无机材料学报 ›› 2020, Vol. 35 ›› Issue (9): 959-971.DOI: 10.15541/jim20190548 CSTR: 32189.14.10.15541/jim20190548
所属专题: 封面文章; MXene材料专辑(2020~2021); 【虚拟专辑】层状MAX,MXene及其他二维材料
• 综述 • 下一篇
收稿日期:2019-10-28
修回日期:2019-12-04
出版日期:2020-09-20
网络出版日期:2020-04-05
作者简介:杨浏鑫(1995-), 女, 博士研究生. E-mail: 基金资助:
YANG Liuxin1,2( ),LUO Wenhua2,WANG Changan1,XU Chen2(
),LUO Wenhua2,WANG Changan1,XU Chen2( )
)
Received:2019-10-28
Revised:2019-12-04
Published:2020-09-20
Online:2020-04-05
Supported by:摘要:
气体膜分离技术是过滤与分离工业的重要技术之一, 相比于传统分离技术更加高效、节能、环保。新型无机二维材料在分离膜领域的应用, 有望同时实现高选择性和高渗透率, 突破商业聚合物膜渗透率和选择性相互制约的瓶颈, 极大地促进高性能分离膜的发展。本文简述了膜的气体分离机制, 综述了石墨烯基、过渡金属硫族化物(TMDs)和二维过渡金属碳化物/氮化物(MXene)等新型无机二维材料近年来在气体分离膜领域的研究进展, 包括其设计、制造和应用, 探讨了不同材料分离膜的特点、面临的挑战和发展前景。此外, 本文对其他新兴二维材料——层状双氢氧化物(LDHs)、六方氮化硼(h-BN)、云母纳米片等的分离膜研究也进行了概述。最后, 对新型无机二维材料在气体分离膜领域的研究方向及面临的挑战作出了评价。
中图分类号:
杨浏鑫,罗文华,汪长安,徐晨. 新型无机二维材料在气体分离膜领域的研究进展[J]. 无机材料学报, 2020, 35(9): 959-971.
YANG Liuxin,LUO Wenhua,WANG Changan,XU Chen. Novel Inorganic Two-dimensional Materials for Gas Separation Membranes[J]. Journal of Inorganic Materials, 2020, 35(9): 959-971.
图1 膜的传输机制示意图, 孔直径定义为Dp = Dc -Dvdw/$\sqrt{2}$, 其中Dc是碳原子中心之间的直径, Dvdw是碳原子的范德华直径[1,10,11,14-17]
Fig. 1 Schematic representation of transport mechanisms in membranes, where Dc is the location of carbon centers, and the pore diameter is defined as Dp = Dc - Dvdw/$\sqrt{2}$, where Dvdw is the van der Waals diameter of a carbon atom[1,10,11,14-17]
图2 石墨烯上形成纳米孔的示意图(a~b)和多孔石墨烯的孔电子密度等值面(c~d)[28]
Fig. 2 Schematic of pores in graphene sheet (a-b) and pore electron density isosurfaces of porous graphene (c-d)[28] (a) Creation of a nitrogen-functionalized pore within a graphene sheet; (b) An all-hydrogen passivated pore in graphene; (c) Pore electron density isosurface of the nitrogen-functionalized porous graphene; (d) The all-hydrogen passivated porous grapheme Color code: C-black; N-green; H-cyan (isovalue is at 0.2 e/nm)
图4 纳米多孔碳膜辅助转移法制备大面积石墨烯膜示意图(a)和石墨烯点阵中本征缺陷的高分辨TEM照片(b)[33]
Fig. 4 Schematic of fabrication of a large-area graphene membrane by the nanoporous carbon film-assisted transfer method (a) and high-resolution TEM images of the intrinsic defects in graphene lattice (b)[33]
图5 1 μm厚GO膜的照片(a), 分离膜的截面SEM照片(b), 分子在GO膜层间的渗透路径示意照片(c)和自支撑的亚微米厚GO膜和对照组PET膜的He渗透性能对比(d)[40] (1 mbar=100 Pa)
Fig. 5 Photo of a 1-μm-thick GO film (a), electron micrograph of the film’s cross section (b), schematic view for possible permeation through the laminates (c), and examples of He-leak measurements for a freestanding submicrometer-thick GO membrane and a reference PET film (d)[40] (1 mbar=100 Pa)
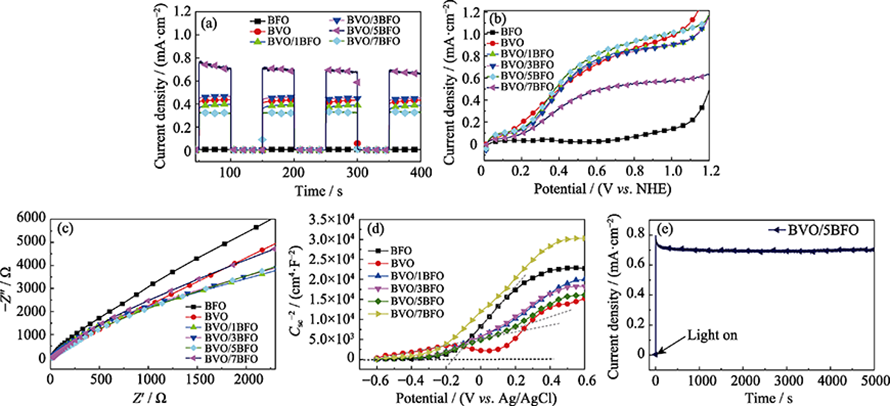
图6 不同的GO膜制备方法示意图(a), GO膜可能的气体传输途径示意图(b), GO膜表面的AFM图与表面深度分布(c, d)[19]
Fig. 6 Schematic illustration of two different GO coating methods (Method one and Method two) (a), schematic illustration of possible gas transport through graphene membrane (b), AFM images of GO membrane surfaces with insert images showing depth profiles of GO membrane surfaces: (c) method one [root mean square roughness (Rq) = 0.8 nm, average roughness (Ra) = 0.614 nm] and (d) method two (Rq = 0.608 nm, Ra = 0.467 nm)[19]
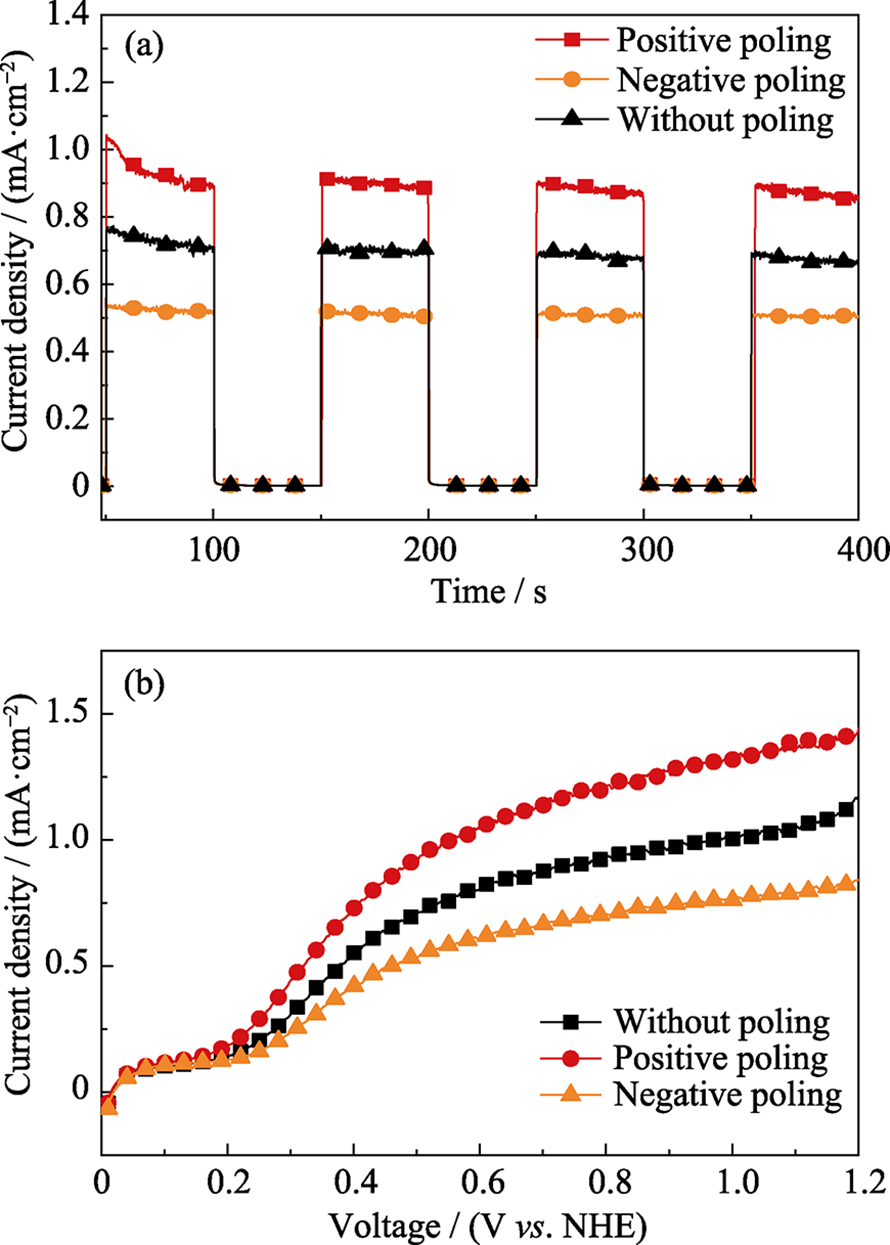
图7 PEBA基膜中组装的GO纳米片示意图(a), 含有0.1wt%GO的膜(b)和TEM照片(黄色虚线指示GO片层) (c), 以及GO-1膜横截面的TEM照片(d)[17]
Fig. 7 Schematic representation of the assembly of GO nanosheets in polymeric environment (a), digital photographs of the membrane with 0.1wt% GO (b), overview (the yellow dashed lines are eye-guiding lines indicating the GO laminates in these regions) (c) and expanded TEM image of the cross section of GO-1 membrane (d)[17]
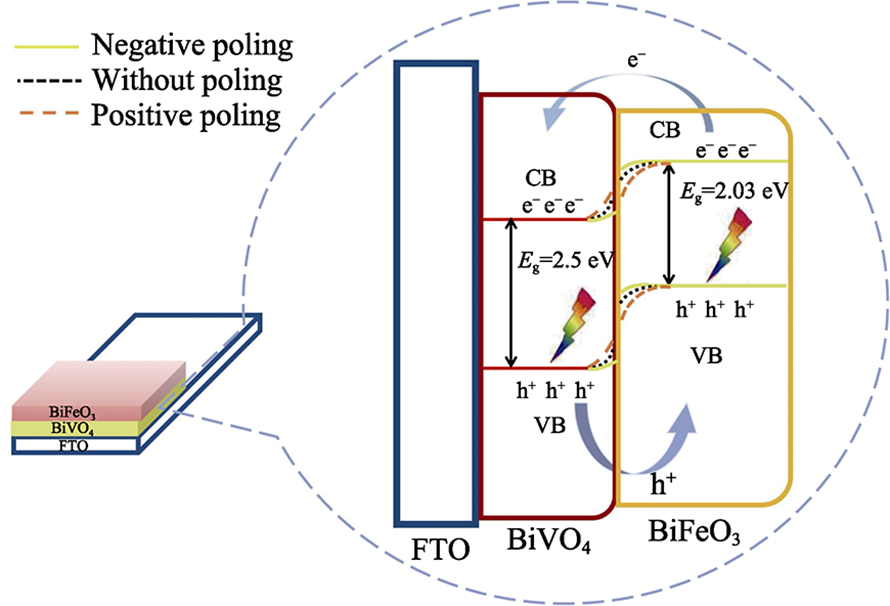
图8 GO纳米片和聚合物链组成的二维通道的受力分析示意图(a), 本征力诱导的无序结构(左)和协同外力驱动的高度有序层状结构(右)示意图(b)[54]
Fig. 8 Force analysis for one 2D channel unit consisting of GO nanosheets and polymer chain (a), schematic illustration of intrinsic force induced disordered structure (left) and highly ordered laminar structures (right) driven by introduced synergistic external forces (b)[54]
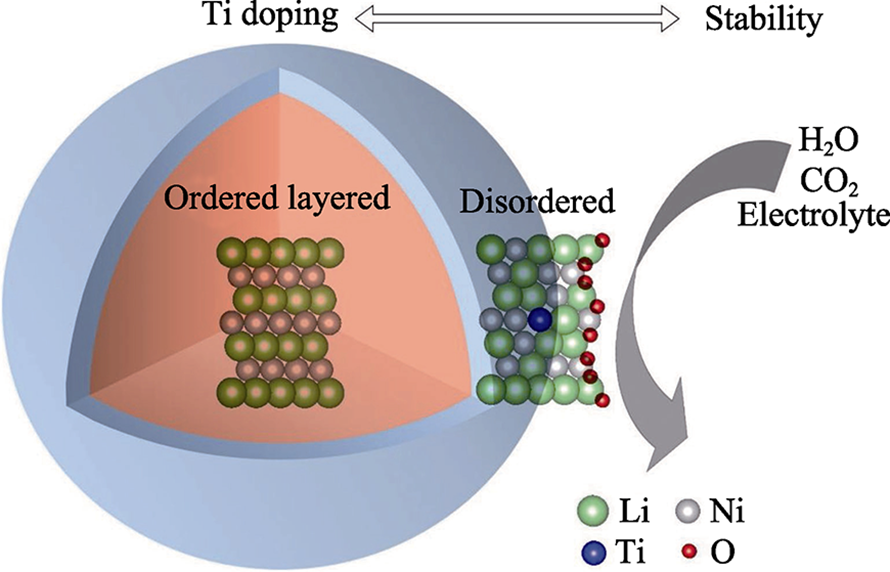
图9 气体通过GO-SILM膜溶解-扩散传输路径示意图(a), 限制在GO纳米通道中的[BMIM][BF4]离子液体分子结构(b), GO膜(c)和GO-SILM膜(d)的截面SEM照片[55]
Fig. 9 Schematic illustration of the gas solution-diffusion transport pathway through a GO-SILM (a), molecular structures of [BMIM][BF4] IL confined in the GO nanochannels (b), the cross section of the GO membrane (c) and GO-SILM (d), respectively[55]
| Materialsa | Preparation method | Feed condition | Selectivity | Permeate rate/ permeance/permeability | Ref. |
|---|---|---|---|---|---|
| Porous graphene monolayer | Simulation All-H passivation | H2/CH4 | 1023 | 10-20 mol?s-1?Pa-1 | [ |
| Porous graphene bilayer | Ultraviolet-induced oxidative etching | H2/CH4 | 104 | 4.5×10-23 mol?s-1?Pa-1 | [ |
| Porous graphene bilayer | Focused ion beam perforation | H2/CO2 | 4.6 | 5.0×103 mol?m-2?s-1?Pa-1 | [ |
| Porous graphene | Ozone functionalization-based pore-etching | H2/CH4 | 25 | 4.1×10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/PES | Spin coating | H2/CO2 | 30 | 4.02×10-17 mol?m?m-2?s-1?Pa-1 | [ |
| Dip and spin coating | H2/CO2 | 20 | 5.69×10-17 mol?m?m-2?s-1?Pa-1 | ||
| GO/AAO | Vacuum filtration | H2/CO2 | 3400 | 10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/AAO | Vacuum filtration | H2/CO2 | 22.5 | 1.14×10-7 mol?m-2?s-1?Pa-1 | [ |
| TU-GOF | Hydrothermal Self-assembly synthesis | H2/CO2 | 225 | 10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/AAO | Spin coating | H2/CO2 | 240 | 3.4×10-7 mol?m-2?s-1?Pa-1 | [ |
| PEBA-GO | Drop casting | CO2/N2 | 91 | 3.35×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| SPEEK/S-GO | Drop casting | CO2/CH4 | 72.2 | 4.44×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| EFDA-GO | Vaccum-spin | H2/CO2 | 29-33 | 2.8-4.0×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| GO-[BMIM][BF4] | Vacuum filtration | CO2/ H2 | 24 | 2.29×10-8 mol?m-2?s-1?Pa-1 | [ |
| CO2/ CH4 | 234 | ||||
| CO2/N2 | 382 |
表1 石墨烯基膜的气体分离性能比较
Table 1 Graphene-based membranes for gas separation
| Materialsa | Preparation method | Feed condition | Selectivity | Permeate rate/ permeance/permeability | Ref. |
|---|---|---|---|---|---|
| Porous graphene monolayer | Simulation All-H passivation | H2/CH4 | 1023 | 10-20 mol?s-1?Pa-1 | [ |
| Porous graphene bilayer | Ultraviolet-induced oxidative etching | H2/CH4 | 104 | 4.5×10-23 mol?s-1?Pa-1 | [ |
| Porous graphene bilayer | Focused ion beam perforation | H2/CO2 | 4.6 | 5.0×103 mol?m-2?s-1?Pa-1 | [ |
| Porous graphene | Ozone functionalization-based pore-etching | H2/CH4 | 25 | 4.1×10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/PES | Spin coating | H2/CO2 | 30 | 4.02×10-17 mol?m?m-2?s-1?Pa-1 | [ |
| Dip and spin coating | H2/CO2 | 20 | 5.69×10-17 mol?m?m-2?s-1?Pa-1 | ||
| GO/AAO | Vacuum filtration | H2/CO2 | 3400 | 10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/AAO | Vacuum filtration | H2/CO2 | 22.5 | 1.14×10-7 mol?m-2?s-1?Pa-1 | [ |
| TU-GOF | Hydrothermal Self-assembly synthesis | H2/CO2 | 225 | 10-7 mol?m-2?s-1?Pa-1 | [ |
| GO/AAO | Spin coating | H2/CO2 | 240 | 3.4×10-7 mol?m-2?s-1?Pa-1 | [ |
| PEBA-GO | Drop casting | CO2/N2 | 91 | 3.35×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| SPEEK/S-GO | Drop casting | CO2/CH4 | 72.2 | 4.44×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| EFDA-GO | Vaccum-spin | H2/CO2 | 29-33 | 2.8-4.0×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| GO-[BMIM][BF4] | Vacuum filtration | CO2/ H2 | 24 | 2.29×10-8 mol?m-2?s-1?Pa-1 | [ |
| CO2/ CH4 | 234 | ||||
| CO2/N2 | 382 |
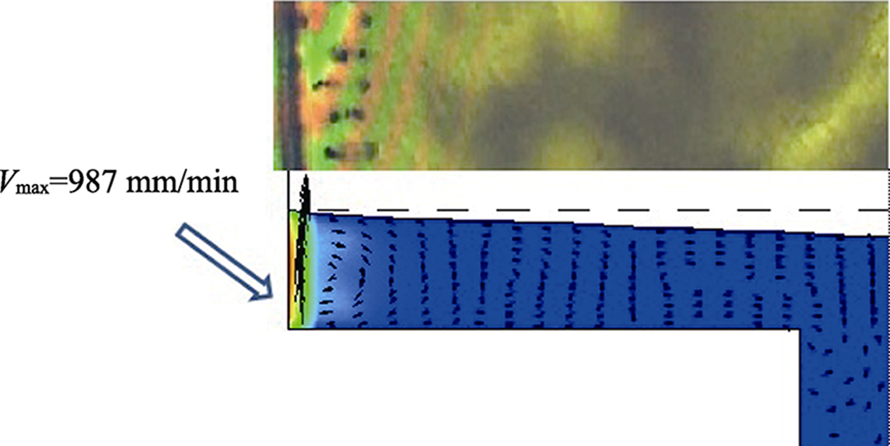
图10 MoS2膜160 ℃热处理前后的气体渗透路径示意图(a)[63], 膜截面的SEM照片(b~e)[64]和MoS2-SILM制备过程示意图(f)[69]
Fig.10 Schematic of gas permeation pathway across MoS2 membranes before and after heating at 160 ℃(a)[63], cross section SEM images of the membrane (b-e)[64], and synthesis process for MoS2-SILM (f)[69](b) GO membrane; (c) MoS2 membrane; (d) GO-MoS2 (50/50) hybrid membrane; (e) GO-MoS2 (75/25) hybrid membrane
图11 AAO衬底上MXene的SEM照片(a) (插图为MXene胶体水溶液的科根达尔效应), MXene膜的截面SEM照片(b) (插图为弯折的膜), MXene片层的AFM图(c), 相邻MXene纳米片示意图(d)[77], 以及MXene膜分别选择性渗透H2和CO2的示意图(e)[78]
Fig. 11 SEM image of the delaminated MXene nanosheets on porous anodic aluminum oxide (AAO) (a) with insert showing the Tyndall scattering effect in MXene colloidal solution in water, cross-sectional SEM image of the MXene membrane (b) with insert showing a tweezer bent membrane, AFM image of the Mxene nanosheet on cleaved mica (c), illustration of the spacing between the neighboring MXene nanosheets in the membrane (d)[77], structures and gas transport of H2-selective and CO2-selective Mxene nanofilms (e)[78]
| Materialsa | Preparation method | Feed Condition | Selectivity | Permeate rate/permeance/ permeability | Ref. |
|---|---|---|---|---|---|
| MoS2/AAO | Vacuum filtration | H2/CO2 | 3.4 | 9.19×10-6 mol?m-2?s-1?Pa-1 | [ |
| MoS2 | Vacuum filtration | H2/CO2 | 8.29 | 3.94×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| GO/MoS2 | Vacuum filtration | H2/CO2 | 26.7 | 8.04×10-7 mol m-2 s-1 Pa-1 | [ |
| MoS2-Pebax | Spin coating | CO2/N2 | 93 | 2.14×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| MoS2-[BMIM][BF4] | Vacuum filtration and drop | CO2/N2 | 131.42 | 1.60×10-8 mol?m-2?s-1?Pa-1 | [ |
| WS2-[BMIM][BF4] | Vacuum filtration and spin-coating | CO2/N2 | 153.21 | 1.58×10-8 mol?m-2?s-1?Pa-1 | [ |
| Ti2C3Tx | Vacuum filtration | H2/CO2 | 160 | 7.37×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| Ti2C3Tx/borate-PEI | Vacuum filtration and spin-coating | CO2/CH4 | 15.3 | 1.17×10-7 mol?m-2?s-1?Pa-1 | [ |
| MgAl-CO3 LDH | Electrophoretic deposition | CO2/N2 | 1.53 | 2.34×10-7 mol?m-2?s-1?Pa-1 | [ |
| MgAl-CO3 LDH | Vacuum-suction | CO2/N2 | 34.4 | 5.5×10-10 mol?m-2?s-1?Pa-1 | [ |
| NiAl-CO3 LDH | In situ growth | H2/CH4 | 80 | 4.5×10-8 mol?m-2?s-1?Pa-1 | [ |
| h-BN-XTR | Drop casting | H2/CH4 | 24.1 | 7.03×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| mica-[BMIM][BF4] | Vacuum filtration and drop-casting | CO2/N2 | 87 | 2.68×10-8 mol?m-2?s-1?Pa-1 | [ |
表2 其他新型无机2D材料气体膜分离性能比较
Table 2 Other novel inorganic 2DMs for membrane gas separation
| Materialsa | Preparation method | Feed Condition | Selectivity | Permeate rate/permeance/ permeability | Ref. |
|---|---|---|---|---|---|
| MoS2/AAO | Vacuum filtration | H2/CO2 | 3.4 | 9.19×10-6 mol?m-2?s-1?Pa-1 | [ |
| MoS2 | Vacuum filtration | H2/CO2 | 8.29 | 3.94×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| GO/MoS2 | Vacuum filtration | H2/CO2 | 26.7 | 8.04×10-7 mol m-2 s-1 Pa-1 | [ |
| MoS2-Pebax | Spin coating | CO2/N2 | 93 | 2.14×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| MoS2-[BMIM][BF4] | Vacuum filtration and drop | CO2/N2 | 131.42 | 1.60×10-8 mol?m-2?s-1?Pa-1 | [ |
| WS2-[BMIM][BF4] | Vacuum filtration and spin-coating | CO2/N2 | 153.21 | 1.58×10-8 mol?m-2?s-1?Pa-1 | [ |
| Ti2C3Tx | Vacuum filtration | H2/CO2 | 160 | 7.37×10-13 mol?m?m-2?s-1?Pa-1 | [ |
| Ti2C3Tx/borate-PEI | Vacuum filtration and spin-coating | CO2/CH4 | 15.3 | 1.17×10-7 mol?m-2?s-1?Pa-1 | [ |
| MgAl-CO3 LDH | Electrophoretic deposition | CO2/N2 | 1.53 | 2.34×10-7 mol?m-2?s-1?Pa-1 | [ |
| MgAl-CO3 LDH | Vacuum-suction | CO2/N2 | 34.4 | 5.5×10-10 mol?m-2?s-1?Pa-1 | [ |
| NiAl-CO3 LDH | In situ growth | H2/CH4 | 80 | 4.5×10-8 mol?m-2?s-1?Pa-1 | [ |
| h-BN-XTR | Drop casting | H2/CH4 | 24.1 | 7.03×10-14 mol?m?m-2?s-1?Pa-1 | [ |
| mica-[BMIM][BF4] | Vacuum filtration and drop-casting | CO2/N2 | 87 | 2.68×10-8 mol?m-2?s-1?Pa-1 | [ |
| [1] |
LIU G, JIN W, XU N. Graphene-based membranes. Chemical Society Reviews, 2015,44(15):5016-5030.
URL PMID |
| [2] |
SHOLL D S, LIVELY R P. Seven chemical separations to change the world. Nature, 2016,532(7600):435-437.
DOI URL PMID |
| [3] |
LI P, WANG Z, QIAO Z, et al. Recent developments in membranes for efficient hydrogen purification. Journal of Membrane Science, 2015. 495:130-168.
DOI URL |
| [4] | JEON Y W, LEE D H. Gas membranes for CO2/CH4 (biogas) separation: a review. Environmental Engineering Science, 2015,32(2):71-85. |
| [5] | DALANE K, DAI Z, MOGSETH G, et al. Potential applications of membrane separation for subsea natural gas processing: a review. Journal of Natural Gas Science and Engineering, 2017,39:101-117. |
| [6] | HIMMA N F, WARDANI A K, PRASETYA N, et al. Recent progress and challenges in membrane-based O2/N2 separation. Reviews in Chemical Engineering, 2019,35(5):591-625. |
| [7] | ZHU J, HOU J, ULIANA A, et al. The rapid emergence of two-dimensional nanomaterials for high-performance separation membranes. Journal of Materials Chemistry A, 2018,6(9):3773-3792. |
| [8] |
GIN D L, NOBLE R D. Designing the next generation of chemical separation membranes. Science, 2011,332(6030):674.
URL PMID |
| [9] | QIU T, KUANG C, ZHENG X, et al. On the research and application trends of global gas membrane separation technology—— based on analysis of SCI articles and patents in recent 20 years. Chemical Industry & Engineering Progress, 2016,35(7):2299-2308. |
| [10] | YAMPOLSKII Y. Polymeric gas separation membranes. Macromolecules, 2012,45(8):3298-3311. |
| [11] | PROZOROVSKA L, KIDAMBI P R. State-of-the-art and future prospects for atomically thin membranes from 2D materials. Advanced Materials, 2018,30(52):1801179. |
| [12] | LIU M, GURR P A, FU Q, et al. Two-dimensional nanosheet-based gas separation membranes. Journal of Materials Chemistry A, 2018,6(46):23169-23196. |
| [13] |
LIU G, JIN W, XU N. Two-dimensional-material membranes: a new family of high-performance separation membranes. Angewandte Chemie International Edition, 2016,55(43):13384-13397.
URL PMID |
| [14] |
WANG L, BOUTILIER M S H, KIDAMBI P R, et al. Fundamental transport mechanisms, fabrication and potential applications of nanoporous atomically thin membranes. Nature Nanotechnology, 2017,12:509.
DOI URL PMID |
| [15] | WIJMANS J G, BAKER R W J. The solution-diffusion model: a review. Journal of Membrane Science, 1995,107:1-21. |
| [16] | LI C, MECKLER S M, SMITH Z P, et al. Engineered transport in microporous materials and membranes for clean energy technologies. Advanced Materials, 2018,30(8):1704953. |
| [17] | SHEN J, LIU G, HUANG K, et al. Membranes with fast and selective gas-transport channels of laminar graphene oxide for efficient CO2 capture. Angewandte Chemie International Edition, 2015,127(2):588-592. |
| [18] |
DRAHUSHUK L W, WANG L, KOENIG S P, et al. Analysis of time-varying, stochastic gas transport through graphene membranes. ACS Nano, 2016,10(1):786-795.
URL PMID |
| [19] |
KIM H W, YOON H W, YOON S M, et al. Selective gas transport through few-layered graphene and graphene oxide membranes. Science, 2013,342(6154):91-95.
URL PMID |
| [20] |
LI H, SONG Z, ZHANG X, et al. Ultrathin, molecular-sieving graphene oxide membranes for selective hydrogen separation. Science, 2013,342(6154):95-98.
URL PMID |
| [21] |
WANG Z, WANG D, ZHANG S, et al. Interfacial design of mixed matrix membranes for improved gas separation performance. Advanced Materials, 2016,28(17):3399-3405.
URL PMID |
| [22] |
ZHU X, TIAN C, DO-THANH C L, et al. Two‐dimensional materials as prospective scaffolds for mixed‐matrix membrane‐based CO2 separation. ChemSusChem, 2017,10(17):3304-3316.
DOI URL PMID |
| [23] |
GEIM A K, NOVOSELOV K S. The rise of graphene. Nature Materials, 2007,6:183.
DOI URL PMID |
| [24] |
PARTOENS B, PEETERS F. From graphene to graphite: electronic structure around the K point. Phys. Rev. B, 2006,74(7):075404.
DOI URL |
| [25] |
BUNCH J S, VERBRIDGE S S, ALDEN J S, et al. Impermeable atomic membranes from graphene sheets. Nano Letters, 2008,8(8):2458-2462.
URL PMID |
| [26] |
BERRY V. Impermeability of graphene and its applications. Carbon, 2013,62:1-10.
DOI URL |
| [27] |
LEE C, WEI X, KYSAR J W, et al. Measurement of the elastic properties and intrinsic strength of monolayer graphene. Science, 2008,321(5887):385-388.
URL PMID |
| [28] |
JIANG D E, COOPER V, DAI S. Porous graphene as the ultimate membrane for gas separation. Nano Letters, 2009,9:4019-4024.
URL PMID |
| [29] | WANG S, DAI S, JIANG D E. Continuously tunable pore size for gas separation via a bilayer nanoporous graphene membrane. ACS Applied Nano Materials, 2019,2(1):379-384. |
| [30] |
KOENIG S P, WANG L, PELLEGRINO J, et al. Selective molecular sieving through porous graphene. Nature Nanotechnology, 2012,7(11):728-732.
URL PMID |
| [31] |
BELL D, LEMME M, STERN L, et al. Precision cutting and patterning of graphene with helium ions. Nanotechnology, 2009,20:455301.
DOI URL PMID |
| [32] |
CELEBI K, BUCHHEIM J, WYSS R M, et al. Ultimate permeation across atomically thin porous graphene. Science, 2014,344(6181):289-292.
URL PMID |
| [33] |
HUANG S, DAKHCHOUNE M, LUO W, et al. Single-layer graphene membranes by crack-free transfer for gas mixture separation. Nature Communications, 2018,9(1):2632-2632.
DOI URL PMID |
| [34] |
FISCHBEIN M D, DRNDIĆ M. Electron beam nanosculpting of suspended graphene sheets. Applied Physics Letters, 2008,93(11):113107.
DOI URL |
| [35] |
LU N, WANG J, FLORESCA H C, et al. In situ studies on the shrinkage and expansion of graphene nanopores under electron beam irradiation at temperatures in the range of 400-1200 ℃. Carbon, 2012,50(8):2961-2965.
DOI URL |
| [36] |
GARAJ S, HUBBARD W, REINA A, et al. Graphene as a subnanometre trans-electrode membrane. Nature, 2010,467:190.
DOI URL PMID |
| [37] | MERCHANT C. DNA translocation through graphene nanopores. Biophysical Journal, 2011,100(3):521a. |
| [38] |
HUMMERS JR W S, OFFEMAN R E. Preparation of graphitic oxide. Journal of the American Chemical Society, 1958,80(6):1339.
DOI URL |
| [39] |
DIMIEV A, TOUR J. Mechanism of graphene oxide formation. ACS Nano, 2014,8(3):3060-3067.
URL PMID |
| [40] |
NAIR R R, WU H A, JAYARAM P N, et al. Unimpeded permeation of water through helium-leak-tight graphene-based membranes. Science, 2012,335(6067):442-444.
DOI URL PMID |
| [41] |
IBRAHIM A, LIN Y S. Gas permeation and separation properties of large-sheet stacked graphene oxide membranes. Journal of Membrane Science, 2018,550:238-245.
DOI URL |
| [42] | YANG J, GONG D, LI G, et al. Self-assembly of thiourea-crosslinked graphene oxide framework membranes toward separation of small molecules. Advanced Materials, 2018,30(16):1705775. |
| [43] |
ABRAHAM J, VASU K S, WILLIAMS C D,et al. Tunable sieving of ions using graphene oxide membranes. Nature Nanotechnology, 2017,12(6):546-550.
URL PMID |
| [44] |
CHI C, WANG X, PENG Y, et al. Facile preparation of graphene oxide membranes for gas separation. Chemistry of Materials, 2016,28(9):2921-2927.
DOI URL |
| [45] | YANG E, HAM M H, PARK H B, et al. Tunable semi-permeability of graphene-based membranes by adjusting reduction degree of laminar graphene oxide layer. Journal of Membrane Science, 2018,547:73-79. |
| [46] |
SU Y, KRAVETS V G, WONG S L, et al. Impermeable barrier films and protective coatings based on reduced graphene oxide. Nature Communications, 2014,5(1):4843.
DOI URL PMID |
| [47] |
SUN P, WANG K, ZHU H. Recent developments in graphene-based membranes: structure, mass-transport mechanism and potential applications. Advanced Materials, 2016,28(12):2287-2310.
DOI URL PMID |
| [48] |
ZHANG Y, ZHANG S, CHUNG T S. Nanometric graphene oxide framework membranes with enhanced heavy metal removal via nanofiltration. Environmental Science & Technology, 2015,49(16):10235-10242.
DOI URL PMID |
| [49] |
BURRESS J W, GADIPELLI S, FORD J, et al. Graphene oxide framework materials: theoretical predictions and experimental results. Angewandte Chemie International Edition, 2010,49(47):8902-8904.
URL PMID |
| [50] | KARUNAKARAN M, VILLALOBOS L F, KUMAR M, et al. Graphene oxide doped ionic liquid ultrathin composite membranes for efficient CO2 capture. Journal of Materials Chemistry A, 2017,5(2):649-656. |
| [51] | WANG S, XIE Y, HE G, et al. Graphene oxide membranes with heterogeneous nanodomains for efficient CO2 separations. Angewandte Chemie, 2017,129(45):14434-14439. |
| [52] | HUANG G, ISFAHANI A P, MUCHTAR A, et al. Pebax/ionic liquid modified graphene oxide mixed matrix membranes for enhanced CO2 capture. Journal of Membrane Science, 2018,565:370-379. |
| [53] | XIN Q, MA F, ZHANG L, et al. Interface engineering of mixed matrix membrane via CO2-philic polymer brush functionalized graphene oxide nanosheets for efficient gas separation. Journal of Membrane Science, 2019,586:23-33. |
| [54] |
SHEN J, LIU G, HUANG K, et al. Subnanometer two-dimensional graphene oxide channels for ultrafast gas sieving. ACS Nano, 2016,10(3):3398-3409.
DOI URL PMID |
| [55] |
YING W, CAI J, ZHOU K, et al. Ionic liquid selectively facilitates CO2 transport through graphene oxide membrane. ACS Nano, 2018,12(6):5385-5393.
URL PMID |
| [56] |
SCHMIDT H, WANG S, CHU L, et al. Transport properties of monolayer MoS2 grown by chemical vapor deposition. Nano Letters, 2014,14(4):1909-1913.
DOI URL PMID |
| [57] |
NICOLOSI V, CHHOWALLA M, G KANATZIDIS M et al. Liquid exfoliation of layered materials. Science, 2013,340:1226419.
DOI URL |
| [58] |
ZHANG W, HUANG J K, CHEN C H, et al. High-gain phototransistors based on a CVD MoS2 monolayer. Advanced Materials, 2013,25(25):3456-3461.
DOI URL PMID |
| [59] |
WANG H, FENG H, LI J. Graphene and graphene-like layered transition metal dichalcogenides in energy conversion and storage. Small, 2014,10(11):2165-2181.
DOI URL PMID |
| [60] | ZENG Z, SUN T, ZHU J, et al. An effective method for the fabrication of few-layer-thick inorganic nanosheets. Angewandte Chemie International Edition, 2012,124(36):9186-9190. |
| [61] | GEE M A, FRINDT R F, JOENSEN P, et al. Inclusion compounds of MoS2. Materials Research Bulletin, 1986,21(5):543-549. |
| [62] |
WANG D, WANG Z, WANG L, et al. Ultrathin membranes of single- layered MoS2 nanosheets for high-permeance hydrogen separation. Nanoscale, 2015,7(42):17649-17652.
DOI URL PMID |
| [63] | ACHARI A, SAHANA S, ESWARAMOORTHY M. High performance MoS2 membranes: effects of thermally driven phase transition on CO2 separation efficiency. Energy & Environmental Science, 2016,9(4):1224-1228. |
| [64] | OSTWAL M, SHINDE D B, WANG X, et al. Graphene oxide- molybdenum disulfide hybrid membranes for hydrogen separation. Journal of Membrane Science, 2018,550:145-154. |
| [65] | ZHAO S, XUE J, KANG W. Gas adsorption on MoS2 monolayer from first-principles calculations. Chemical Physics Letters, 2014, 595-596:35-42. |
| [66] |
HE Q, ZENG Z, YIN Z, et al. Fabrication of flexible MoS2 thin-film transistor arrays for practical gas-sensing applications. Small, 2012,8(19):2994-2999.
URL PMID |
| [67] |
BEREAN K J, OU J Z, DAENEKE T, et al. 2D MoS2 pdms nanocomposites for NO2 separation. Small, 2015,11(38):5035-5040.
URL PMID |
| [68] |
SHEN Y, WANG H, ZHANG X, et al. MoS2 nanosheets functionalized composite mixed matrix membrane for enhanced CO2 capture via surface drop-coating method. ACS Applied Materials & Interfaces, 2016,8(35):23371-23378.
DOI URL PMID |
| [69] |
CHEN D K, YING W, GUO Y, et al. Enhanced gas separation through nanoconfined ionic liquid in laminated MoS2 membrane. ACS Applied Materials & Interfaces, 2017,9(50):44251-44257.
URL PMID |
| [70] | CHEN D, WANG W, YING W, et al. CO2-philic WS2 laminated membranes with a nanoconfined ionic liquid. Journal of Materials Chemistry A, 2018,6(34):16566-16573. |
| [71] | ALHABEB M, MALESKI K, ANASORI B, et al. Guidelines for synthesis and processing of two-dimensional titanium carbide (Ti3C2Tx MXene). Chemistry of Materials, 2017,29(18):7633-7644. |
| [72] |
WANG J T, CHEN P P, SHI B B, et al. A regularly channeled lamellar membrane for unparalleled water and organics permeation. Angewandte Chemie-International Edition, 2018,57(23):6814-6818.
DOI URL PMID |
| [73] |
DING L, WEI Y, WANG Y, et al. A two-dimensional lamellar membrane: MXene nanosheet stacks. Angewandte Chemie International Edition, 2017,56(7):1825-1829.
DOI URL PMID |
| [74] |
REN C E, HATZELL K B, ALHABEB M, et al. Charge- and size-selective ion sieving through Ti3C2Tx MXene membranes. The Journal of Physical Chemistry Letters, 2015,6(20):4026-4031.
URL PMID |
| [75] |
RASOOL K, MAHMOUD K A, JOHNSON D J, et al. Efficient antibacterial membrane based on two-dimensional Ti3C2Tx (MXene) nanosheets. Sci. Rep, 2017,7(1):1598.
DOI URL PMID |
| [76] |
RASOOL K, HELAL M, ALI A, et al. Antibacterial activity of Ti3C2Tx MXene. ACS Nano, 2016,10(3):3674-3684.
DOI URL PMID |
| [77] |
DING L, WEI Y, LI L, et al. MXene molecular sieving membranes for highly efficient gas separation. Nature Communications, 2018,9:155.
DOI URL PMID |
| [78] |
SHEN J, LIU G Z, JI Y F, et al. 2D MXene nanofilms with tunable gas transport channels. Advanced Functional Materials, 2018,28(31):1801511.
DOI URL |
| [79] |
LI L, ZHANG T, DUAN Y, et al. Selective gas diffusion in two-dimensional MXene lamellar membranes: insights from molecular dynamics simulations. Journal of Materials Chemistry A, 2018,6(25):11734-11742.
DOI URL |
| [80] |
WANG Q, O’HARE D. Recent advances in the synthesis and application of layered double hydroxide (LDH) nanosheets. Chemical Reviews, 2012,112(7):4124-4155.
URL PMID |
| [81] | LU P, LIU Y, ZHOU T, et al. Recent advances in layered double hydroxides (LDHs) as two-dimensional membrane materials for gas and liquid separations. Journal of Membrane Science, 2018,567:89-103. |
| [82] | KIM T W, SAHIMI M, TSOTSIS T T. Preparation of hydrotalcite thin films using an electrophoretic technique. Industrial & Engineering Chemistry Research, 2008,47(23):9127-9132. |
| [83] | KIM T W, SAHIMI M, TSOTSIS T T. The preparation and characterization of hydrotalcite thin films. Industrial & Engineering Chemistry Research, 2009,48(12):5794-5801. |
| [84] |
LIU Y, WANG N, CAO Z, et al. Molecular sieving through interlayer galleries. Journal of Materials Chemistry A, 2014,2(5):1235-1238.
DOI URL |
| [85] |
WANG Y, LOW Z X, KIM S, et al. Functionalized boron nitride nanosheets: a thermally rearranged polymer nanocomposite membrane for hydrogen separation. Angewandte Chemie International Edition, 2018,57(49):16056-16061.
URL PMID |
| [86] |
KIM S, HOU J, WANG Y, et al. Highly permeable thermally rearranged polymer composite membranes with a graphene oxide scaffold for gas separation. Journal of Materials Chemistry A, 2018,6(17):7668-7674.
DOI URL |
| [87] |
ZHANG X, HE Y, LI R, et al. 2D mica crystal as electret in organic field-effect transistors for multistate memory. Advanced Materials, 2016,28(19):3755-3760.
DOI URL PMID |
| [88] |
WANG D, YUAN G, HAO G, et al. All-inorganic flexible piezoelectric energy harvester enabled by two-dimensional mica. Nano Energy, 2018,43:351-358.
DOI URL |
| [89] |
YING W, HAN B, LIN H, et al. Laminated mica nanosheets supported ionic liquid membrane for CO2 separation. Nanotechnology, 2019,30(38):385705.
DOI URL PMID |
| [90] | ZHANG H P, HU W, DU A, et al. Doped phosphorene for hydrogen capture: a DFT study. Applied Surface Science, 2018,433:249-255. |
| [91] | ZHANG H P, DU A, SHI Q B, et al. Adsorption behavior of CO2 on pristine and doped phosphorenes: a dispersion corrected DFT study. Journal of CO2 Utilization, 2018,24:463-470. |
| [1] | 朱文杰, 唐璐, 陆继长, 刘江平, 罗永明. 钙钛矿型氧化物催化氧化挥发性有机化合物的研究进展[J]. 无机材料学报, 2025, 40(7): 735-746. |
| [2] | 胡智超, 杨鸿宇, 杨鸿程, 孙成礼, 杨俊, 李恩竹. P-V-L键理论在微波介质陶瓷性能调控中的应用[J]. 无机材料学报, 2025, 40(6): 609-626. |
| [3] | 董晨雨, 郑维杰, 马一帆, 郑春艳, 温峥. 压电力显微镜表征Pb(Mg,Nb)O3-PbTiO3超薄膜弛豫特性[J]. 无机材料学报, 2025, 40(6): 675-682. |
| [4] | 吴琼, 沈炳林, 张茂华, 姚方周, 邢志鹏, 王轲. 铅基织构压电陶瓷研究进展[J]. 无机材料学报, 2025, 40(6): 563-574. |
| [5] | 张碧辉, 刘小强, 陈湘明. Ruddlesden-Popper结构杂化非常规铁电体的研究进展[J]. 无机材料学报, 2025, 40(6): 587-608. |
| [6] | 吴杰, 杨帅, 王明文, 李景雷, 李纯纯, 李飞. 铅基织构压电陶瓷的发展历程、现状与挑战[J]. 无机材料学报, 2025, 40(6): 575-586. |
| [7] | 姜昆, 李乐天, 郑木鹏, 胡永明, 潘勤学, 吴超峰, 王轲. PZT陶瓷的低温烧结研究进展[J]. 无机材料学报, 2025, 40(6): 627-638. |
| [8] | 田睿智, 兰正义, 殷杰, 郝南京, 陈航榕, 马明. 基于微流控技术的纳米无机生物材料制备: 原理及其研究进展[J]. 无机材料学报, 2025, 40(4): 337-347. |
| [9] | 张继国, 吴田, 赵旭, 杨钒, 夏天, 孙士恩. 钠离子电池正极材料循环稳定性提升策略及产业化进程[J]. 无机材料学报, 2025, 40(4): 348-362. |
| [10] | 殷杰, 耿佳毅, 王康龙, 陈忠明, 刘学建, 黄政仁. SiC陶瓷的3D打印成形与致密化新进展[J]. 无机材料学报, 2025, 40(3): 245-255. |
| [11] | 谌广昌, 段小明, 朱金荣, 龚情, 蔡德龙, 李宇航, 杨东雷, 陈彪, 李新民, 邓旭东, 余瑾, 刘博雅, 何培刚, 贾德昌, 周玉. 直升机特定结构先进陶瓷材料研究进展与应用展望[J]. 无机材料学报, 2025, 40(3): 225-244. |
| [12] | 王悦, 王欣, 于显利. 室温铁磁性还原氧化石墨烯基全碳膜[J]. 无机材料学报, 2025, 40(3): 305-313. |
| [13] | 范晓波, 祖梅, 杨向飞, 宋策, 陈晨, 王子, 罗文华, 程海峰. 质子调控型电化学离子突触研究进展[J]. 无机材料学报, 2025, 40(3): 256-270. |
| [14] | 海热古·吐逊, 郭乐, 丁嘉仪, 周嘉琪, 张学良, 努尔尼沙·阿力甫. 上转换荧光探针辅助的光学成像技术在肿瘤显影中的应用研究进展[J]. 无机材料学报, 2025, 40(2): 145-158. |
| [15] | 孙树娟, 郑南南, 潘昊坤, 马猛, 陈俊, 黄秀兵. 单原子催化剂制备方法的研究进展[J]. 无机材料学报, 2025, 40(2): 113-127. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||