无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 432-444.DOI: 10.15541/jim20250144 CSTR: 32189.14.10.15541/jim20250144
李璇1,2( ), 叶奎材1, 冯佳音1, 邱家军3, 钱文昊1(
), 叶奎材1, 冯佳音1, 邱家军3, 钱文昊1( ), 邢敏1(
), 邢敏1( )
)
收稿日期:2025-04-07
修回日期:2025-06-05
出版日期:2026-04-20
网络出版日期:2025-06-27
通讯作者:
邢 敏, 副研究员. E-mail: xingmin0821@126.com;作者简介:李 璇(2000-), 女, 硕士研究生. E-mail: 2233092@mail.dhu.edu.cn
基金资助:
LI Xuan1,2( ), YE Kuicai1, FENG Jiayin1, QIU Jiajun3, QIAN Wenhao1(
), YE Kuicai1, FENG Jiayin1, QIU Jiajun3, QIAN Wenhao1( ), XING Min1(
), XING Min1( )
)
Received:2025-04-07
Revised:2025-06-05
Published:2026-04-20
Online:2025-06-27
Contact:
XING Min, associate professor. E-mail: xingmin0821@126.com;About author:LI Xuan (2000-), female, Master candidate. E-mail: 2233092@mail.dhu.edu.cn
Supported by:摘要:
钛及钛合金因其优异的力学性能、耐腐蚀性和生物相容性, 被广泛用作牙种植体材料。然而, 钛基牙种植体在临床使用中软组织封闭不佳, 易使细菌侵入诱发种植体周炎, 导致种植体植入手术失败。因此, 为提升钛基牙种植体的软组织封闭性能, 降低种植体植入手术的失败率, 国内外学者开展了持续、深入的研究。本文综述了近年来钛基牙种植体软组织封闭表面改性研究系列进展, 着重介绍了钛表面化学组分调控和微纳结构构建方法, 指出了当前研究面临的挑战以及未来发展趋势, 以期为该领域的进一步研究提供参考。
中图分类号:
李璇, 叶奎材, 冯佳音, 邱家军, 钱文昊, 邢敏. 钛基牙种植体表面改性促进软组织封闭的研究进展[J]. 无机材料学报, 2026, 41(4): 432-444.
LI Xuan, YE Kuicai, FENG Jiayin, QIU Jiajun, QIAN Wenhao, XING Min. Surface Modification of Titanium-based Dental Implants for Soft Tissue Sealing: A Review[J]. Journal of Inorganic Materials, 2026, 41(4): 432-444.
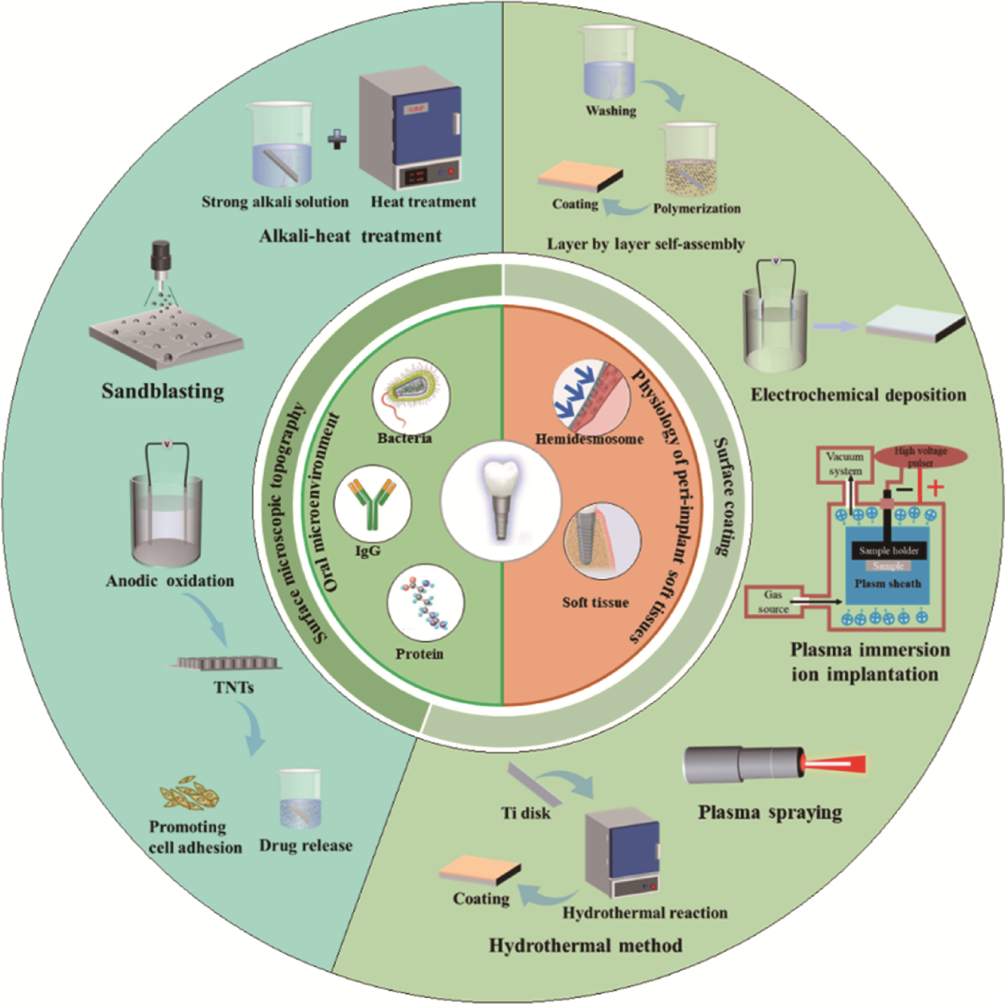
图1 钛基牙种植体口腔微环境、软组织生理学及各种表面改性方法[7]
Fig. 1 Oral microenvironment and soft tissue around titanium-based dental implants along with various surface modification techniques[7]
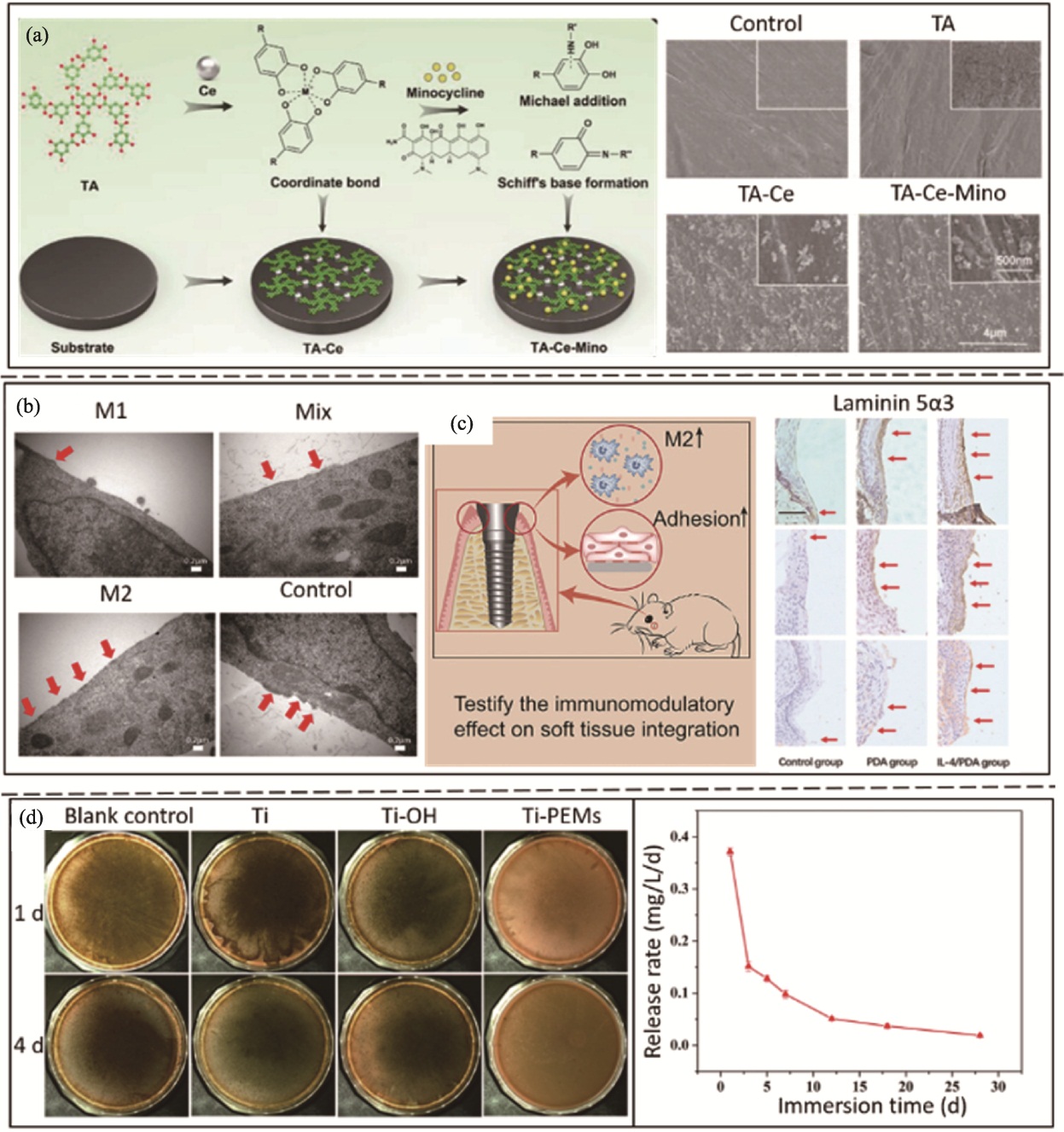
图3 采用层层自组装法的软组织封闭及抗菌效果[27-28,30]
Fig. 3 Soft tissue sealing and antibacterial effects promoted by layer-by-layer self-assembly method[27-28,30] (a) Schemetic diagram of TA-Ce-Mino coating design and SEM images showing topography of the coating[27]; (b) Hemidesmosome detected by TEM[28]; (c) Immunohistochemistry staining results of expression of the hemidesmosome related protein laminin 5α3[28]; (d) Antibacterial efficacy of Ti-PEMs samples against P. gingivalis[30]

图4 电化学沉积法制备钛基涂层示意图及该涂层对细胞迁移的作用[34]
Fig. 4 Schematic diagram of titanium-based coating prepared by electrochemical deposition and its effect on promoting cell migration[34] (a) Electrochemical deposition model for modification of Col-Ι on Ti[34]; (b1-b3) SEM images of L929 fibroblast cell reseeding on (b1) untreated Ti, (b2) TiO2 porous surface and (b3) coated Ti/Col-Ι (scale bar: 10 and 2 μm, respectively)[34]
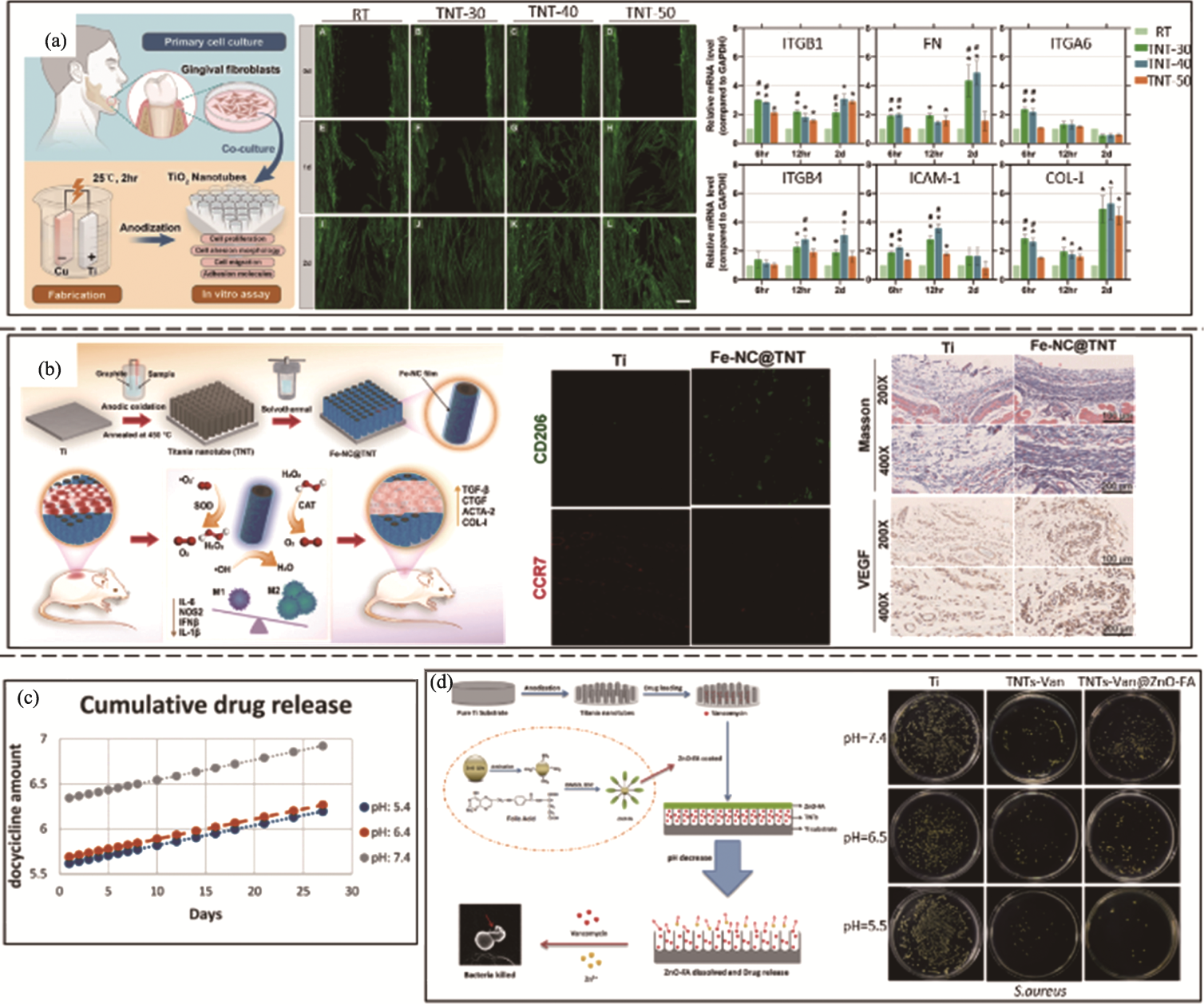
图6 采用阳极氧化法的软组织封闭及抗菌效果[75-76,78 -79]
Fig. 6 Soft tissue sealing and antibacterial effects promoted by anodic oxidation method [75-76,78 -79] (a) Soft tissue sealing efficacy promoted by TNT-30, TNT-40 and TNT-50 (scale bar: 100 μm)[75]; (b) Soft tissue sealing efficacy promoted by Fe-NC@TNT[76]; (c) Thirty-day cumulative average quantity of doxycycline released under pH 5.4, 6.4 and 7.4[78]; (d) Antibacterial activity of TNTs-Van@ZnO-FA under different pH against S. aureus[79]
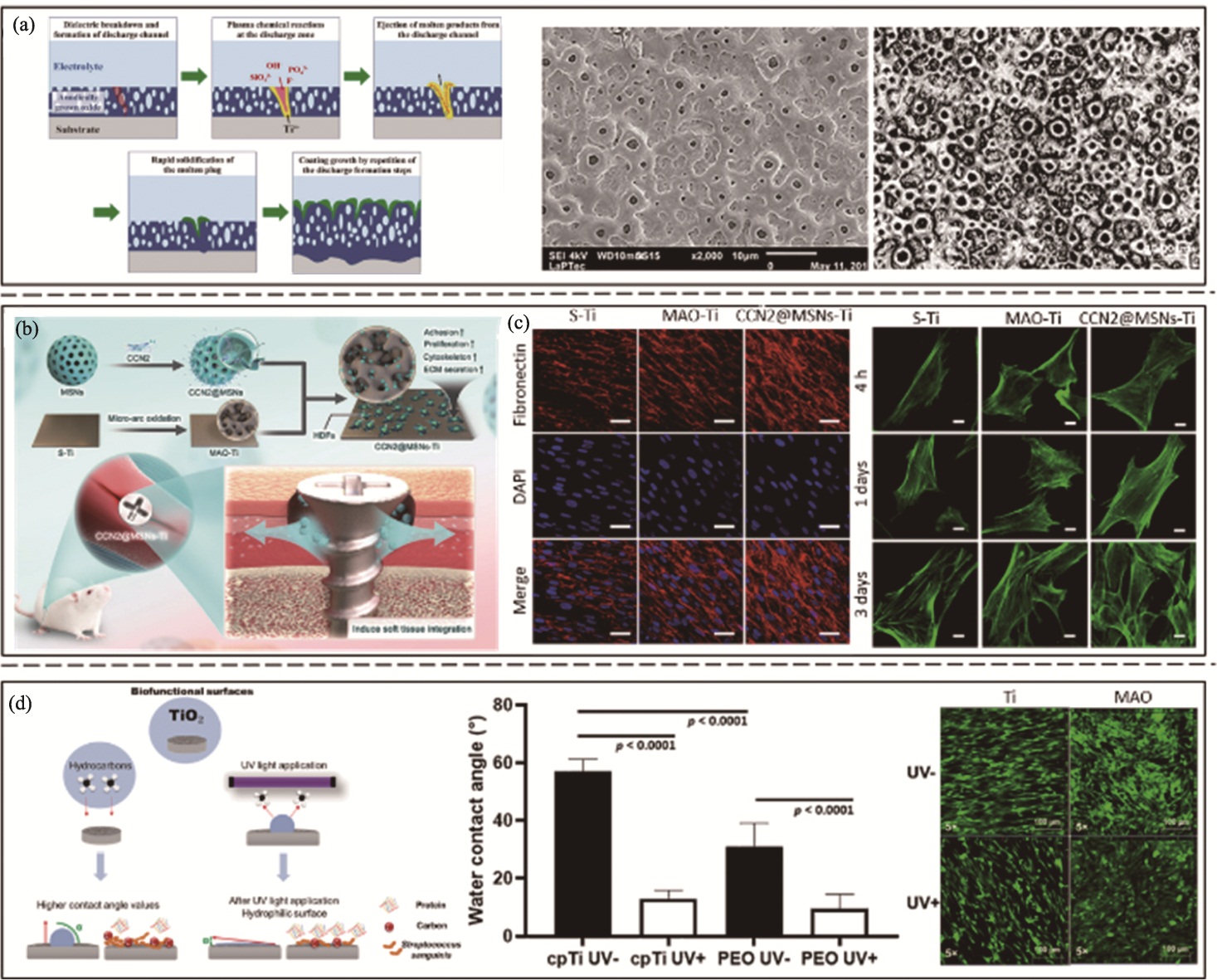
图7 微弧氧化法制备钛基涂层示意图及其软组织封闭效果[82-84]
Fig. 7 Titanium-based coating prepared by micro-arc oxidation and its effect on promoting soft tissue sealing[82-84] (a) Schematic illustration of titanium-based coating fabrication via micro-arc oxidation and characteristic surface morphology[82-83]; (b) Schematic diagram of CCN2@MSNs-Ti prepared by micro-arc oxidation[84]; (c) Effects of S-Ti, MAO-Ti, and CCN2@MSNs-Ti on HDFs migration (scale bar: 50 (left) and 10 (right) μm, respectively)[84]; (d) Fluorescence staining of HGFs for incubation on Ti and MAO treated surfaces without UV treatment (UV−) and with UV treatment (UV+)[82]
| [1] | AGARWAL S, MISTRY L, MISTRY S, et al. The per-ingvar Brånemark era (1929-2014): evolution of a no compromise prosthetic dental replacement. Cureus, 2024, 16(10): e71708. |
| [2] |
SAFAEI M, MOHAMMADI H, BEDDU S, et al. Surface topography steer soft tissue response and antibacterial function at the transmucosal region of titanium implant. International Journal of Nanomedicine, 2024, 19: 4835.
DOI PMID |
| [3] |
WU X Q, LI L Q, TAO W, et al. Built-up sodium alginate/ chlorhexidine multilayer coating on dental implants with initiating anti-infection and cyto-compatibility sequentially for soft-tissue sealing. Biomaterials Advances, 2023, 151: 213491.
DOI URL |
| [4] | SHRIVAS S, SAMAUR H, YADAV V, et al. Soft and hard tissue integration around percutaneous bone-anchored titanium prostheses: toward achieving holistic biointegration. ACS Biomaterials Science & Engineering, 2024, 10(4): 1966. |
| [5] |
YU C, YU Y, LU Y, et al. UiO-66/AgNPs coating for dental implants in preventing bacterial infections. Journal of Dental Research, 2024, 103(5): 516.
DOI PMID |
| [6] |
ABDALLAH M N, BADRAN Z, CIOBANU O, et al. Strategies for optimizing the soft tissue seal around osseointegrated implants. Advanced Healthcare Materials, 2017, 6(20): 1700549.
DOI URL |
| [7] | SUN X D, LIU T T, WANG Q Q, et al. Surface modification and functionalities for titanium dental implants. ACS Biomaterials Science & Engineering, 2023, 9(8): 4442. |
| [8] |
BERNABE E, MARCENES W, HERNANDEZ C R, et al. Global, regional, and national levels and trends in burden of oral conditions from 1990 to 2017: a systematic analysis for the global burden of disease 2017 study. Journal of Dental Research, 2020, 99(4): 362.
DOI PMID |
| [9] |
SOUZA J G S, BERTOLINI M M, COSTA R C, et al. Targeting implant-associated infections: titanium surface loaded with antimicrobial. iScience, 2021, 24(1): 102008.
DOI URL |
| [10] |
JIA B, ZHANG B B, LI J H, et al. Emerging polymeric materials for treatment of oral diseases: design strategy towards a unique oral environment. Chemical Society Reviews, 2024, 53(7): 3273.
DOI URL |
| [11] | MOUNTCASTLE S E, COX S C, SAMMONS R L, et al. A review of co-culture models to study the oral microenvironment and disease. Journal of Oral Microbiology, 2020, 12(1): 17732. |
| [12] | NAYAK S C, LATHA P B, KANDANATTU B, et al. The oral microbiome and systemic health: bridging the gap between dentistry and medicine. Cureus, 2025, 17(2): e78918. |
| [13] |
CUI Z W, WANG P, GAO W Y. Microbial dysbiosis in periodontitis and peri-implantitis: pathogenesis, immune responses, and therapeutic. Frontiers in Cellular and Infection Microbiology, 2025, 15: 1517154.
DOI URL |
| [14] |
INCHINGOLO F, INCHINGOLO A M, MALCANGI G, et al. The benefits of probiotics on oral health: systematic review of the literature. Pharmaceuticals, 2023, 16(9): 1313.
DOI URL |
| [15] | WELCH J L M, RAMÍREZ-PUEBLA S T, BORISY G G. Oral microbiome geography: micron-scale habitat and niche. Cell Host & Microbe, 2020, 28(2): 160. |
| [16] |
GAO H, JIANG N, NIU Q N, et al. Biocompatible nanostructured silver-incorporated implant surfaces show effective antibacterial, osteogenic, and anti-inflammatory effects in vitro and in rat model. International Journal of Nanomedicine, 2023, 18: 7359.
DOI URL |
| [17] |
MONJE A, NART J. Management and sequelae of dental implant removal. Periodontology 2000, 2022, 88(1): 182.
DOI PMID |
| [18] |
GUO T Q, GULATI K, ARORA H, et al. Race to invade: understanding soft tissue integration at the transmucosal region of titanium dental implants. Dental Materials, 2021, 37(5): 816.
DOI PMID |
| [19] |
WANG M, LIU Y B, TONG W M, et al. Periodontitis history shapes the early peri-implant microbiome formation: a metagenomic analysis. Journal of Clinical Periodontology, 2025, 52(7): 1011.
DOI PMID |
| [20] |
CINQUINI C, MARCHIO V, DI DONNA E, et al. Histologic evaluation of soft tissues around dental implant abutments: a narrative review. Materials, 2022, 15(11): 3811.
DOI URL |
| [21] | MATHUR A, KHARBANDA O P, KOUL V, et al. Fabrication and evaluation of antimicrobial biomimetic nanofiber coating for improved dental implant bioseal: an in vitro study. Journal of Periodontology, 2022, 93(10): 1578. |
| [22] |
DENG Z M, LIANG J, FANG N, et al. Integration of collagen fibers in connective tissue with dental implant in the transmucosal region. International Journal of Biological Macromolecules, 2022, 208: 833.
DOI PMID |
| [23] |
WALKO G, CASTAÑÓN M J, WICHE G. Molecular architecture and function of the hemidesmosome. Cell and Tissue Research, 2015, 360(2): 363.
DOI PMID |
| [24] |
YANG J, ZHANG Z H, YAO W H, et al. Recent developments in coatings on biodegradable Mg alloys: a review. Journal of Magnesium and Alloys, 2025, 13(4): 1405.
DOI URL |
| [25] |
SÁNCHEZ-BODÓN J, DEL OLMO J A, ALONSO J M, et al. Bioactive coatings on titanium: a review on hydroxylation, self-assembled monolayers (SAMs) and surface modification strategies. Polymers, 2021, 14(1): 165.
DOI URL |
| [26] |
QIU W Z, YANG H C, XU Z K. Dopamine-assisted co-deposition: an emerging and promising strategy for surface modification. Advances in Colloid and Interface Science, 2018, 256: 111.
DOI URL |
| [27] |
LI D Z, TAN X, ZHENG L W, et al. A dual-antioxidative coating on transmucosal component of implant to repair connective tissue barrier for treatment of peri-implantitis. Advanced Healthcare Materials, 2023, 12(30): 2301733.
DOI URL |
| [28] |
LIU R H, CHEN S C, HUANG P N, et al. Immunomodulation-based strategy for improving soft tissue and metal implant integration and its implications in the development of metal soft tissue materials. Advanced Functional Materials, 2020, 30(21): 1910672.
DOI URL |
| [29] |
XIE X Y, TANG J, XING Y X, et al. Intervention of polydopamine assembly and adhesion on nanoscale interfaces: state-of-the-art designs and biomedical applications. Advanced Healthcare Materials, 2021, 10(9): 2002138.
DOI URL |
| [30] |
LI W, YANG Y W, ZHANG H C, et al. Improvements on biological and antimicrobial properties of titanium modified by AgNPs-loaded chitosan-heparin polyelectrolyte multilayers. Journal of Materials Science: Materials in Medicine, 2019, 30(5): 52.
DOI |
| [31] | 杜佳恒, 范鑫丽, 肖东琴, 等. 钛表面电化学沉积氢氧化镁与氧化镁涂层的抗菌性能与生物活性. 硅酸盐学报, 2024, 52(5): 1570. |
| [32] |
YI J L, LI M, ZHU J X, et al. Recent development and applications of electrodeposition biocoatings on medical titanium for bone repair. Journal of Materials Chemistry B, 2024, 12(39): 9863.
DOI URL |
| [33] |
GRIGORIEV S, SOTOVA C, VERESCHAKA A, et al. Modifying coatings for medical implants made of titanium alloys. Metals, 2023, 13(4): 718.
DOI URL |
| [34] |
TRUC N T, MINH H H, KHANH L L, et al. Modification of type I collagen on TiO2 surface using electrochemical deposition. Surface and Coatings Technology, 2018, 344: 664.
DOI URL |
| [35] |
GUO T Q, SCIMECA J C, IVANOVSKI S, et al. Enhanced corrosion resistance and local therapy from nano-engineered titanium dental implants. Pharmaceutics, 2023, 15(2): 315.
DOI URL |
| [36] | MELENTIEV R, YUDHANTO A, TAO R, et al. Metallization of polymers and composites: state-of-the-art approaches. Materials & Design, 2022, 221: 110958. |
| [37] |
RATHA I, DATTA P, BALLA V K, et al. Effect of doping in hydroxyapatite as coating material on biomedical implants by plasma spraying method: a review. Ceramics International, 2021, 47(4): 4426.
DOI URL |
| [38] |
WANG Q, TANG Z L, HEROUT R, et al. Axial suspension plasma sprayed Ag-TiO2 coating for enhanced photocatalytic and antimicrobial properties. Surfaces and Interfaces, 2024, 45: 103856.
DOI URL |
| [39] |
LUO Y, GAO L P, HU J Q, et al. Mechanical properties and in vitro human gingival fibroblasts compatibility of plasma-sprayed zirconia-coated titanium alloy abutment. Materials Letters, 2022, 324: 132702.
DOI URL |
| [40] |
WANG F F, LI C J, ZHANG S, et al. Tantalum coated on titanium dioxide nanotubes by plasma spraying enhances cytocompatibility for dental implants. Surface and Coatings Technology, 2020, 382: 125161.
DOI URL |
| [41] |
夏超, 钱仕, 王东辉, 等. 碳离子注入医用Ti性能研究. 金属学报, 2017, 53(10): 1393.
DOI |
| [42] |
TIAN X, ZHANG P, XU J. Incorporating zinc ion into titanium surface promotes osteogenesis and osteointegration in implantation early phase. Journal of Materials Science: Materials in Medicine, 2023, 34(11): 55.
DOI |
| [43] |
ZHOU J J, WU Y H, TAN J, et al. Constructing hydroxylated graphite-like film with protein regulation on PEEK for integrated bone-soft tissue therapy. Advanced Functional Materials, 2025, 35(27): 2423821.
DOI URL |
| [44] |
LIN Z J, WU S L, LIU X Y, et al. A surface-engineered multifunctional TiO2 based nano-layer simultaneously elevates the corrosion resistance, osteoconductivity and antimicrobial property of a magnesium alloy. Acta Biomaterialia, 2019, 99: 495.
DOI URL |
| [45] |
ZHU Y, ZHANG C N, GU Y X, et al. The responses of human gingival fibroblasts to magnesium-doped titanium. Journal of Biomedical Materials Research Part A, 2020, 108(2): 267.
DOI PMID |
| [46] |
SHIAU D K, YANG C H, SUN Y S, et al. Enhancing the blood response and antibacterial adhesion of titanium surface through oxygen plasma immersion ion implantation treatment. Surface and Coatings Technology, 2019, 365: 173.
DOI URL |
| [47] |
OSHIRO W, AYUKAWA Y, ATSUTA I, et al. Effects of CaCl2 hydrothermal treatment of titanium implant surfaces on early epithelial sealing. Colloids and Surfaces B: Biointerfaces, 2015, 131: 141.
DOI URL |
| [48] | SHI X L, XU L L, MUNAR M L, et al. Hydrothermal treatment for TiN as abrasion resistant dental implant coating and its fibroblast response. Materials Science & Engineering: C, 2015, 49: 1. |
| [49] |
MA L, WANG C S, MA Y, et al. Achieving high strength and ductility in laser powder bed fusion-manufactured pure titanium through the addition of carbon nanotubes. Rare Metals, 2025, 44(6): 4149.
DOI URL |
| [50] |
TUAN T Q, TOAN L V, PHAM V H. Synthesis of heterostructured TiO2 nanopores/nanotubes by anodizing at high voltages. Materials, 2024, 17(13): 3347.
DOI URL |
| [51] |
NICOLAS-SILVENTE A I, VELASCO-ORTEGA E, ORTIZ- GARCIA I, et al. Influence of the titanium implant surface treatment on the surface roughness and chemical composition. Materials, 2020, 13(2): 314.
DOI URL |
| [52] |
CAO X, WU K Y, WANG C Y, et al. Graphene oxide loaded on TiO2-nanotube-modified Ti regulates the behavior of human gingival fibroblasts. International Journal of Molecular Sciences, 2022, 23(15): 8723.
DOI URL |
| [53] |
OSMAN M A, ALAMOUSH R A, KUSHNEREV E, et al. Biological response of epithelial and connective tissue cells to titanium surfaces with different ranges of roughness: an in-vitro study. Dental Materials, 2022, 38(11): 1777.
DOI URL |
| [54] |
ZHAO B R, VAN DER MEI H C, SUBBIAHDOSS G, et al. Soft tissue integration versus early biofilm formation on different dental implant materials. Dental Materials, 2014, 30(7): 716.
DOI PMID |
| [55] | MÜHL A, SZABÓ P, KRAFCSIK O, et al. Comparison of surface aspects of turned and anodized titanium dental implant, or abutment material for an optimal soft tissue integration. Heliyon, 2022, 8(8): e10263. |
| [56] |
AN N, RAUSCH-FAN X, WIELAND M, et al. Initial attachment, subsequent cell proliferation/viability and gene expression of epithelial cells related to attachment and wound healing in response to different titanium surfaces. Dental Materials, 2012, 28(12): 1207.
DOI PMID |
| [57] |
FISCHER N G, APARICIO C. Junctional epithelium and hemidesmosomes: tape and rivets for solving the “percutaneous device dilemma” in dental and other permanent implants. Bioactive Materials, 2022, 18: 178.
DOI URL |
| [58] |
LI N B, XIAO G Y, TSAI I H, et al. Transformation of the surface compositions of titanium during alkali and heat treatment at different vacuum degrees. New Journal of Chemistry, 2018, 42(14): 11991.
DOI URL |
| [59] |
LIU W J, LI W Q, WANG H R, et al. Surface modification of porous titanium and titanium alloy implants manufactured by selective laser melting: a review. Advanced Engineering Materials, 2023, 25(21): 2300765.
DOI URL |
| [60] |
HUANG Y Z, HE S K, GUO Z J, et al. Nanostructured titanium surfaces fabricated by hydrothermal method: influence of alkali conditions on the osteogenic performance of implants. Materials Science and Engineering: C, 2019, 94: 1.
DOI URL |
| [61] |
JAGGESSAR A, MATHEW A, WANG H X, et al. Mechanical, bactericidal and osteogenic behaviours of hydrothermally synthesised TiO2 nanowire arrays. Journal of the Mechanical Behavior of Biomedical Materials, 2018, 80: 311.
DOI URL |
| [62] | KATO E, SAKURAI K, YAMADA M. Periodontal-like gingival connective tissue attachment on titanium surface with nano- ordered spikes and pores created by alkali-heat treatment. Dental Materials, 2015, 31(5): e116. |
| [63] |
OSAK P, MASZYBROCKA J, ZUBKO M, et al. Influence of sandblasting process on tribological properties of titanium grade 4 in artificial saliva for dentistry applications. Materials, 2021, 14(24): 7536.
DOI URL |
| [64] |
KITYK A, HNATKO M, PAVLIK V, et al. Advancing biomedical substrate engineering: an eco-friendly route for synthesizing micro- and nanotextures on 3D printed Ti-6Al-4V. Journal of Materials Research and Technology, 2024, 28: 2098.
DOI URL |
| [65] | LIU R, TANG Y L, LIU H, et al. Effects of combined chemical design (Cu addition) and topographical modification (SLA) of Ti-Cu/SLA for promoting osteogenic, angiogenic and antibacterial activities. Journal of Materials Science & Technology, 2020, 47: 202. |
| [66] | ABDULLA M A, HASAN R H, AL-HYANI O H. Impact of Er, Cr: YSGG laser, sandblast, and acid etching surface modification on surface topography of biodental titanium implants. Journal of Lasers in Medical Sciences, 2023, 14: e38. |
| [67] |
ROY M, CORTI A, DOMINICI S, et al. Biocompatibility of subperiosteal dental implants: effects of differently treated titanium surfaces on the expression of ECM-related genes in gingival fibroblasts. Journal of Functional Biomaterials, 2023, 14(2): 59.
DOI URL |
| [68] |
RAUSCH M A, SHOKOOHI-TABRIZI H, WEHNER C, et al. Impact of implant surface material and microscale roughness on the initial attachment and proliferation of primary human gingival fibroblasts. Biology, 2021, 10(5): 356.
DOI URL |
| [69] |
JIN S Q, YU Y M, ZHANG T, et al. Surface modification strategies to reinforce the soft tissue seal at transmucosal region of dental implants. Bioactive Materials, 2024, 42: 404.
DOI PMID |
| [70] |
PAN Y, CAO L L, CHEN L B, et al. Enhanced bacterial and biofilm adhesion resistance of ALD nano-TiO2 coatings compared to AO coatings on titanium abutments. International Journal of Nanomedicine, 2024, 19: 11143.
DOI URL |
| [71] | GUO T Q, IVANOVSKI S, GULATI K. Optimizing titanium implant nano-engineering via anodization. Materials & Design, 2022, 223: 111110. |
| [72] | GUO T Q, IVANOVSKI S, GULATI K. Fresh or aged: short time anodization of titanium to understand the influence of electrolyte aging on titania nanopores. Journal of Materials Science & Technology, 2022, 119: 245. |
| [73] |
ŠÍSTKOVÁ J, FIALOVÁ T, SVOBODA E, et al. Insight into antibacterial effect of titanium nanotubular surfaces with focus on Staphylococcus aureus and Pseudomonas aeruginosa. Scientific Reports, 2024, 14: 17303.
DOI |
| [74] |
WANG W Z, LIU H P, GUO Z L, et al. Various antibacterial strategies utilizing titanium dioxide nanotubes prepared via electrochemical anodization biofabrication method. Biomimetics, 2024, 9(7): 408.
DOI URL |
| [75] |
DENG Z M, YU L R, KUANG Y S, et al. Highly ordered nanotube-like microstructure on titanium dental implant surface fabricated via anodization enhanced cell adhesion and migration of human gingival fibroblasts. International Journal of Nanomedicine, 2024, 19: 2469.
DOI URL |
| [76] |
ZHU H Q, ZHANG H F, CHEN S H, et al. Fe-NC nanozymes-loaded TiO2 nanotube arrays endow titanium implants with excellent antioxidant capacity for inflammation inhibition and soft tissue integration. Composites Part B: Engineering, 2023, 267: 111054.
DOI URL |
| [77] |
KUNRATH M F, FARINA G, STURMER L B S, et al. TiO2 nanotubes as an antibacterial nanotextured surface for dental implants: systematic review and meta-analysis. Dental Materials, 2024, 40(6): 907.
DOI URL |
| [78] |
ALÉCIO A B W, FERREIRA C F, BABU J, et al. Doxycycline release of dental implants with nanotube surface, coated with poly lactic-co-glycolic acid for extended pH-controlled drug delivery. The Journal of Oral Implantology, 2019, 45(4): 267.
DOI URL |
| [79] | XIANG Y M, LIU X M, MAO C Y, et al. Infection-prevention on Ti implants by controlled drug release from folic acid/ZnO quantum dots sealed titania nanotubes. Materials Science & Engineering: C, 2018, 85: 214. |
| [80] |
GUO Y T, WANG X, WANG C Y, et al. In vitro behaviour of human gingival fibroblasts cultured on 3D-printed titanium alloy with hydrogenated TiO2 nanotubes. Journal of Materials Science. Materials in Medicine, 2022, 33(3): 27.
DOI |
| [81] |
XU L, WU C, LEI X C, et al. Effect of oxidation time on cytocompatibility of ultrafine-grained pure Ti in micro-arc oxidation treatment. Surface and Coatings Technology, 2018, 342: 12.
DOI URL |
| [82] | DINI C, NAGAY B E, CORDEIRO J M, et al. UV-photofunctionalization of a biomimetic coating for dental implants application. Materials Science & Engineering: C, 2020, 110: 110657. |
| [83] |
LI G Q, MA F C, LIU P, et al. Review of micro-arc oxidation of titanium alloys: mechanism, properties and applications. Journal of Alloys and Compounds, 2023, 948: 169773.
DOI URL |
| [84] | ZHOU M H, WANG J, WANG J, et al. Construction of a localized and long-acting CCN2 delivery system on percutaneous Ti implant surfaces for enhanced soft-tissue integration. ACS Applied Materials & Interfaces, 2023, 15(19): 22864. |
| [85] |
WEN X Y, LIU Y, XI F Q, et al. Micro-arc oxidation (MAO) and its potential for improving the performance of titanium implants in biomedical applications. Frontiers in Bioengineering and Biotechnology, 2023, 11: 1282590.
DOI URL |
| [86] |
MOLAEI M, FATTAH-ALHOSSEINI A, NOURI M, et al. Enhancing cytocompatibility, antibacterial activity and corrosion resistance of PEO coatings on titanium using incorporated ZrO2 nanoparticles. Surfaces and Interfaces, 2022, 30: 101967.
DOI URL |
| [87] |
JAYASREE A, CARTMELL S, IVANOVSKI S, et al. Electrically stimulated dental implants triggers soft-tissue integration and bactericidal functions. Advanced Functional Materials, 2024, 34(23): 2311027.
DOI URL |
| [88] |
HAN W, LIU Z Q, YU H, et al. An artificial piezoelectric- conductive integrated peri-implant gingiva enables efficient bacterial inhibition and soft-tissue integration. Advanced Fiber Materials, 2025, 7: 1128.
DOI |
| [1] | 彭德招, 李瑞, 王文鸿, 王梓瑞, 章志珍. 钠氯化物固态电解质研究进展[J]. 无机材料学报, 2026, 41(4): 409-420. |
| [2] | 陈坤, 姜勇刚, 冯军宗, 李良军, 胡艺洁, 冯坚. 锆酸镧多孔隔热材料研究进展[J]. 无机材料学报, 2026, 41(4): 421-431. |
| [3] | 石金瑜, 雷一明, 王晨旭, 张洁, 王京阳. 不同化学计量碳化钛的离子辐照损伤行为研究[J]. 无机材料学报, 2026, 41(3): 322-330. |
| [4] | 韦连金, 齐志杰, 汪信, 朱俊武, 付永胜. 纳米金刚石改性及其在电催化氧还原反应中的应用[J]. 无机材料学报, 2026, 41(3): 273-288. |
| [5] | 刘占一, 李勉, 欧阳晓平, 柴之芳, 黄庆. 干法后处理熔盐中Sr/Cs去除方法的研究进展[J]. 无机材料学报, 2026, 41(2): 150-158. |
| [6] | 孙炼, 张磊磊, 薛泽旭, 吴坤, 陈晔, 李志远, 王鲁凯, 王尊刚. 面向辐射探测应用的零维金属卤化物闪烁体研究进展[J]. 无机材料学报, 2026, 41(2): 159-176. |
| [7] | 任先培, 李超, 胡启威, 向晖, 彭跃红. 金属/过渡金属化合物莫特-肖特基析氢催化剂研究进展[J]. 无机材料学报, 2026, 41(2): 137-149. |
| [8] | 蒋君, 杨攻旅, 杨雨帆, 李毅, 袁宁一, 丁建宁. 有机胺盐调控钙钛矿薄膜结晶提升太阳能电池光电转换效率和稳定性[J]. 无机材料学报, 2026, 41(2): 186-192. |
| [9] | 范雨竹, 王媛, 王林燕, 向美玲, 鄢雨婷, 黎本慧, 李敏, 文志东, 王海超, 陈永福, 邱会东, 赵波, 周成裕. 氧化石墨烯基吸附材料去除水体中Pb(II): 制备、性能及机理[J]. 无机材料学报, 2026, 41(1): 12-26. |
| [10] | 蒋妮玉, 孙浩宸, 林明梅, 王定远, 刘来君. 铌镁酸铅-钛酸铅(PMN-PT)陶瓷的电卡效应[J]. 无机材料学报, 2026, 41(1): 96-104. |
| [11] | 徐锦涛, 高攀, 何唯一, 蒋圣楠, 潘秀红, 汤美波, 陈锟, 刘学超. 3C-SiC晶体制备研究进展[J]. 无机材料学报, 2026, 41(1): 1-11. |
| [12] | 余升阳, 苏海军, 姜浩, 余明辉, 姚佳彤, 杨培鑫. 激光增材制造超高温氧化物陶瓷孔隙缺陷形成及抑制研究进展[J]. 无机材料学报, 2025, 40(9): 944-956. |
| [13] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [14] | 肖晓琳, 王玉祥, 谷佩洋, 朱圳荣, 孙勇. 二维无机材料调控病损皮肤组织再生的研究进展[J]. 无机材料学报, 2025, 40(8): 860-870. |
| [15] | 马景阁, 吴成铁. 无机生物材料用于毛囊和毛发再生的研究[J]. 无机材料学报, 2025, 40(8): 901-910. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||