 | 0.03 | 180 | 0.1 | 100 | [ |
| Below 100 | 2 | |||||
| NPUCS | 257.7 | 0.1 | 255.1 | 0.1 | 200 | [ |
| 157.0 | 5 | 110.0 | 5 | 2000 | ||
| PB-1000 | 205 | 0.1 | 146.9 | 2 | 600 | [ |
| 136.6 | 2 | |||||
| CPB-PL | 293 | 0.02 | 193 | 0.1 | 200 | [ |
| 77 | 1 | |||||
| SC-800 | 229 | 0.05 | 189 | 0.05 | 50 | [ |
| 82.0 | 1 | 81.0 | 0.5 | 2000 | ||
| SAL | 285 | 0.02 | 141.6 | 0.2 | 1000 | [ |
| 150 | 0.5 | |||||
| HC-1000 | 242.1 | 0.025 | Below 60 | 1 | 800 | [ |
| Below 100 | 0.8 | |||||
| YT-1200 | 261 | 0.1 | 235 | 0.1 | 200 | [ |
| 206 | 2 | |||||
| N-FLG | 264.3 | 0.1 | 211.3 | 0.5 | 2000 | [ |
| 148.5 | 5 | |||||
| B@N@NHCS-900 | 274.8 | 0.1 | 281.8 | 0.1 | 100 | This work |
| 179.4 | 2 | 122.5 | 2 | 500 | ||
表S2 B@N@NHCS-900负极材料与近期文献报道的钠离子电池负极材料的电化学性能对比
Table S2 Comparison of electrochemical performance of B@N@NHCS-900 anode with other carbon anode materials for sodium ion storage reported in previous literatures
| Sample | Rate performance | Cycling performance | Ref. | |||
|---|---|---|---|---|---|---|
| Capacity/(mAh·g-1) | Current density/(A·g-1) | Capacity/(mAh·g-1) | Current density /(A·g-1) | Cycle number | ||
| BHCS-1200 | 234 | 0.03 | 180 | 0.1 | 100 | [ |
| Below 100 | 2 | |||||
| NPUCS | 257.7 | 0.1 | 255.1 | 0.1 | 200 | [ |
| 157.0 | 5 | 110.0 | 5 | 2000 | ||
| PB-1000 | 205 | 0.1 | 146.9 | 2 | 600 | [ |
| 136.6 | 2 | |||||
| CPB-PL | 293 | 0.02 | 193 | 0.1 | 200 | [ |
| 77 | 1 | |||||
| SC-800 | 229 | 0.05 | 189 | 0.05 | 50 | [ |
| 82.0 | 1 | 81.0 | 0.5 | 2000 | ||
| SAL | 285 | 0.02 | 141.6 | 0.2 | 1000 | [ |
| 150 | 0.5 | |||||
| HC-1000 | 242.1 | 0.025 | Below 60 | 1 | 800 | [ |
| Below 100 | 0.8 | |||||
| YT-1200 | 261 | 0.1 | 235 | 0.1 | 200 | [ |
| 206 | 2 | |||||
| N-FLG | 264.3 | 0.1 | 211.3 | 0.5 | 2000 | [ |
| 148.5 | 5 | |||||
| B@N@NHCS-900 | 274.8 | 0.1 | 281.8 | 0.1 | 100 | This work |
| 179.4 | 2 | 122.5 | 2 | 500 | ||
| [1] |
MAHMUD S, RAHMAN M, KAMRUZZAMAN M, et al. Recent advances in lithium-ion battery materials for improved electrochemical performance: a review. Results in Engineering, 2022, 15: 100472.
DOI URL |
| [2] |
JUNG S K, HWANG I, CHANG D, et al. Nanoscale phenomena in lithium-ion batteries. Chemical Reviews, 2020, 120(14): 6684.
DOI URL |
| [3] |
SUN J, XU Y, LV Y, et al. Recent advances in covalent organic framework electrode materials for alkali metal-ion batteries. CCS Chemistry, 2023, 5(6): 1259.
DOI URL |
| [4] |
HUANG Y, ZHONG X, HU X, et al. Rationally designing closed pore structure by carbon dots to evoke sodium storage sites of hard carbon in low-potential region. Advanced Functional Materials, 2024, 34(4): 2308392.
DOI URL |
| [5] |
LI L, CHENG D, ZOU G, et al. Carbon anode from carbon dots-regulated polypyrrole for enhanced potassium storage. Journal of Alloys and Compounds, 2023, 958: 170481.
DOI URL |
| [6] |
SIMONE V, BOULINEAU A, DE GEYER A, et al. Hard carbon derived from cellulose as anode for sodium ion batteries: dependence of electrochemical properties on structure. Journal of Energy Chemistry, 2016, 25(5): 761.
DOI |
| [7] |
WANG Y, ZHANG M, SHEN X, et al. Biomass-derived carbon materials: controllable preparation and versatile applications. Small, 2021, 17(40): 2008079.
DOI URL |
| [8] |
CHU Y, ZHANG J, ZHANG Y, et al. Reconfiguring hard carbons with emerging sodium-ion batteries: a perspective. Advanced Materials, 2023, 35(31): 2212186.
DOI URL |
| [9] |
SU D, HUANG M, ZHANG J, et al. High N-doped hierarchical porous carbon networks with expanded interlayers for efficient sodium storage. Nano Research, 2020, 13(10): 2862.
DOI |
| [10] |
POTHAYA S, POOCHAI C, TAMMANOON N, et al. Bamboo-derived hard carbon/carbon nanotube composites as anode material for long-life sodium-ion batteries with high charge/ discharge capacities. Rare Metals, 2024, 43(1): 124.
DOI URL |
| [11] |
CHEN B, MENG Q, WANG T, et al. Cross-linking matters: building hard carbons with enhanced sodium-ion storage plateau capacities. Journal of Power Sources, 2024, 624: 235566.
DOI URL |
| [12] |
ZENG Y, WANG F, CHENG Y, et al. Identifying the importance of functionalization evolution during pre-oxidation treatment in producing economical asphalt-derived hard carbon for Na-ion batteries. Energy Storage Materials, 2024, 73: 103808.
DOI URL |
| [13] |
XIE L, SHEN G, LI B, et al. Porous carbon materials derived from waste tea leaves as high-capacity anodes for lithium/sodium ion batteries. Journal of Energy Storage, 2024, 100: 113598.
DOI URL |
| [14] |
LU B, SONG J X, DENG D R, et al. Self-doped porous sorghum husk-derived carbon as anode for high performance sodium-ion batteries at low temperatures. Journal of Energy Storage, 2024, 102: 114056.
DOI URL |
| [15] |
KIM D Y, LI O L, KANG J. Novel synthesis of highly phosphorus-doped carbon as an ultrahigh-rate anode for sodium ion batteries. Carbon, 2020, 168: 448.
DOI URL |
| [16] |
HE B, FENG L, HONG G, et al. A generic F-doped strategy for biomass hard carbon to achieve fast and stable kinetics in sodium/ potassium-ion batteries. Chemical Engineering Journal, 2024, 490: 151636.
DOI URL |
| [17] |
QUAN B, JIN A, YU S H, et al. Solvothermal-derived S-doped graphene as an anode material for sodium-ion batteries. Advanced Science, 2018, 5(5): 1700880.
DOI URL |
| [18] |
ZHANG T, ZHANG T, WANG F, et al. High-efficiently doping nitrogen in kapok fiber-derived hard carbon used as anode materials for boosting rate performance of sodium-ion batteries. Journal of Energy Chemistry, 2024, 96: 472.
DOI URL |
| [19] |
LIU Z, JIANG L, SHENG L, et al. Oxygen clusters distributed in graphene with “paddy land” structure: ultrahigh capacitance and rate performance for supercapacitors. Advanced Functional Materials, 2018, 28(5): 1705258.
DOI URL |
| [20] |
WANG Y, LI H, ZHAI B, et al. Highly crystalline poly(heptazine imide)-based carbonaceous anodes for ultralong lifespan and low- temperature sodium-ion batteries. ACS Nano, 2024, 18(4): 3456.
DOI URL |
| [21] |
FENG W, FENG N, LIU W, et al. Liquid-state templates for constructing B, N, Co-doping porous carbons with a boosting of potassium-ion storage performance. Advanced Energy Materials, 2021, 11(4): 2003215.
DOI URL |
| [22] |
CUI K, WANG C, LUO Y, et al. Enhanced sodium storage kinetics of nitrogen rich cellulose-derived hierarchical porous carbon via subsequent boron doping. Applied Surface Science, 2020, 531: 147302.
DOI URL |
| [23] |
OH J A S, DEYSHER G, RIDLEY P, et al. High-performing all-solid-state sodium-ion batteries enabled by the presodiation of hard carbon. Advanced Energy Materials, 2023, 13(26): 2300776.
DOI URL |
| [24] |
WARREN B E. X-ray diffraction in random layer lattices. Physical Review, 1941, 59(9): 693.
DOI URL |
| [25] |
FENG S, LI K, HU P, et al. Solvent-free synthesis of hollow carbon nanostructures for efficient sodium storage. ACS Nano, 2023, 17(22): 23152.
DOI PMID |
| [26] | QIU C, LI M, QIU D, et al. Ultra-high sulfur-doped hierarchical porous hollow carbon sphere anodes enabling unprecedented durable potassium-ion hybrid capacitors. ACS Applied Materials & Interfaces, 2021, 13(42): 49942. |
| [27] |
ZHU C L, WANG H L, FAN W J, et al. Large-scale doping- engineering enables boron/nitrogen dual-doped porous carbon for high-performance zinc ion capacitors. Rare Metals, 2022, 41(7): 2505.
DOI URL |
| [28] |
XIANG J, MA L, SUN Y, et al. Ball-milling-assisted N/O codoping for enhanced sodium storage performance of coconut- shell-derived hard carbon anodes in sodium-ion batteries. Langmuir, 2024, 40(45): 23853.
DOI URL |
| [29] |
LIU Y, YIN J, WU R, et al. Molecular engineering of pore structure/interfacial functional groups toward hard carbon anode in sodium-ion batteries. Energy Storage Materials, 2025, 75: 104008.
DOI URL |
| [30] |
ZHANG F, XIONG P, GUO X, et al. A nitrogen, sulphur dual-doped hierarchical porous carbon with interconnected conductive polyaniline coating for high-performance sodium- selenium batteries. Energy Storage Materials, 2019, 19: 251.
DOI URL |
| [31] |
XIA Q, YANG H, WANG M, et al. High energy and high power lithium-ion capacitors based on boron and nitrogen dual-doped 3d carbon nanofibers as both cathode and anode. Advanced Energy Materials, 2017, 7(22): 1701336.
DOI URL |
| [32] |
BALAJI S S, KARNAN M, ANANDHAGANESH P, et al. Performance evaluation of B-doped graphene prepared via two different methods in symmetric supercapacitor using various electrolytes. Applied Surface Science, 2019, 491: 560.
DOI URL |
| [33] | WU D, SUN F, QU Z, et al. Multi-scale structure optimization of boron-doped hard carbon nanospheres boosting the plateau capacity for high performance sodium ion batteries. Journal of Materials Chemistry A, 2022, 10(33): 1722. |
| [34] |
YUAN M, CAO B, LIU H, et al. Sodium storage mechanism of nongraphitic carbons: a general model and the function of accessible closed pores. Chemistry of Materials, 2022, 34(7): 3489.
DOI URL |
| [35] |
ALVIN S, CAHYADI H S, HWANG J, et al. Revealing the intercalation mechanisms of lithium, sodium, and potassium in hard carbon. Advanced Energy Materials, 2020, 10(20): 2000283.
DOI URL |
| [36] |
CHAO D, LIANG P, CHEN Z, et al. Pseudocapacitive Na-ion storage boosts high rate and areal capacity of self-branched 2D layered metal chalcogenide nanoarrays. ACS Nano, 2016, 10(11): 10211.
PMID |
| [37] |
HONG Z, ZHEN Y, RUAN Y, et al. Rational design and general synthesis of S-doped hard carbon with tunable doping sites toward excellent Na-ion storage performance. Advanced Materials, 2018, 30(29): 1802035.
DOI URL |
| [38] |
WU S, PENG H, XU J, et al. Nitrogen/phosphorus co-doped ultramicropores hard carbon spheres for rapid sodium storage. Carbon, 2024, 218: 118756.
DOI URL |
| [1] | 闫共芹, 王晨, 蓝春波, 洪雨昕, 叶维超, 付向辉. Al掺杂P2型Na0.8Ni0.33Mn0.67-xAlxO2钠离子电池正极材料的制备与电化学性能[J]. 无机材料学报, 2025, 40(9): 1005-1012. |
| [2] | 万俊池, 杜路路, 张永上, 李琳, 刘建德, 张林森. Na4FexP4O12+x/C钠离子电池正极材料的结构演变及其电化学性能[J]. 无机材料学报, 2025, 40(5): 497-503. |
| [3] | 张继国, 吴田, 赵旭, 杨钒, 夏天, 孙士恩. 钠离子电池正极材料循环稳定性提升策略及产业化进程[J]. 无机材料学报, 2025, 40(4): 348-362. |
| [4] | 杨舒琪, 杨存国, 牛慧祝, 石唯一, 舒珂维. GeP3/科琴黑复合材料作为钠离子电池高性能负极材料[J]. 无机材料学报, 2025, 40(3): 329-336. |
| [5] | 朱志杰, 申明远, 吴涛, 李文翠. Cu和Mg协同取代抑制钠离子电池正极材料P2-Na2/3Ni1/3Mn2/3O2的P2-O2相变[J]. 无机材料学报, 2025, 40(2): 184-195. |
| [6] | 王琨鹏, 刘兆林, 林存生, 王治宇. 基于低含水量普鲁士蓝正极的准固态钠离子电池[J]. 无机材料学报, 2024, 39(9): 1005-1012. |
| [7] | 孔剑锋, 黄杰成, 刘兆林, 林存生, 王治宇. 基于DPEPA聚合物凝胶电解质的准固态钠离子电池[J]. 无机材料学报, 2024, 39(12): 1331-1338. |
| [8] | 周靖渝, 李兴宇, 赵晓琳, 王有伟, 宋二红, 刘建军. Ti和Cu掺杂β-NaMnO2正极材料:钠离子电池的倍率和循环性能[J]. 无机材料学报, 2024, 39(12): 1404-1412. |
| [9] | 胡梦菲, 黄丽萍, 李贺, 张国军, 吴厚政. 锂/钠离子电池硬碳负极材料的研究进展[J]. 无机材料学报, 2024, 39(1): 32-44. |
| [10] | 孔国强, 冷明哲, 周战荣, 夏池, 沈晓芳. Sb掺杂O3型Na0.9Ni0.5Mn0.3Ti0.2O2钠离子电池正极材料[J]. 无机材料学报, 2023, 38(6): 656-662. |
| [11] | 王禹桐, 张非凡, 许乃才, 王春霞, 崔立山, 黄国勇. 水系锂离子电池负极材料LiTi2(PO4)3的研究进展[J]. 无机材料学报, 2022, 37(5): 481-492. |
| [12] | 赵伟, 徐阳, 万颖杰, 蔡天逊, 穆金潇, 黄富强. 金属氰胺化合物的结构、合成及电化学储能应用[J]. 无机材料学报, 2022, 37(2): 140-151. |
| [13] | 王晶, 徐守冬, 卢中华, 赵壮壮, 陈良, 张鼎, 郭春丽. 钠离子电池中空结构CoSe2/C负极材料的制备及储钠性能研究[J]. 无机材料学报, 2022, 37(12): 1344-1350. |
| [14] | 李昆儒, 胡省辉, 张正富, 郭玉忠, 黄瑞安. 源于溪木贼的高性能锂离子电池三维多孔生物质硅/碳复合负极材料[J]. 无机材料学报, 2021, 36(9): 929-935. |
| [15] | 张晓君, 李佳乐, 邱吴劼, 杨淼森, 刘建军. 钠离子电池正极材料P2-Nax[Mg0.33Mn0.67]O2的电化学活性研究[J]. 无机材料学报, 2021, 36(6): 623-628. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||