
Journal of Inorganic Materials ›› 2026, Vol. 41 ›› Issue (4): 445-454.DOI: 10.15541/jim20250272
• RESEARCH ARTICLE • Previous Articles Next Articles
WANG Yuhe1,2( ), LUO Yixiu1(
), LUO Yixiu1( ), GUO Huiming3, ZHANG Guangheng4, ZHANG Siyan4, SUN Luchao1, WANG Jiemin1, WANG Jingyang1(
), GUO Huiming3, ZHANG Guangheng4, ZHANG Siyan4, SUN Luchao1, WANG Jiemin1, WANG Jingyang1( )
)
Received:2025-06-27
Revised:2025-08-22
Published:2026-04-20
Online:2025-08-26
Contact:
LUO Yixiu, professor. E-mail: yxluo13s@imr.ac.cn;About author:WANG Yuhe (2001-), male, Master candidate. E-mail: yhwang23s@imr.ac.cn
Supported by:CLC Number:
WANG Yuhe, LUO Yixiu, GUO Huiming, ZHANG Guangheng, ZHANG Siyan, SUN Luchao, WANG Jiemin, WANG Jingyang. First-principles Investigation of Elastic and Thermophysical Properties of High-entropy Rare-earth Oxide Thermal Barrier Coating Materials[J]. Journal of Inorganic Materials, 2026, 41(4): 445-454.
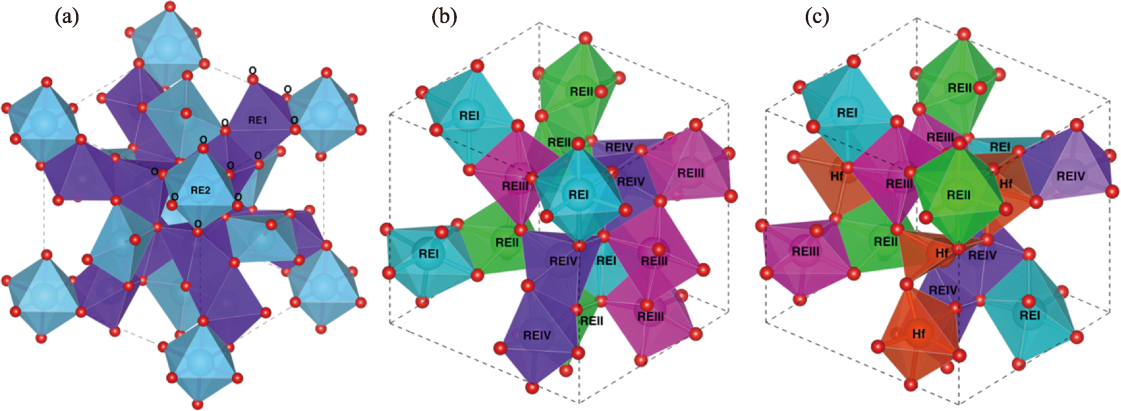
Fig. 1 Crystal structures of high-entropy rare-earth oxide materials (a) C-RE2O3; (b) C-(REI1/4REII1/4REIII1/4REIV1/4)2O3; (c) C-(REI3/16REII3/16REIII3/16REIV3/16Hf1/4)2O3
| Compound | Method | a/Å | b/Å | c/Å | α/(°) | β/(°) | γ/(°) | ρ/(g•cm-3) |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | Calc | 9.13 | 9.14 | 9.15 | 109.49 | 109.52 | 109.46 | 7.81 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | Calc | 9.05 | 9.04 | 9.04 | 109.43 | 109.51 | 109.46 | 9.10 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | Calc | 9.01 | 9.06 | 9.01 | 109.34 | 109.37 | 109.55 | 8.43 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | Calc | 8.92 | 9.00 | 8.98 | 109.53 | 109.74 | 109.47 | 9.46 |
Table 1 Calculated lattice parameters of four high-entropy rare-earth oxide materials
| Compound | Method | a/Å | b/Å | c/Å | α/(°) | β/(°) | γ/(°) | ρ/(g•cm-3) |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | Calc | 9.13 | 9.14 | 9.15 | 109.49 | 109.52 | 109.46 | 7.81 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | Calc | 9.05 | 9.04 | 9.04 | 109.43 | 109.51 | 109.46 | 9.10 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | Calc | 9.01 | 9.06 | 9.01 | 109.34 | 109.37 | 109.55 | 8.43 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | Calc | 8.92 | 9.00 | 8.98 | 109.53 | 109.74 | 109.47 | 9.46 |
| Compound | c11 | c12 | c13 | c22 | c23 | c33 | c44 | c55 | c66 |
|---|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 244.79 | 95.97 | 95.90 | 240.43 | 101.79 | 239.03 | 69.14 | 62.94 | 63.10 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 257.22 | 100.81 | 99.30 | 251.14 | 107.11 | 250.74 | 72.87 | 66.14 | 66.16 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 250.73 | 98.29 | 98.37 | 248.15 | 97.99 | 241.45 | 70.74 | 69.71 | 69.93 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 259.08 | 102.63 | 100.97 | 250.68 | 97.74 | 251.63 | 77.04 | 71.63 | 75.61 |
Table 2 Calculated second-order elastic coefficients of four high-entropy rare-earth oxide materials (GPa)
| Compound | c11 | c12 | c13 | c22 | c23 | c33 | c44 | c55 | c66 |
|---|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 244.79 | 95.97 | 95.90 | 240.43 | 101.79 | 239.03 | 69.14 | 62.94 | 63.10 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 257.22 | 100.81 | 99.30 | 251.14 | 107.11 | 250.74 | 72.87 | 66.14 | 66.16 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 250.73 | 98.29 | 98.37 | 248.15 | 97.99 | 241.45 | 70.74 | 69.71 | 69.93 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 259.08 | 102.63 | 100.97 | 250.68 | 97.74 | 251.63 | 77.04 | 71.63 | 75.61 |
| Compound | B/GPa | G/GPa | E/GPa | Emax/GPa | Emin/GPa | Emax/Emin | G/B | ν/GPa |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 146 | 67 | 175 | 192 | 151 | 1.2753 | 0.4606 | 0.3004 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 153 | 71 | 183 | 201 | 159 | 1.2639 | 0.4624 | 0.2997 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 148 | 72 | 185 | 197 | 174 | 1.1323 | 0.4852 | 0.2912 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 151 | 75 | 194 | 209 | 180 | 1.1605 | 0.4975 | 0.2866 |
Table 3 Calculated elastic modulus of four high-entropy rare-earth oxide materials
| Compound | B/GPa | G/GPa | E/GPa | Emax/GPa | Emin/GPa | Emax/Emin | G/B | ν/GPa |
|---|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 146 | 67 | 175 | 192 | 151 | 1.2753 | 0.4606 | 0.3004 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 153 | 71 | 183 | 201 | 159 | 1.2639 | 0.4624 | 0.2997 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 148 | 72 | 185 | 197 | 174 | 1.1323 | 0.4852 | 0.2912 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 151 | 75 | 194 | 209 | 180 | 1.1605 | 0.4975 | 0.2866 |
| Compound | vs/(m·s-1) | vl/(m·s-1) | vm/(m·s-1) | ΘD/K | γ | αL/(×10-6, K-1) | kmin/(W·m-1·K-1) |
|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 2932 | 5489 | 3276 | 398 | 1.7749 | 11.4574 | 0.7567 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 2785 | 5207 | 3111 | 382 | 1.7707 | 11.2533 | 0.7334 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 2915 | 5371 | 3253 | 400 | 1.7194 | 11.3375 | 0.7690 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 2823 | 5162 | 3148 | 390 | 1.6928 | 11.1577 | 0.7566 |
Table 4 Thermophysical parameters of four high-entropy rare-earth oxide materials
| Compound | vs/(m·s-1) | vl/(m·s-1) | vm/(m·s-1) | ΘD/K | γ | αL/(×10-6, K-1) | kmin/(W·m-1·K-1) |
|---|---|---|---|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | 2932 | 5489 | 3276 | 398 | 1.7749 | 11.4574 | 0.7567 |
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | 2785 | 5207 | 3111 | 382 | 1.7707 | 11.2533 | 0.7334 |
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | 2915 | 5371 | 3253 | 400 | 1.7194 | 11.3375 | 0.7690 |
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | 2823 | 5162 | 3148 | 390 | 1.6928 | 11.1577 | 0.7566 |
| Parameter | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf | Diff(1-3)/% | Diff(2-4)/% | Diff(1-2)/% | Diff(3-4)/% |
|---|---|---|---|---|---|---|---|---|
| kT | 898.98 | 892.94 | 1006.36 | 1050.75 | 12.07 | 17.70 | -0.68 | 4.32 |
| 1.44×10-12 | 1.62×10-12 | 1.49×10-12 | 1.62×10-12 | 3.15 | -0.01 | 11.90 | 8.48 | |
| ΘD3 | 6.31×107 | 5.58×107 | 6.40×107 | 5.95×107 | 1.36 | 6.69 | -11.69 | -7.05 |
| A(γ)/γ2 | 9.85×106 | 9.90×106 | 1.06×107 | 1.09×107 | 7.19 | 10.33 | 0.52 | 3.47 |
Table 5 Quantitative relationship of k~αT-1 and origin of differences in four high-entropy rare-earth oxide materials
| Parameter | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf | Diff(1-3)/% | Diff(2-4)/% | Diff(1-2)/% | Diff(3-4)/% |
|---|---|---|---|---|---|---|---|---|
| kT | 898.98 | 892.94 | 1006.36 | 1050.75 | 12.07 | 17.70 | -0.68 | 4.32 |
| 1.44×10-12 | 1.62×10-12 | 1.49×10-12 | 1.62×10-12 | 3.15 | -0.01 | 11.90 | 8.48 | |
| ΘD3 | 6.31×107 | 5.58×107 | 6.40×107 | 5.95×107 | 1.36 | 6.69 | -11.69 | -7.05 |
| A(γ)/γ2 | 9.85×106 | 9.90×106 | 1.06×107 | 1.09×107 | 7.19 | 10.33 | 0.52 | 3.47 |
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XREⅠ=XREⅡ=XREⅢ= XREIV=1/4 |
Table S1 Number of sites on sublattice and composition for C-(REⅠ1/4REⅡ1/4REⅢ1/4REⅣ1/4)2O3
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XREⅠ=XREⅡ=XREⅢ= XREIV=1/4 |
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=0 XREⅡ=XREⅢ=XREⅣ= XHf =1/4 | XREⅠ=XHf=1/4 XREⅡ=XREⅢ=XREⅣ= 1/6 | |
| No.2 | NS | 4 | 12 |
| XS | XREⅡ=0 XREⅠ=XREⅢ=XREⅣ= XHf=1/4 | XREⅡ=XHf=1/4 XREⅠ=XREⅢ=XREⅣ= 1/6 | |
| No.3 | NS | 4 | 12 |
| XS | XREⅢ=0 XREⅠ=XREⅡ=XREⅣ= XHf=1/4 | XREⅢ=XHf=1/4 XREⅠ=XREⅡ=XREⅣ= 1/6 | |
| No.4 | NS | 4 | 12 |
| XS | XREⅣ=0 XREⅠ=XREⅡ=XREⅢ= XHf=1/4 | XREⅣ=XHf=1/4 XREⅠ=XREⅡ=XREⅢ= 1/6 | |
| No.5 | NS | 4 | 12 |
| XS | XHf=0 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XHf=1/3 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/6 |
Table S2 Number of sites on sublattice and composition for C-(REⅠ3/16REⅡ3/16REⅢ3/16REⅣ3/16Hf1/4)2O3
| Site | RE1 | RE2 | |
|---|---|---|---|
| No.1 | NS | 4 | 12 |
| XS | XREⅠ=0 XREⅡ=XREⅢ=XREⅣ= XHf =1/4 | XREⅠ=XHf=1/4 XREⅡ=XREⅢ=XREⅣ= 1/6 | |
| No.2 | NS | 4 | 12 |
| XS | XREⅡ=0 XREⅠ=XREⅢ=XREⅣ= XHf=1/4 | XREⅡ=XHf=1/4 XREⅠ=XREⅢ=XREⅣ= 1/6 | |
| No.3 | NS | 4 | 12 |
| XS | XREⅢ=0 XREⅠ=XREⅡ=XREⅣ= XHf=1/4 | XREⅢ=XHf=1/4 XREⅠ=XREⅡ=XREⅣ= 1/6 | |
| No.4 | NS | 4 | 12 |
| XS | XREⅣ=0 XREⅠ=XREⅡ=XREⅢ= XHf=1/4 | XREⅣ=XHf=1/4 XREⅠ=XREⅡ=XREⅢ= 1/6 | |
| No.5 | NS | 4 | 12 |
| XS | XHf=0 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/4 | XHf=1/3 XREⅠ=XREⅡ=XREⅢ= XREⅣ=1/6 |
| Compound | Unit | Average bond length/Å | |
|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | YO6 | 2.2868 | |
| HoO6 | 2.2794 | ||
| ErO6 | 2.2700 | ||
| YbO6 | 2.2518 | ||
| REO6 | 2.2720 | ||
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | ErO6 | 2.2631 | |
| TmO6 | 2.2535 | ||
| YbO6 | 2.2446 | ||
| LuO6 | 2.2356 | ||
| REO6 | 2.2492 | ||
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | YO6 | 2.2991 | |
| HoO6 | 2.2880 | ||
| ErO6 | 2.2794 | ||
| YbO6 | 2.2451 | ||
| REO6 | 2.2779 | ||
| HfO6 | 2.1893 | ||
| XO6* | 2.2558 | ||
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | ErO6 | 2.2684 | |
| TmO6 | 2.2541 | ||
| YbO6 | 2.2437 | ||
| LuO6 | 2.2399 | ||
| REO6 | 2.2515 | ||
| HfO6 | 2.1936 | ||
| XO6* | 2.2370 | ||
Table S3 Average bond length of polyhedral of four high-entropy rare-earth oxide materials
| Compound | Unit | Average bond length/Å | |
|---|---|---|---|
| (Y1/4Ho1/4Er1/4Yb1/4)2O3 | YO6 | 2.2868 | |
| HoO6 | 2.2794 | ||
| ErO6 | 2.2700 | ||
| YbO6 | 2.2518 | ||
| REO6 | 2.2720 | ||
| (Er1/4Tm1/4Yb1/4Lu1/4)2O3 | ErO6 | 2.2631 | |
| TmO6 | 2.2535 | ||
| YbO6 | 2.2446 | ||
| LuO6 | 2.2356 | ||
| REO6 | 2.2492 | ||
| (Y3/16Ho3/16Er3/16Yb3/16Hf1/4)2O3 | YO6 | 2.2991 | |
| HoO6 | 2.2880 | ||
| ErO6 | 2.2794 | ||
| YbO6 | 2.2451 | ||
| REO6 | 2.2779 | ||
| HfO6 | 2.1893 | ||
| XO6* | 2.2558 | ||
| (Er3/16Tm3/16Yb3/16Lu3/16Hf1/4)2O3 | ErO6 | 2.2684 | |
| TmO6 | 2.2541 | ||
| YbO6 | 2.2437 | ||
| LuO6 | 2.2399 | ||
| REO6 | 2.2515 | ||
| HfO6 | 2.1936 | ||
| XO6* | 2.2370 | ||
| Material | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf |
|---|---|---|---|---|
| s11 | 0.005239 | 0.004968 | 0.005147 | 0.005011 |
| s12 | -0.001469 | -0.001396 | -0.001445 | -0.001504 |
| s13 | -0.001491 | -0.001365 | -0.001516 | -0.001426 |
| s14 | -0.000049 | -0.000006 | -0.000043 | -0.000120 |
| s15 | -0.000082 | -0.000019 | 0.000079 | 0.000086 |
| s16 | 0.000013 | 0.000002 | 0.000050 | -0.000122 |
| s22 | 0.005544 | 0.005321 | 0.005213 | 0.005182 |
| s23 | -0.001794 | -0.001739 | -0.001541 | -0.001420 |
| s24 | -0.000058 | 0.000003 | 0.000073 | -0.000020 |
| s25 | 0.000077 | -0.000031 | -0.000081 | -0.000353 |
| s26 | 0.000974 | 0.000913 | 0.000381 | 0.000603 |
| s33 | 0.005584 | 0.005303 | 0.005388 | 0.005103 |
| s34 | 0.000051 | -0.000014 | 0.000040 | 0.000062 |
| s35 | 0.000000 | 0.000049 | 0.000058 | 0.000069 |
| s36 | -0.000990 | -0.000888 | -0.000400 | -0.000397 |
| s44 | 0.014726 | 0.013938 | 0.014169 | 0.012992 |
| s45 | -0.002028 | -0.001789 | -0.000457 | -0.000304 |
| s46 | 0.000017 | -0.000002 | -0.000460 | 0.000002 |
| s55 | 0.016174 | 0.015356 | 0.014366 | 0.014004 |
| s56 | -0.000060 | 0.000077 | -0.000237 | -0.000343 |
| s66 | 0.016120 | 0.015338 | 0.014369 | 0.013311 |
Table S4 Elastic compliance constants of four high-entropy rare-earth oxide materials
| Material | 1-YHoErYb | 2-ErTmYbLu | 3-YHoErYbHf | 4-ErTmYbLuHf |
|---|---|---|---|---|
| s11 | 0.005239 | 0.004968 | 0.005147 | 0.005011 |
| s12 | -0.001469 | -0.001396 | -0.001445 | -0.001504 |
| s13 | -0.001491 | -0.001365 | -0.001516 | -0.001426 |
| s14 | -0.000049 | -0.000006 | -0.000043 | -0.000120 |
| s15 | -0.000082 | -0.000019 | 0.000079 | 0.000086 |
| s16 | 0.000013 | 0.000002 | 0.000050 | -0.000122 |
| s22 | 0.005544 | 0.005321 | 0.005213 | 0.005182 |
| s23 | -0.001794 | -0.001739 | -0.001541 | -0.001420 |
| s24 | -0.000058 | 0.000003 | 0.000073 | -0.000020 |
| s25 | 0.000077 | -0.000031 | -0.000081 | -0.000353 |
| s26 | 0.000974 | 0.000913 | 0.000381 | 0.000603 |
| s33 | 0.005584 | 0.005303 | 0.005388 | 0.005103 |
| s34 | 0.000051 | -0.000014 | 0.000040 | 0.000062 |
| s35 | 0.000000 | 0.000049 | 0.000058 | 0.000069 |
| s36 | -0.000990 | -0.000888 | -0.000400 | -0.000397 |
| s44 | 0.014726 | 0.013938 | 0.014169 | 0.012992 |
| s45 | -0.002028 | -0.001789 | -0.000457 | -0.000304 |
| s46 | 0.000017 | -0.000002 | -0.000460 | 0.000002 |
| s55 | 0.016174 | 0.015356 | 0.014366 | 0.014004 |
| s56 | -0.000060 | 0.000077 | -0.000237 | -0.000343 |
| s66 | 0.016120 | 0.015338 | 0.014369 | 0.013311 |
| [1] |
LEE K N, FOX D S, BANSAL N P. Rare earth silicate environmental barrier coatings for SiC/SiC composites and Si3N4 ceramics. Journal of the European Ceramic Society, 2005, 25(10): 1705.
DOI URL |
| [2] |
MARTIN D, BENNETT C, HUSSAIN T. A review on environmental barrier coatings: history, current state of the art and future developments. Journal of the European Ceramic Society, 2021, 41(3): 1747.
DOI URL |
| [3] |
DUAN Z, DENG L, LU K, et al. Thermal cycle and water oxygen performance of multi-layered Y3Al5O12/Yb2SiO5/Yb2Si2O7 thermal/ environmental barrier coatings. Ceramics International, 2024, 50(9): 16309.
DOI URL |
| [4] |
ZINKEVICH M. Thermodynamics of rare earth sesquioxides. Progress in Materials Science, 2007, 52(4): 597.
DOI URL |
| [5] |
ZHANG G H, ZHANG J, WANG J Y. Synthesis and characterization of ytterbium oxide: a novel CMAS-resistant environmental barrier coating material. Journal of the American Ceramic Society, 2023, 106(1): 621.
DOI URL |
| [6] |
ZHANG G H, SHI J Y, ZHANG J, et al. Investigation on crystallization behavior between (ScxYb1-x)O1.5 and CMAS: a new insight in the effect of Sc substitution. Journal of Advanced Ceramics, 2024, 13(6): 789.
DOI URL |
| [7] |
WRIGHT A J, WANG Q Y, HUANG C Y, et al. From high-entropy ceramics to compositionally-complex ceramics: a case study of fluorite oxides. Journal of the European Ceramic Society, 2020, 40(5): 2120.
DOI URL |
| [8] |
SUN L C, REN X M, DU T F, et al. High entropy engineering: new strategy for the critical property optimizations of rare earth silicates. Journal of Inorganic Materials, 2021, 36(4): 339.
DOI |
| [9] |
WANG Y D, GUO Q, ZHOU Q J, et al. Research progress in high temperature functional coatings for advanced aeroengines. Journal of Aeronautical Materials, 2024, 44(5): 48.
DOI |
| [10] |
PAN W, LIU G H, WANG X L, et al. Review of the development of thermal barrier coating materials and technologies. Thermal Spray Technology, 2025, 17(1): 1.
DOI URL |
| [11] | LUO X W, XU C H, DUAN S S, et al. Research progress of high-entropy thermal barrier coatings ceramic materials. Aeronautical Manufacturing Technology, 2022, 65(3): 82. |
| [12] |
SUN Y N, XIANG H M, DAI F Z, et al. Preparation and properties of CMAS resistant bixbyite structured high-entropy oxides RE2O3 (RE=Sm, Eu, Er, Lu, Y, and Yb): promising environmental barrier coating materials for Al2O3f/Al2O3 composites. Journal of Advanced Ceramics, 2021, 10: 596.
DOI |
| [13] |
ARDREY K D, RIDLEY M J, WANG K, et al. Opportunities for novel refractory alloy thermal/environmental barrier coatings using multicomponent rare earth oxides. Scripta Materialia, 2024, 251: 116206.
DOI URL |
| [14] |
PING X Y, MENG B, YU X H, et al. Structural, mechanical and thermal properties of cubic bixbyite-structured high-entropy oxides. Chemical Engineering Journal, 2023, 464: 142649.
DOI URL |
| [15] |
MENG H, YU R, TANG Z, et al. Formation ability descriptors for high-entropy diborides established through high-throughput experiments and machine learning. Acta Materialia, 2023, 256: 119132.
DOI URL |
| [16] |
GYORFFY B L. Coherent-potential approximation for a nonoverlap muffin-tin-potential model of random substitutional alloys. Physical Review B, 1972, 5(6): 2382.
DOI URL |
| [17] |
BELLAICHE L, VANDERBILT D. Virtual crystal approximation revisited: application to dielectric and piezoelectric properties of perovskites. Physical Review B, 2000, 61(12): 7877.
DOI URL |
| [18] |
VAN DE WALLE A, TIWARY P, DE JONG M, et al. Efficient stochastic generation of special quasirandom structures. Calphad, 2013, 42: 13.
DOI URL |
| [19] |
REN X M, TIAN Z L, ZHANG J, et al. Equiatomic quaternary (Y1/4Ho1/4Er1/4Yb1/4)2SiO5 silicate: a perspective multifunctional thermal and environmental barrier coating material. Scripta Materialia, 2019, 168: 47.
DOI URL |
| [20] |
SUN L C, LUO Y X, TIAN Z L, et al. High temperature corrosion of (Er1/4Tm1/4Yb1/4Lu1/4)2Si2O7 environmental barrier coating material subjected to water vapor and molten calcium-magnesium- aluminosilicate (CMAS). Corrosion Science, 2020, 175: 108881.
DOI URL |
| [21] |
KRESSE G, FURTHMULLER J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Physical Review B, 1996, 54(16): 11169.
DOI URL |
| [22] |
BLOCHL P E. Projector augmented-wave method. Physical Review B, 1994, 50(24): 17953.
DOI PMID |
| [23] |
PERDEW J P, BURKE K, ERNZERHOF M. Generalized gradient approximation made simple. Physical Review Letters, 1997, 77(18): 3865.
DOI URL |
| [24] |
MONKHORST H J, PACK J D. Special points for Brillouin-zone integrations. Physical Review B, 1976, 13(12): 5188.
DOI URL |
| [25] |
LUO Y X, WANG J, SUN L C, et al. Phononic origin of the infrared dielectric properties of RE2O3 (RE=Y, Gd, Ho, Lu) compounds. Modelling and Simulation in Materials Science and Engineering, 2024, 32(5): 055009.
DOI |
| [26] | VOIG W. Lehrbuch der Kristallphysik 962. Leipzig: Teubner, 1928. |
| [27] | REUSS A. Berechnung der fließgrenze von mischkristallen auf grund der plastizitätsbedingung für einkristalle. Journal of Applied Mathematics and Mechanics, 1929, 9(1): 49. |
| [28] | HILL R. The elastic behaviour of a crystalline aggregate. Proceedings of the Physical Society. Section A, 1952, 65(5): 349. |
| [29] |
LUO Y X, WANG J M, LI J N, et al. Theoretical study on crystal structures, elastic stiffness, and intrinsic thermal conductivities of β-, γ-, and δ-Y2Si2O7. Journal of Materials Research, 2015, 30(4): 493.
DOI URL |
| [30] | NYE J F. Physical properties of crystals:their representation by tensors and matrices. Oxford: Oxford University Press, 1985. |
| [31] | KITTEL C, MCEUEN P. Introduction to solid state physics. New York: John Wiley & Sons, 2018. |
| [32] |
LUO Y X, SUN L C, WANG J M, et al. Material-genome perspective towards tunable thermal expansion of rare-earth disilicates. Journal of the European Ceramic Society, 2018, 38(10): 3547.
DOI URL |
| [33] | CLARKE D R. Materials selection guidelines for low thermal conductivity thermal barrier coatings. Surface and Coatings Technology, 2003, 163: 67. |
| [34] | MORELLI D T, SLACK G A. High lattice thermal conductivity solids. New York: Springer New York, 2006: 37-68. |
| [35] |
ANDERSON O L. A simplified method for calculating the Debye temperature from elastic constants. Journal of Physics and Chemistry of Solids, 1963, 24(7): 909.
DOI URL |
| [36] |
SANDITOV B D, TSYDYPOV S B, SANDITOV D S. Relation between the Grüneisen constant and Poisson’s ratio of vitreous systems. Acoustical Physics, 2007, 53: 594.
DOI URL |
| [37] | BRULSR J. The thermal conductivity of magnesium silicon nitride, MgSiN2, ceramics and related materials. Eindhoven: PhD in Chemical Engineering and Chemistry from Eindhoven University of Technology, 2000. |
| [38] |
LUO Y X, WANG JM J, LI Y R, et al. Giant phonon anharmonicity and anomalous pressure dependence of lattice thermal conductivity in Y2Si2O7 silicate. Scientific Reports, 2016, 6: 29801.
DOI |
| [39] |
WEI G, ZUO Y, LUO F, et al. Investigation of mechanical and thermodynamic properties of La2Zr2O7 pyrochlore. International Journal of Energy Research, 2022, 46(2): 2011.
DOI URL |
| [40] |
LUO F, LI B, GUO Z, et al. Ab initio calculation of mechanical and thermodynamic properties of Gd2Zr2O7 pyrochlore. Materials Chemistry and Physics, 2020, 243: 122565.
DOI URL |
| [41] |
CAO X Q, VASSEN R, STOVER D. Ceramic materials for thermal barrier coatings. Journal of the European Ceramic Society, 2004, 24(1): 1.
DOI URL |
| [42] |
TIAN Z L, ZHENG L Y, WANG J M, et al. Theoretical and experimental determination of the major thermo-mechanical properties of RE2SiO5 (RE=Tb, Dy, Ho, Er, Tm, Yb, Lu, and Y) for environmental and thermal barrier coating applications. Journal of the European Ceramic Society, 2016, 36(1): 189.
DOI URL |
| [43] |
TIAN Z L, ZHENG L Y, LI Z, et al. Exploration of the low thermal conductivities of γ-Y2Si2O7, β-Y2Si2O7, β-Yb2Si2O7, and β-Lu2Si2O7 as novel environmental barrier coating candidates. Journal of the European Ceramic Society, 2016, 36(11): 2813.
DOI URL |
| [1] | WU Yuhao, PENG Renci, CHENG Chunyu, YANG Li, ZHOU Yichun. First-principles Study on Mechanical Properties and Melting Curve of HfxTa1-xC System [J]. Journal of Inorganic Materials, 2024, 39(7): 761-768. |
| [2] | ZHOU Jingyu, LI Xingyu, ZHAO Xiaolin, WANG Youwei, SONG Erhong, LIU Jianjun. Rate and Cycling Performance of Ti and Cu Doped β-NaMnO2 as Cathode of Sodium-ion Battery [J]. Journal of Inorganic Materials, 2024, 39(12): 1404-1412. |
| [3] | ZHOU Yunkai, DIAO Yaqi, WANG Minglei, ZHANG Yanhui, WANG Limin. First-principles Calculation Study of the Oxidation Resistance of PANI Modified Ti3C2(OH)2 [J]. Journal of Inorganic Materials, 2024, 39(10): 1151-1158. |
| [4] | WU Xiaowei, ZHANG Han, ZENG Biao, MING Chen, SUN Yiyang. Comparison of Hybrid Functionals HSE and PBE0 in Calculating the Defect Properties of CsPbI3 [J]. Journal of Inorganic Materials, 2023, 38(9): 1110-1116. |
| [5] | GU Junyi, FAN Wugang, ZHANG Zhaoquan, YAO Qin, ZHAN Hongquan. Preparation and Thermal Property of PrAlO3 Ceramics [J]. Journal of Inorganic Materials, 2023, 38(10): 1200-1206. |
| [6] | AN Wenran, HUANG Jingqi, LU Xiangrong, JIANG Jianing, DENG Longhui, CAO Xueqiang. Effect of Heat-treatment Temperature on Thermal and Mechanical Properties of LaMgAl11O19 Coating [J]. Journal of Inorganic Materials, 2022, 37(9): 925-932. |
| [7] | WEN Zhiqin, HUANG Binrong, LU Taoyi, ZOU Zhengguang. Pressure on the Structure and Thermal Properties of PbTiO3: First-principle Study [J]. Journal of Inorganic Materials, 2022, 37(7): 787-794. |
| [8] | XIAO Meixia, LI Miaomiao, SONG Erhong, SONG Haiyang, LI Zhao, BI Jiaying. Halogenated Ti3C2 MXene as High Capacity Electrode Material for Li-ion Batteries [J]. Journal of Inorganic Materials, 2022, 37(6): 660-668. |
| [9] | YUAN Gang, MA Xinguo, HE Hua, DENG Shuiquan, DUAN Wangyang, CHENG Zhengwang, ZOU Wei. Plane Strain on Band Structures and Photoelectric Properties of 2D Monolayer MoSi2N4 [J]. Journal of Inorganic Materials, 2022, 37(5): 527-533. |
| [10] | WEI Hailang, CAO Xueqiang, DENG Longhui, JIANG Jianing. Thermodynamic Properties and Thermal Cycling Lifetimes of LaMeAl11O19/YSZ Thermal Barrier Coatings [J]. Journal of Inorganic Materials, 2022, 37(12): 1259-1266. |
| [11] | PENG Junhui, TIKHONOV Evgenii. Vacancy on Structures, Mechanical and Electronic Properties of Ternary Hf-Ta-C System: a First-principles Study [J]. Journal of Inorganic Materials, 2022, 37(1): 51-57. |
| [12] | ZHU Jiatong, LOU Zhihao, ZHANG Ping, ZHAO Jia, MENG Xuanyu, XU Jie, GAO Feng. Preparation and Thermal Properties of Rare Earth Tantalates (RETaO4) High-Entropy Ceramics [J]. Journal of Inorganic Materials, 2021, 36(4): 411-417. |
| [13] | YAN Yuxing, WANG Fan, ZHANG Juexuan, LI Fushao. First Principles Study of Electronic Structure and Optical Properties of ZnNb2O6 with Vacancy Defects [J]. Journal of Inorganic Materials, 2021, 36(3): 269-276. |
| [14] | ZHAO Linyan, LIU Yangsi, XI Xiaoli, MA Liwen, NIE Zuoren. First-principles Study on Nanoscale Tungsten Oxide: a Review [J]. Journal of Inorganic Materials, 2021, 36(11): 1125-1136. |
| [15] | JI Xiaojuan,YU Yueguang,LU Xiaoliang. Effects of Impurities on Properties of YSZ Thermal Barrier Coatings [J]. Journal of Inorganic Materials, 2020, 35(6): 669-674. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||