无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 527-535.DOI: 10.15541/jim20250165 CSTR: 32189.14.10.15541/jim20250165
王浩宇1,2( ), 柯学1,2, 关世伟1,2, 钱仕1,2(
), 柯学1,2, 关世伟1,2, 钱仕1,2( ), 刘宣勇1,2
), 刘宣勇1,2
收稿日期:2025-04-19
修回日期:2025-05-11
出版日期:2026-04-20
网络出版日期:2025-06-05
通讯作者:
钱 仕, 研究员. E-mail: qianshi@mail.sic.ac.cn作者简介:王浩宇(2000-), 男, 硕士研究生. E-mail: wanghaoyu222@mails.ucas.ac.cn
WANG Haoyu1,2( ), KE Xue1,2, GUAN Shiwei1,2, QIAN Shi1,2(
), KE Xue1,2, GUAN Shiwei1,2, QIAN Shi1,2( ), LIU Xuanyong1,2
), LIU Xuanyong1,2
Received:2025-04-19
Revised:2025-05-11
Published:2026-04-20
Online:2025-06-05
Contact:
QIAN Shi, professor. E-mail: qianshi@mail.sic.ac.cnAbout author:WANG Haoyu (2000-), male, Master candidate. E-mail: wanghaoyu222@mails.ucas.ac.cn
Supported by:摘要:
牙周炎是临床极富挑战的问题, 由口腔致病菌过度生长失衡引起, 可导致牙周组织的不可逆损伤。与清创术相比, 抗菌声动力疗法(aSDT)具有优异的生物相容性和组织穿透能力, 是一种有前景的替代方案。本工作制备了铁掺杂的钛基金属有机框架(Fe/Ti-MOFs), 研究了铁掺杂与声催化效率间的量效关系。引入Fe能够调节MOFs的能带结构和结晶度, 且未改变其晶格结构。结果表明, Fe/Ti-MOFs在超声作用下表现出增强的羟基自由基(•OH)产生能力, 从而催化杀灭细菌病原体。其中, Fe/Ti摩尔比为0.025 : 1的样品表现出较佳的声催化性能, 且受带隙工程、铁掺杂浓度和结晶度的共同调节。此外, 牙龈成纤维细胞增殖和活死染色结果表明, Fe/Ti-MOFs具有优异的生物相容性。超声响应的Fe/Ti-MOFs材料为牙周炎的aSDT提供了新的见解和策略, 有望避免抗生素的不良反应。
中图分类号:
王浩宇, 柯学, 关世伟, 钱仕, 刘宣勇. Fe掺杂Ti-MOFs用于牙周炎抗菌声动力治疗[J]. 无机材料学报, 2026, 41(4): 527-535.
WANG Haoyu, KE Xue, GUAN Shiwei, QIAN Shi, LIU Xuanyong. Fe Doped Ti-MOFs for Enhanced Antibacterial Sonodynamic Therapy of Periodontitis[J]. Journal of Inorganic Materials, 2026, 41(4): 527-535.
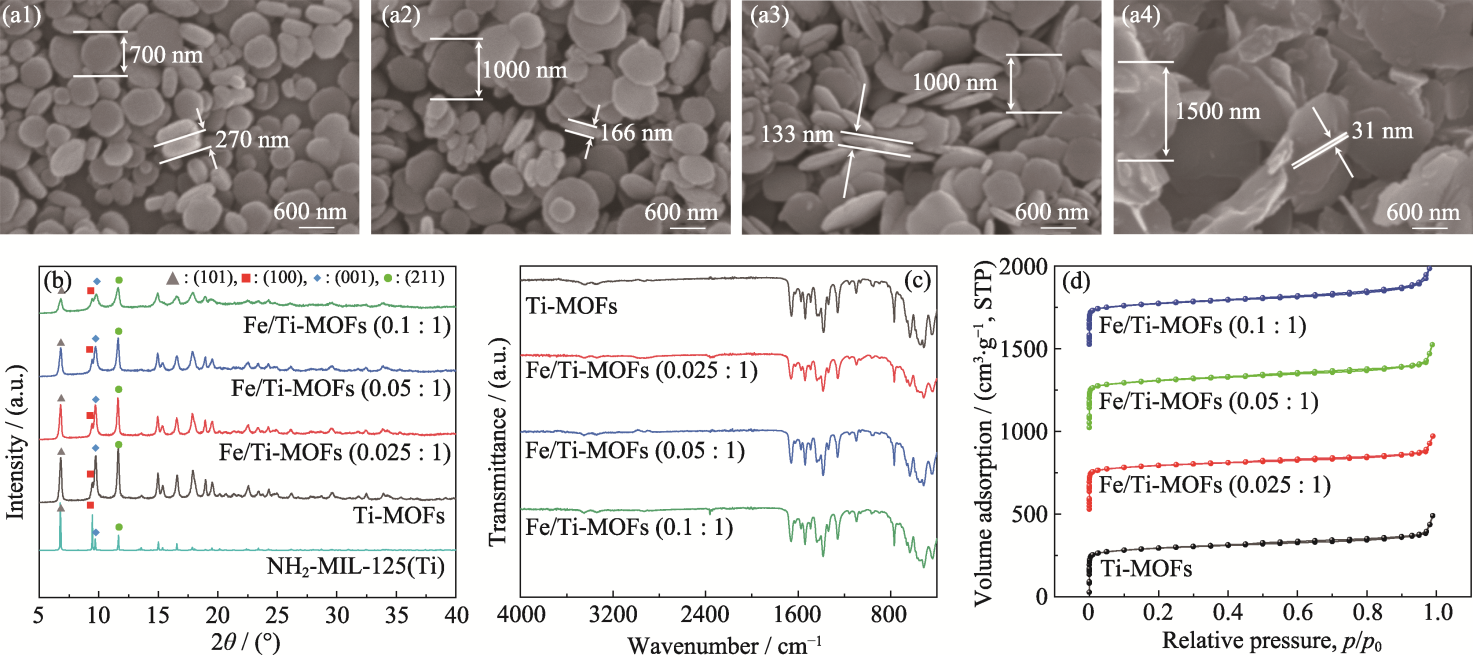
Fig. 1 SEM images of Ti-MOFs (a1), Fe/Ti-MOFs (0.025 : 1) (a2), Fe/Ti-MOFs (0.05 : 1) (a3), and Fe/Ti-MOFs (0.1 : 1) (a4); XRD patterns (b), FT-IR spectra (c), and N2 adsorption desorption curves (d) of samples
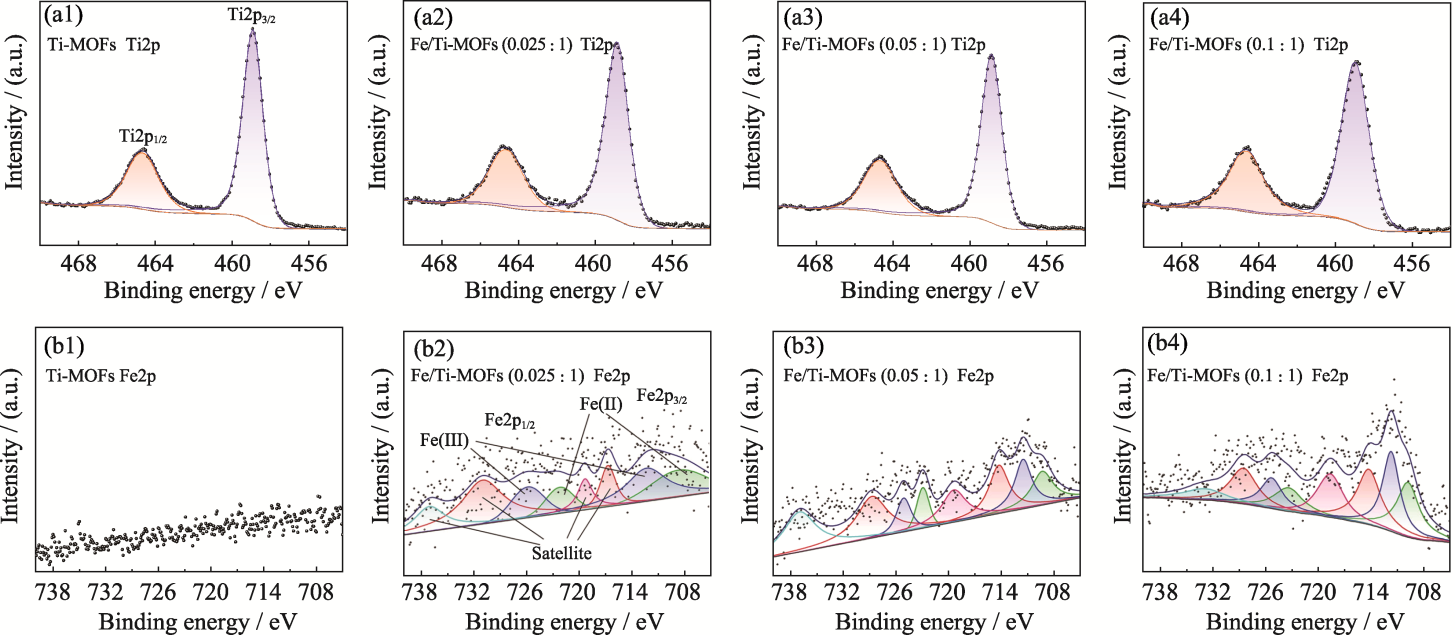
Fig. 2 High resolution XPS spectra of Ti2p (a1-a4) and Fe2p (b1-b4) of Ti-MOFs, Fe/Ti-MOFs (0.025 : 1), Fe/Ti-MOFs (0.05 : 1), and Fe/Ti-MOFs (0.1 : 1)
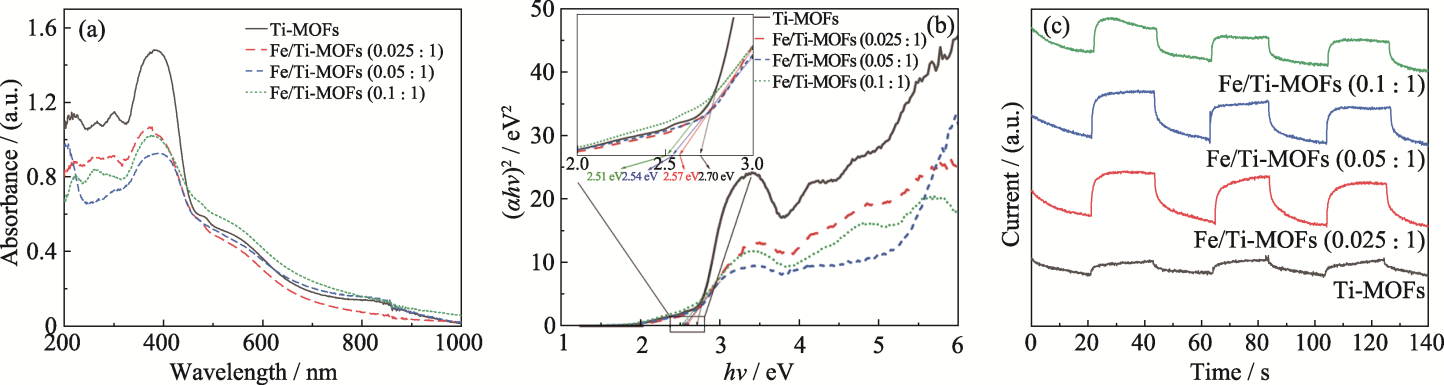
Fig. 3 UV-Vis spectra (a), bandgap fitting (b), and photoelectric response (c) of Ti-MOFs, Fe/Ti-MOFs (0.025 : 1), Fe/Ti-MOFs (0.05 : 1), and Fe/Ti-MOFs (0.1 : 1)
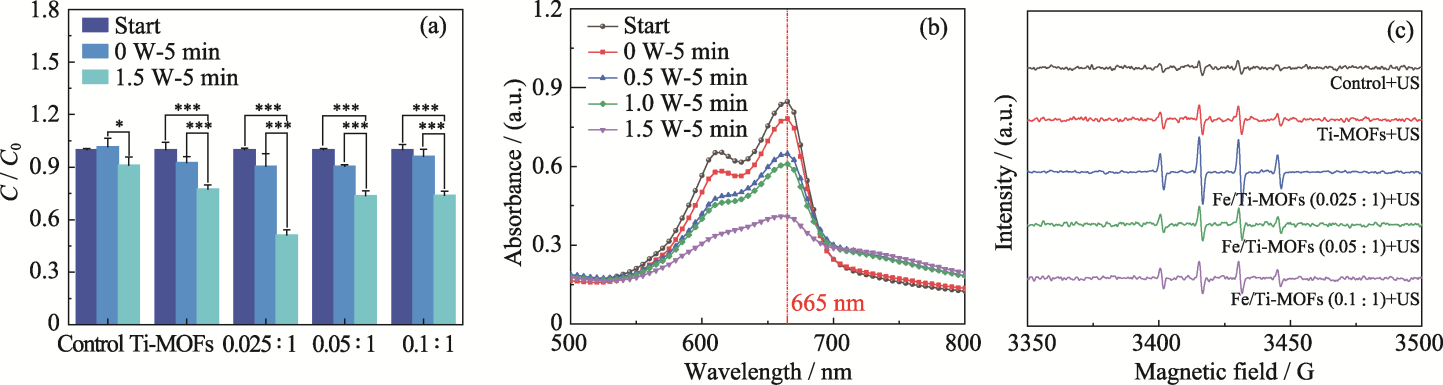
Fig. 4 MB degradation efficiencies of samples (a), absorption curves of Fe/Ti-MOFs (0.025 : 1) before and after ultrasound at different powers (b), and ESR spectra of samples under ultrasound (c) Colorful figures are available on website
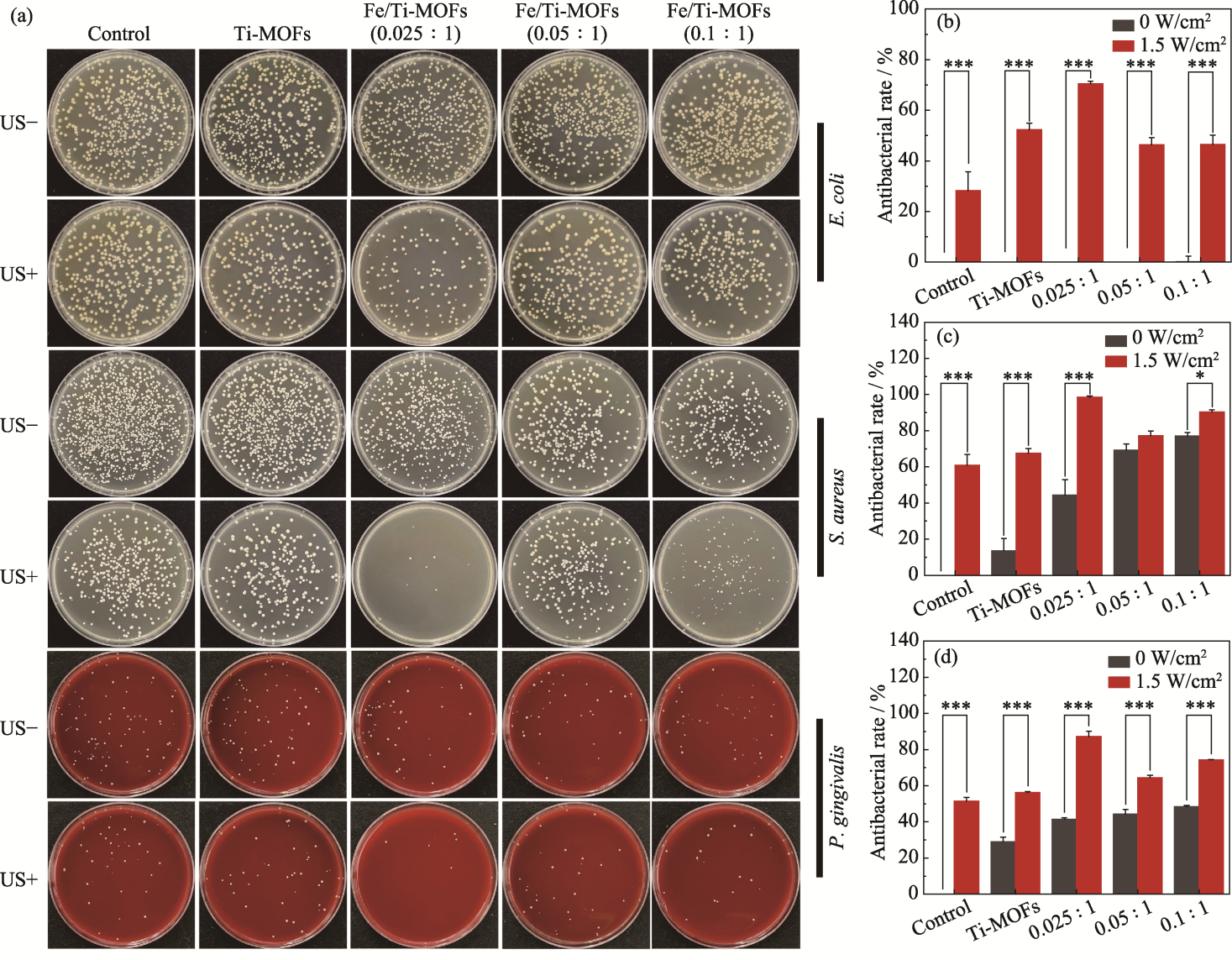
Fig. 5 Photos of re-cultivated bacterial colonies (a) and antibacterial rates against E. coli (b), S. aureus (c) and P. gingivalis (d) of Ti-MOFs, Fe/Ti-MOFs (0.025 : 1), Fe/Ti-MOFs (0.05 : 1), and Fe/Ti-MOFs (0.1 : 1) before and after ultrasonic treatment US+ and US- represent under ultrasonic and non-ultrasonic conditions, respectively. Colorful figures are available on website
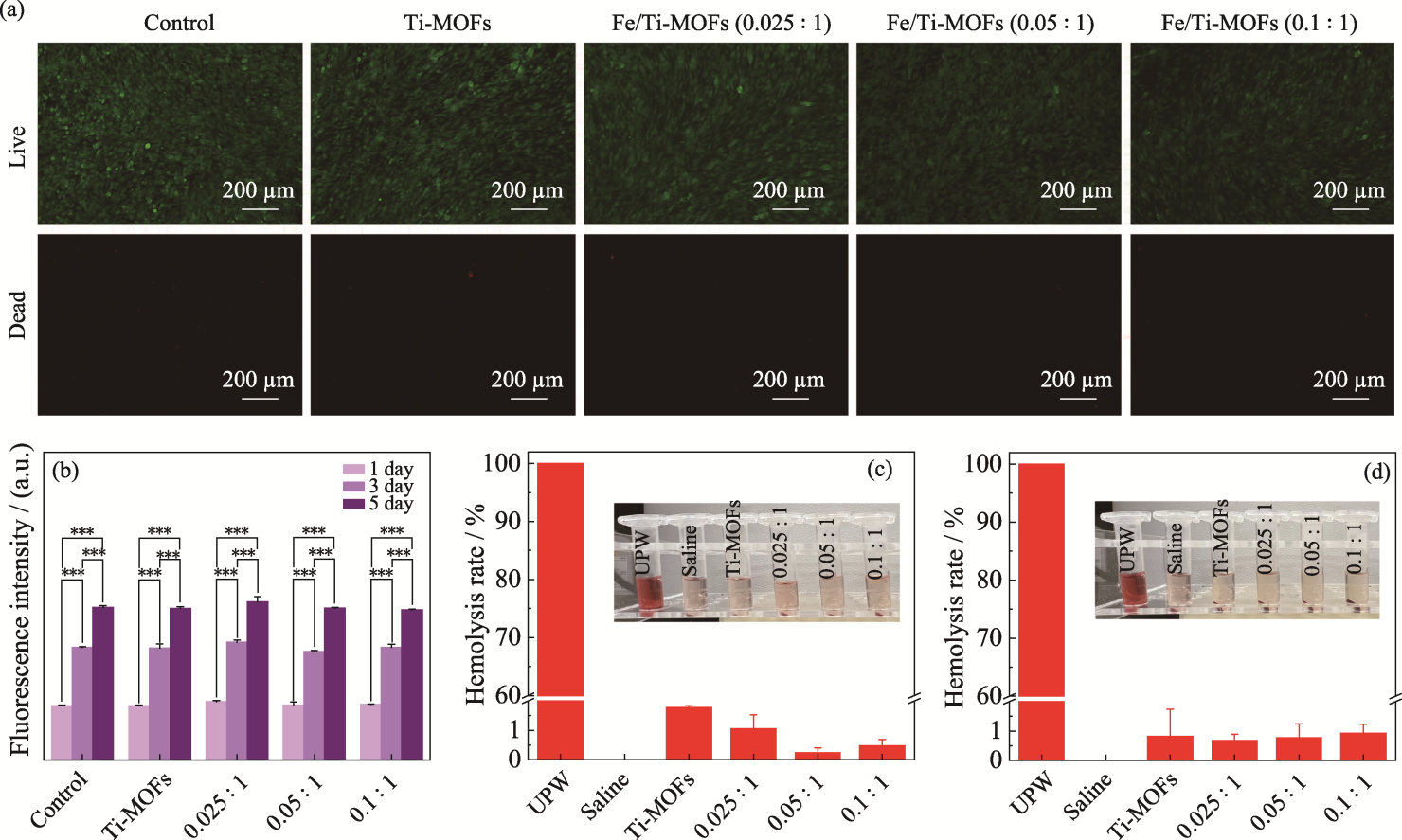
Fig. 6 Live-dead staining images (a), cell proliferation (b), and hemolysis tests (c, d) of Ti-MOFs, Fe/Ti-MOFs (0.025 : 1), Fe/Ti-MOFs (0.05 : 1), and Fe/Ti-MOFs (0.1 : 1) Colorful figures are available on website
| Sample | H2BDC-NH2/g | Fe(acac)3/mg | TTIP/mL | DMF/mL | CH3OH/mL |
|---|---|---|---|---|---|
| Ti-MOFs | 2.72 | 0 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.025 : 1) | 2.72 | 33.1 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.05 : 1) | 2.72 | 66.1 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.1 : 1) | 2.72 | 132.2 | 1.15 | 45.0 | 5.00 |
Table S1 Raw material usage in preparation process of Fe/Ti-MOFs with different Fe : Ti ratios
| Sample | H2BDC-NH2/g | Fe(acac)3/mg | TTIP/mL | DMF/mL | CH3OH/mL |
|---|---|---|---|---|---|
| Ti-MOFs | 2.72 | 0 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.025 : 1) | 2.72 | 33.1 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.05 : 1) | 2.72 | 66.1 | 1.15 | 45.0 | 5.00 |
| Fe/Ti-MOFs (0.1 : 1) | 2.72 | 132.2 | 1.15 | 45.0 | 5.00 |
| Sample | Ti-MOFs | Fe/Ti-MOFs (0.025 : 1) | Fe/Ti-MOFs (0.05 : 1) | Fe/Ti-MOFs (0.1 : 1) | |
|---|---|---|---|---|---|
| (101) | Intensity | 1858 | 1405 | 1123 | 546 |
| FWHM/° | 0.196 | 0.192 | 0.238 | 0.317 | |
| (100) | Intensity | 483 | 386 | 382 | 205 |
| FWHM/° | 0.160 | 0.147 | 0.164 | 0.174 | |
| (001) | Intensity | 1885 | 1340 | 1093 | 631 |
| FWHM/° | 0.208 | 0.215 | 0.303 | 0.543 | |
| (211) | Intensity | 2171 | 1612 | 1427 | 865 |
| FWHM/° | 0.180 | 0.170 | 0.207 | 0.362 | |
Table S2 Diffraction peak parameters of different crystal planes of Ti-MOFs and different Fe/Ti-MOFs materials
| Sample | Ti-MOFs | Fe/Ti-MOFs (0.025 : 1) | Fe/Ti-MOFs (0.05 : 1) | Fe/Ti-MOFs (0.1 : 1) | |
|---|---|---|---|---|---|
| (101) | Intensity | 1858 | 1405 | 1123 | 546 |
| FWHM/° | 0.196 | 0.192 | 0.238 | 0.317 | |
| (100) | Intensity | 483 | 386 | 382 | 205 |
| FWHM/° | 0.160 | 0.147 | 0.164 | 0.174 | |
| (001) | Intensity | 1885 | 1340 | 1093 | 631 |
| FWHM/° | 0.208 | 0.215 | 0.303 | 0.543 | |
| (211) | Intensity | 2171 | 1612 | 1427 | 865 |
| FWHM/° | 0.180 | 0.170 | 0.207 | 0.362 | |
| Sample | SBET/(m2·g-1) | SDFT/(m2·g-1) | Vpore/(cm3·g-1) |
|---|---|---|---|
| Ti-MOFs | 1132.08 | 1722.72 | 0.69 |
| Fe/Ti-MOFs (0.025 : 1) | 1134.90 | 1702.66 | 0.66 |
| Fe/Ti-MOFs (0.05 : 1) | 1175.66 | 1736.29 | 0.74 |
| Fe/Ti-MOFs (0.1 : 1) | 1033.73 | 1533.40 | 0.77 |
Table S3 Specific surface area and pore volume data of Ti-MOFs and different Fe/Ti-MOFs
| Sample | SBET/(m2·g-1) | SDFT/(m2·g-1) | Vpore/(cm3·g-1) |
|---|---|---|---|
| Ti-MOFs | 1132.08 | 1722.72 | 0.69 |
| Fe/Ti-MOFs (0.025 : 1) | 1134.90 | 1702.66 | 0.66 |
| Fe/Ti-MOFs (0.05 : 1) | 1175.66 | 1736.29 | 0.74 |
| Fe/Ti-MOFs (0.1 : 1) | 1033.73 | 1533.40 | 0.77 |
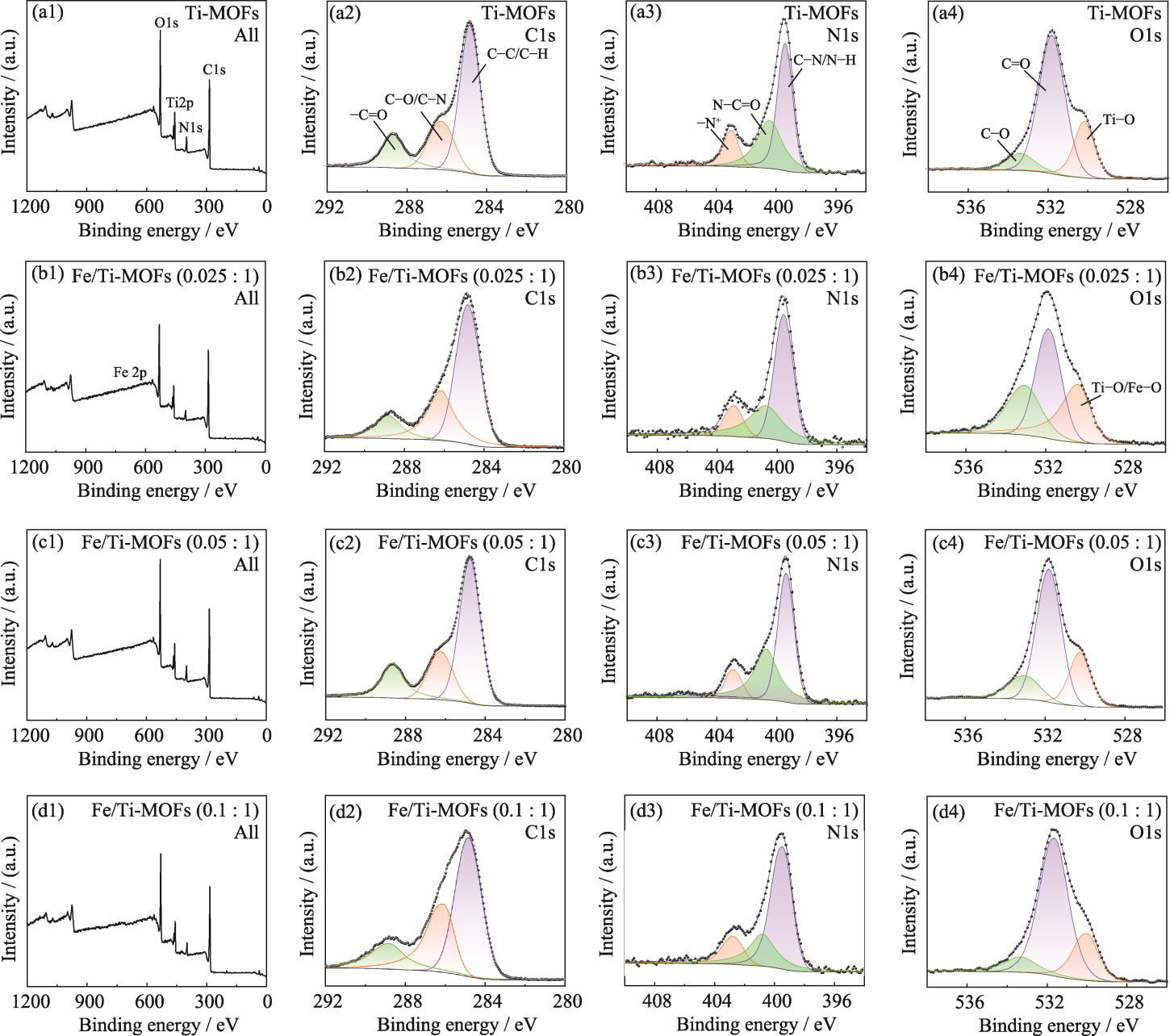
Fig. S2 Full XPS spectra (a1-d1) and high-resolution C1s (a2-d2), N1s (a3-d3), and O1s (a4-d4) XPS spectra of Ti-MOFs, Fe/Ti-MOFs (0.025 : 1), Fe/Ti-MOFs (0.05 : 1), and Fe/Ti-MOFs (0.1 : 1)
| [1] | TAN X, LIU S, HU X, et al. Near-infrared-enhanced dual enzyme- mimicking Ag-TiO2-x@alginate microspheres with antibactericidal and oxygeneration abilities to treat periodontitis. ACS Applied Materials and Interfaces, 2023, 15(1): 391. |
| [2] |
MIYAUCHI S, KAWADA-MATSUO M, FURUSHO H, et al. Atrial translocation of Porphyromonas gingivalis exacerbates atrial fibrosis and atrial fibrillation. Circulation, 2025, 151(21): 1527.
DOI URL |
| [3] |
LIU H, NAN Z, ZHAO C, et al. Emerging synergistic strategies for enhanced antibacterial sonodynamic therapy: advances and prospects. Ultrasonics Sonochemistry, 2025, 116: 107288.
DOI URL |
| [4] |
ZHANG K, WANG T, HUANG X, et al. Ultrasound-mediated nanomaterials for the treatment of inflammatory diseases. Ultrasonics Sonochemistry, 2025, 114: 107270.
DOI URL |
| [5] |
LI K, XU W, CHEN Y, et al. Piezoelectric nanostructured surface for ultrasound-driven immunoregulation to rescue titanium implant infection. Advanced Functional Materials, 2023, 33(28): 2214522.
DOI URL |
| [6] |
CHEN H, LIU L, MA A, et al. Noninvasively immunogenic sonodynamic therapy with manganese protoporphyrin liposomes against triple-negative breast cancer. Biomaterials, 2021, 269: 120639.
DOI URL |
| [7] |
PAN X, WU N, TIAN S, et al. Inhalable MOF-derived nanoparticles for sonodynamic therapy of bacterial pneumonia. Advanced Functional Materials, 2022, 32(25): 2112145.
DOI URL |
| [8] |
QIN W, YANG Q, ZHU C, et al. A Distinctive insight into inorganic sonosensitizers: design principles and applicationdo- mains. Small, 2024, 20(25): 2311228.
DOI URL |
| [9] |
LI D, YANG Y, LI D, et al. Organic sonosensitizers for sonodynamic therapy: from small molecules and nanoparticles toward clinical development. Small, 2021, 17(42): 2101976.
DOI URL |
| [10] |
YANG F, DONG J, LI Z, et al. Metal-organic frameworks (MOF)-assisted sonodynamic therapy in anticancer applications. ACS Nano, 2023, 17(5): 4102.
DOI PMID |
| [11] |
DAN-HARDI M, SERRE C, FROT T, et al. A new photoactive crystalline highly porous titanium(IV) dicarboxylate. Journal of the American Chemical Society, 2009, 131(31): 10857.
DOI PMID |
| [12] |
VERMOORTELE F, MAES M, MOGHADAM P Z, et al. p-xylene-selective metal-organic frameworks: a case of topology-directed selectivity. Journal of the American Chemical Society, 2011, 133(46): 18526.
DOI URL |
| [13] |
NGUYEN H L. Perspectives on titanium-based metal-organic frameworks. Journal of Physics: Energy, 2021, 3(2): 021003.
DOI |
| [14] |
CHEN J, CHENG F, LUO D, et al. Recent advances in Ti-based MOFs in biomedical applications. Dalton Transactions, 2022, 51(39): 14817.
DOI URL |
| [15] |
LIANG S, XIAO X, BAI L, et al. Conferring Ti-based MOFs with defects for enhanced sonodynamic cancer therapy. Advanced Materials, 2021, 33(18): 2100333.
DOI URL |
| [16] |
WANG Q, TIAN Y, YAO M, et al. Bimetallic organic frameworks of high piezovoltage for sono-piezo dynamic therapy. Advanced Materials, 2023, 35(41): 2301784.
DOI URL |
| [17] |
LI Y, REN Z, HE Z, et al. Crystallinity-defect matching relationship of g-C3N4: experimental and theoretical perspectives. Green Energy and Environment, 2024, 9(4): 623.
DOI URL |
| [18] |
JIN Y, LIU F, LI Y, et al. Efficient adsorption of azo anionic dye Congo Red by micro-nano metal-organic framework MIL-68(Fe) and MIL-68(Fe) /chitosan composite sponge: preparation, characterization and adsorption performance. International Journal of Biological Macromolecules, 2023, 252: 126198.
DOI URL |
| [19] |
WANG Y, FENG W, LI J, et al. A novel route for the facile synthesis of NH2-MIL-53(Fe) and its highly efficient and selective adsorption of Congo Red. Inorganica Chimica Acta, 2023, 547: 121332.
DOI URL |
| [20] |
OLCZAK T, SIMPSON W, LIU X, et al. Iron and heme utilization in Porphyromonas gingivalis. FEMS Microbiology Reviews, 2005, 29(1): 119.
DOI URL |
| [21] | WÓJTOWICZ H, GUEVARA T, TALLANT C, et al. Unique structure and stability of HmuY, a novel heme-binding protein of Porphyromonas gingivalis. PLOS Pathogens, 2009, 5(5): e1000419. |
| [22] |
WANG X, ZHONG X, BAI L, et al. Ultrafine titanium monoxide (TiO1+x) nanorods for enhanced sonodynamic therapy. Journal of the American Chemical Society, 2020, 142(14): 6527.
DOI URL |
| [23] |
ZHANG D Y, LIU H, YOUNIS M R, et al. In-situ TiO2-x decoration of titanium carbide MXene for photo/sono-responsive antitumor theranostics. Journal of Nanobiotechnology, 2022, 20(1): 53.
DOI |
| [24] |
GUAN S, XU W, TAN J, et al. Metainterface heterostructure enhances sonodynamic therapy for disrupting secondary biofilms. ACS Nano, 2024, 18(23): 15114.
DOI PMID |
| [1] | 李璇, 叶奎材, 冯佳音, 邱家军, 钱文昊, 邢敏. 钛基牙种植体表面改性促进软组织封闭的研究进展[J]. 无机材料学报, 2026, 41(4): 432-444. |
| [2] | 朱开煌, 杨世杰, 李欣格, 宋贯卿, 史淦升, 王焱, 任小孟, 陆遥, 徐新宏, 孙静. 基于UiO-66骨架的氧化石墨烯改性金属有机框架凝胶的制备及其对甲苯的高效吸附性能[J]. 无机材料学报, 2026, 41(4): 519-526. |
| [3] | 聂晓双, 李丹丹, 王芳, 欧阳丽萍, 李恒, 邱家军. Ti3C2Tx压电复合水凝胶用于感染创面修复研究[J]. 无机材料学报, 2026, 41(2): 234-244. |
| [4] | 江宗玉, 黄红花, 清江, 王红宁, 姚超, 陈若愚. 铝离子掺杂MIL-101(Cr)的制备及其VOCs吸附性能研究[J]. 无机材料学报, 2025, 40(7): 747-753. |
| [5] | 赵丽华, 王言帅, 尹昕妩, 毛叶琼, 牛德超. 负载硫化铋纳米簇的硅基杂化胶束的制备及其光热抗菌性能[J]. 无机材料学报, 2025, 40(10): 1129-1136. |
| [6] | 张博, 付一敏, 陈政, 石澳, 朱敏. 近红外光响应的双相抗菌介孔生物活性玻璃复合支架的制备及抗菌性能研究[J]. 无机材料学报, 2025, 40(10): 1137-1144. |
| [7] | 王月月, 黄佳慧, 孔红星, 李怀珠, 姚晓红. 载银放射状介孔二氧化硅的制备及其在牙科树脂中的应用[J]. 无机材料学报, 2025, 40(1): 77-83. |
| [8] | 瞿牡静, 张淑兰, 朱梦梦, 丁浩杰, 段嘉欣, 代恒龙, 周国红, 李会利. CsPbBr3@MIL-53纳米复合荧光粉的合成、性能及其白光LEDs应用[J]. 无机材料学报, 2024, 39(9): 1035-1043. |
| [9] | 李承瑜, 丁自友, 韩颖超. 锰掺杂纳米羟基磷灰石的体外抗菌-促成骨性能研究[J]. 无机材料学报, 2024, 39(3): 313-320. |
| [10] | 张志民, 葛敏, 林翰, 施剑林. 新型磁电催化纳米粒子的活性氮释放与抗菌性能研究[J]. 无机材料学报, 2024, 39(10): 1114-1124. |
| [11] | 张淑敏, 奚晓雯, 孙磊, 孙平, 王德强, 魏杰. 基于声动力和类酶活性的铌基涂层: 抗菌及促进细胞增殖与分化[J]. 无机材料学报, 2024, 39(10): 1125-1134. |
| [12] | 谢家晔, 李力文, 朱强. 三种临床盖髓剂的抗菌性及生物相容性对比研究[J]. 无机材料学报, 2023, 38(12): 1449-1456. |
| [13] | 杜佳恒, 范鑫丽, 肖东琴, 尹一然, 李忠, 贺葵, 段可. 电泳沉积制备微弧氧化钛表面氧化镁涂层及其生物学性能[J]. 无机材料学报, 2023, 38(12): 1441-1448. |
| [14] | 吴雪彤, 张若飞, 阎锡蕴, 范克龙. 纳米酶: 一种抗微生物感染新方法[J]. 无机材料学报, 2023, 38(1): 43-54. |
| [15] | 盛丽丽, 常江. 光/磁热Fe2SiO4/Fe3O4双相生物陶瓷及其复合电纺丝膜制备及抗菌性能研究[J]. 无机材料学报, 2022, 37(9): 983-990. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||