
Journal of Inorganic Materials ›› 2014, Vol. 29 ›› Issue (7): 763-768.DOI: 10.3724/SP.J.1077.2014.13519
• Orginal Article • Previous Articles Next Articles
DUAN Lian-Feng1,2, ZHANG Yu2, WANG Li-Min1, JIN Song-Zhe2, WU Hua2
Received:2013-10-10
Revised:2013-12-06
Published:2014-07-20
Online:2014-06-20
CLC Number:
DUAN Lian-Feng, ZHANG Yu, WANG Li-Min, JIN Song-Zhe, WU Hua. Synthesis and Characterization of MnFe2O4 with Different Morphologies and Their Application in Water Treatment[J]. Journal of Inorganic Materials, 2014, 29(7): 763-768.
Add to citation manager EndNote|Ris|BibTeX
| Samples | S2 | S3 | S4 |
|---|---|---|---|
| Ms /(Am2·kg-1) | 58.7 | 76.7 | 61.3 |
| Mr /(Am2·kg-1) | 6.5 | 1.3 | 7.4 |
| Hc /(kA·m-1) | 6.23 | 0.95 | 6.87 |
Table 1 Magnetism parameters of different samples
| Samples | S2 | S3 | S4 |
|---|---|---|---|
| Ms /(Am2·kg-1) | 58.7 | 76.7 | 61.3 |
| Mr /(Am2·kg-1) | 6.5 | 1.3 | 7.4 |
| Hc /(kA·m-1) | 6.23 | 0.95 | 6.87 |
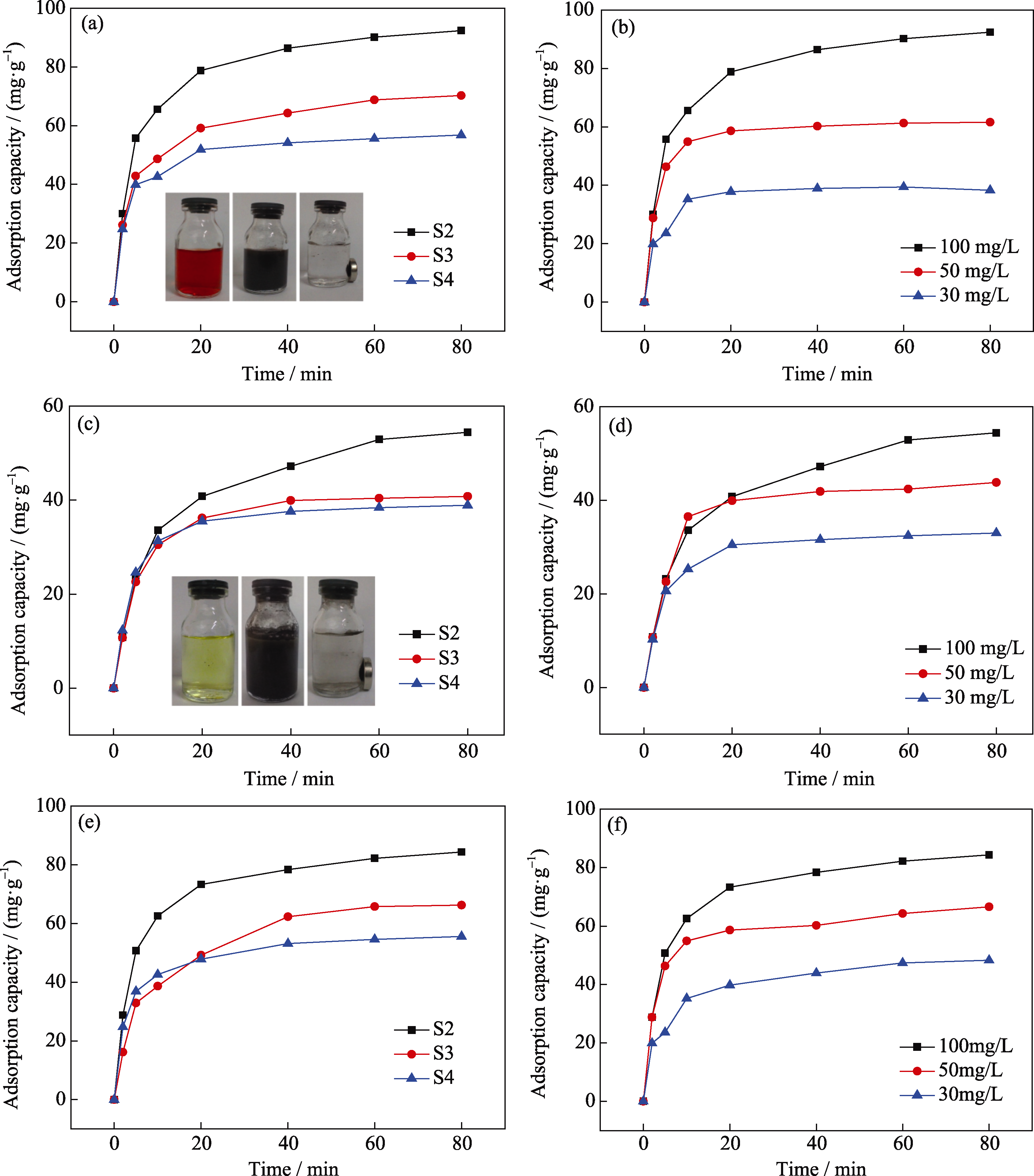
Fig. 5 Adsorption capacity of samples S2, S3 and S4 adsorbed CR, Cr6+ and Pb2+ solution (a, c and e) and adsorption capacity of sample S2 adsorbed CR, Cr6+ and Pb2+solution (b, d and f) with different concentrations
| [1] | CUI Y, LIEBER C M. Functional nanoscale electronic devices assembled using silicon nanowire building blocks. Science, 2001, 291(851): 851-853. |
| [2] | WANG X, ZHUANG J, PENG Q, et al. A general strategy for nanocrystal synthesis. Nature, 2005, 437(121-124): 121-124. |
| [3] | DUAN L F, JIA S S, ZHAO L J. Study on morphologies of Co microcrystals produced by solvothermal method with different solvents. Materials Research Bulletin, 2010, 45(4): 373-376. |
| [4] | LIU Q,LV L,WANG Y. Electric and magnetic properties of NiFe2O4- BaTiO3 composites synthesized by hydrothermalmethod. Bulletin of the Chinese Ceramic Society, 2012, 33(1): 50-55. |
| [5] | FU F L, WANG Q. Removal of heavy metal ions from wastewaters: a review. J. Environ. Manage., 2011, 92(3): 407-418. |
| [6] | WANG Y H, LIN S H, JUAN R S. Removal of heavy metal ions from aqueous solutions using various low-cost adsorbents. J. Hazard. Mater., 2003, 102(2/3): 291-302. |
| [7] | KURNIAWAN T A, CHAN G Y S, LO W H, et al. Physico- chemical treatment techniques for wastewater laden with heavy metals. Chem. Eng. J. , 2006, 118(1/2): 83-98. |
| [8] | LI M, QIAN F Y, LI X Y. Characterization of organic pollutants in bio-treated effluents of dyeing and textile wastewater and removal behavior during magnesium sulfate coagulation process. Environmental Chemistry, 2012, 31(1): 88-93. |
| [9] | MERIC S, KAPTAN D, OLMEZ T. Color and COD removal from wast-e-water containing Reactive Black 5 using Fenton's oxidation process. Chemosphere, 2004, 54(3): 435-441. |
| [10] | PAN B J, PAN B C, ZHANG W M, et al. Development of polymeric and polymer-based hybrid adsorbents for pollutants removal from waters. Chem. Eng. J., 2009, 151(1/2/3): 19-29. |
| [11] | MISHRA S P, SINGH V K, TIWARI D. Radiotracer technique in adsorption study. 14. Efficient removal of mercury from aqueous solutions by hydrous zirco-nium oxide. Appl. Radiat. Isot. , 1996 47(1): 15-21. |
| [12] | VANBENSCHOTEN J E, REED B E, MATSUMOTO M R, et al. Metal removal by soil washing for an iron-oxide coated sandy soil. Water Environ. Res. , 1994(66): 168-174. |
| [13] | DING Z H, FENG J M. Surface characters of iron-(hydro) oxides andtheir adsorption of heavy metal ions. Acta Mineralogica Sinica, 2000(20): 349-352. |
| [14] | AGRAWAL A, SAHU K K. Kinetic and isotherm studies of cadmium adsorption on manganese nodule residue. J. Hazard. Mater., 2006(137): 915-924. |
| [15] | MANDAVIAN A R, MIRRAHIMI M A S. Efficient separation of heavy metal cations by anchoring polyacrylic acid on superparamagnetic magnetite nanoparticles through surface modification. Chem. Eng. J. , 2010, 159(1/2/3): 264-271. |
| [16] | ZHAO X, LV L, PAN B, et al. Polymer-supported nanoco-m-posites for environmental application-a review. Chem. Eng. J., 2011, 170(2/3): 381-394. |
| [17] | ZHAO M Y, DUAN L F, WU H, et al. Synthesis of hollow Fe3O4 submicrospheres and their adsorption-desorption capability for Congo red. Chin. J. Inorg. Chem., 2012, 28(10): 2186-2192. |
| [18] | WANG Y Q, CHENG R M, WEN Z, et al. Synthesis and characteriz-ation of single-crystalline MnFe2O4 ferrite nanocrystals and their possible application in water treatment. Eur. J.Inorg.Chem., 2011, 19(1): 2942-2947. |
| [19] | ZHUO NA, LI LI, GAO YU, et al. CTAB-assisted synthesis of nano-composite Ag/ZnO-TiO2 and its UV photocatalytic activity. Chin. J. Inorg. Chem., 2013, 29(5): 991-998. |
| [20] | ZHU Y F, ZHAO W R, CHEN H R, et al. A Simple one-pot self-assem-bly route to nanoporous and monodispersed Fe3O4 particles with oriented attachment structure and magnetic property. Phys. J. Chem. C, 2007, 111(14): 5281-5285. |
| [21] | DIMITROV D A, WYSIN G M. Effects of surface anisotropy on hysteresis in fine magnetic particles. Phys. Rev. B, 1994, 50(5): 3077-3084. |
| [22] | KODOMA R H, BERKOWITZ A E. Atomic-scale magnetic modeling of oxide nanoparticles. Phys. Rev. B, 1999, 59(9): 6321-6336. |
| [23] | WANG L X, LI J C, JIANG Q, et al. Water-soluble Fe3O4 nanopart-icles with high solubility for removal of heavy-metal ions from waste water. Dalton Trans., 2012, 41(15): 4544-4551. |
| [24] | GUO MING, HUANG FENG-QIN, LIU MI-MI, et al. Performance of binding interaction between Cr(Ⅵ) and serum proteome. Chin. J. Inorg.Chem., 2013, 29(5): 1037-1044. |
| [25] | WANG X S, CHEN J P. Biosorption of congo red from aqueous solution using wheat bran and rice bran: batch studies. Sep.Sci. Technol. , 2009, 44(6): 1452-1466. |
| [26] | PANDAG C, DAS S K, GUHA A K. Effect of method of crystallization on the IV-III and IV-II polymorphic transitions of ammonium nitrate. J. Hazard. Mater., 2009, 161(1): 373-379. |
| [27] | JIANG M Q, WANG Q P, JIN X Y, et al. Removal of Pb(II) from aqueous solution using modified and unmodified kaolinite clay. J. Hazard. Mater., 2009, 170(1): 332-339. |
| [28] | HU J, LO I M C, CHEN G H. Fast removal and recovery of Cr(VI) using surface-modified jacobsite (MnFe2O4) nanoparticles. Langmuir, 2005, 21(24): 11173-11179. |
| [1] | MU Haojie, ZHANG Yuanjiang, YU Bin, FU Xiumei, ZHOU Shibin, LI Xiaodong. Preparation and Properties of ZrO2 Doped Y2O3-MgO Nanocomposite Ceramics [J]. Journal of Inorganic Materials, 2025, 40(3): 281-289. |
| [2] | FAN Wugang, CAO Xiong, ZHOU Xiang, LI Ling, ZHAO Guannan, ZHANG Zhaoquan. Anticorrosion Performance of 8YSZ Ceramics in Simulated Aqueous Environment of Pressurized Water Reactor [J]. Journal of Inorganic Materials, 2024, 39(7): 803-809. |
| [3] | CHEN Qian, SU Haijun, JIANG Hao, SHEN Zhonglin, YU Minghui, ZHANG Zhuo. Progress of Ultra-high Temperature Oxide Ceramics: Laser Additive Manufacturing and Microstructure Evolution [J]. Journal of Inorganic Materials, 2024, 39(7): 741-753. |
| [4] | JIANG Lingyi, PANG Shengyang, YANG Chao, ZHANG Yue, HU Chenglong, TANG Sufang. Preparation and Oxidation Behaviors of C/SiC-BN Composites [J]. Journal of Inorganic Materials, 2024, 39(7): 779-786. |
| [5] | ZHENG Yawen, ZHANG Cuiping, ZHANG Ruijie, XIA Qian, RU Hongqiang. Fabrication of Boron Carbide Ceramic Composites by Boronic Acid Carbothermal Reduction and Silicon Infiltration Reaction Sintering [J]. Journal of Inorganic Materials, 2024, 39(6): 707-714. |
| [6] | XUE Yifan, LI Weijie, ZHANG Zhongwei, PANG Xu, LIU Yu. Process Control of PyC Interphases Microstructure and Uniformity in Carbon Fiber Cloth [J]. Journal of Inorganic Materials, 2024, 39(4): 399-408. |
| [7] | SUN Chuan, HE Pengfei, HU Zhenfeng, WANG Rong, XING Yue, ZHANG Zhibin, LI Jinglong, WAN Chunlei, LIANG Xiubing. SiC-based Ceramic Materials Incorporating GNPs Array: Preparation and Mechanical Characterization [J]. Journal of Inorganic Materials, 2024, 39(3): 267-273. |
| [8] | LI Xianke, ZHANG Chaoyi, HUANG Lin, SUN Peng, LIU Bo, XU Jun, TANG Huili. High-quality Indium-doped Gallium Oxide Single Crystal Growth by Floating Zone Method [J]. Journal of Inorganic Materials, 2024, 39(12): 1384-1390. |
| [9] | CAI Hao, WANG Qihang, ZOU Zhaoyong. Crystallization Pathway of Monohydrocalcite via Amorphous Calcium Carbonate Regulated by Magnesium Ion [J]. Journal of Inorganic Materials, 2024, 39(11): 1275-1282. |
| [10] | HAO Yongxin, QIN Juan, SUN Jun, YANG Jinfeng, LI Qinglian, HUANG Guijun, XU Jingjun. Impact of Crucible Bottom Shape on the Growth of Congruent Lithium Niobate Crystals by Czochralski Method [J]. Journal of Inorganic Materials, 2024, 39(10): 1167-1174. |
| [11] | ZHENG Jiaqian, LU Xiao, LU Yajie, WANG Yingjun, WANG Zhen, LU Jianxi. Functional Bioadaptability in Medical Bioceramics: Biological Mechanism and Application [J]. Journal of Inorganic Materials, 2024, 39(1): 1-16. |
| [12] | QIN Juan, LIANG Dandan, SUN Jun, YANG Jinfeng, HAO Yongxin, LI Qinglian, ZHANG Ling, XU Jingjun. Flat Shoulder Congruent Lithium Niobate Crystals Grown by the Czochralski Method [J]. Journal of Inorganic Materials, 2023, 38(8): 978-986. |
| [13] | HE Danqi, WEI Mingxu, LIU Ruizhi, TANG Zhixin, ZHAI Pengcheng, ZHAO Wenyu. Heavy-Fermion YbAl3 Materials: One-step Synthesis and Enhanced Thermoelectric Performance [J]. Journal of Inorganic Materials, 2023, 38(5): 577-582. |
| [14] | WU Shuang, GOU Yanzi, WANG Yongshou, SONG Quzhi, ZHANG Qingyu, WANG Yingde. Effect of Heat Treatment on Composition, Microstructure and Mechanical Property of Domestic KD-SA SiC Fibers [J]. Journal of Inorganic Materials, 2023, 38(5): 569-576. |
| [15] | LIN Siqi, LI Airan, FU Chenguang, LI Rongbing, JIN Min. Crystal Growth and Thermoelectric Properties of Zintl Phase Mg3X2 (X=Sb, Bi) Based Materials: a Review [J]. Journal of Inorganic Materials, 2023, 38(3): 270-279. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||