
Journal of Inorganic Materials ›› 2018, Vol. 33 ›› Issue (5): 494-500.DOI: 10.15541/jim20170326
Special Issue: 离子电池材料
• RESEARCH PAPER • Previous Articles Next Articles
XIAO Na1, PANG Yang1, SONG Yun1, WU Xiao-Jing1, FU Zheng-Wen2, ZHOU Yong-Ning1
Received:2017-07-03
Revised:2017-10-24
Published:2018-05-20
Online:2018-04-26
About author:XIAO Na. E-mail: 15210300014@fudan.edu.cn
Supported by:CLC Number:
XIAO Na, PANG Yang, SONG Yun, WU Xiao-Jing, FU Zheng-Wen, ZHOU Yong-Ning. Electrochemical Behavior of Sb-Si Nanocomposite Thin Films as Anode Materials for Sodium-ion Batteries[J]. Journal of Inorganic Materials, 2018, 33(5): 494-500.
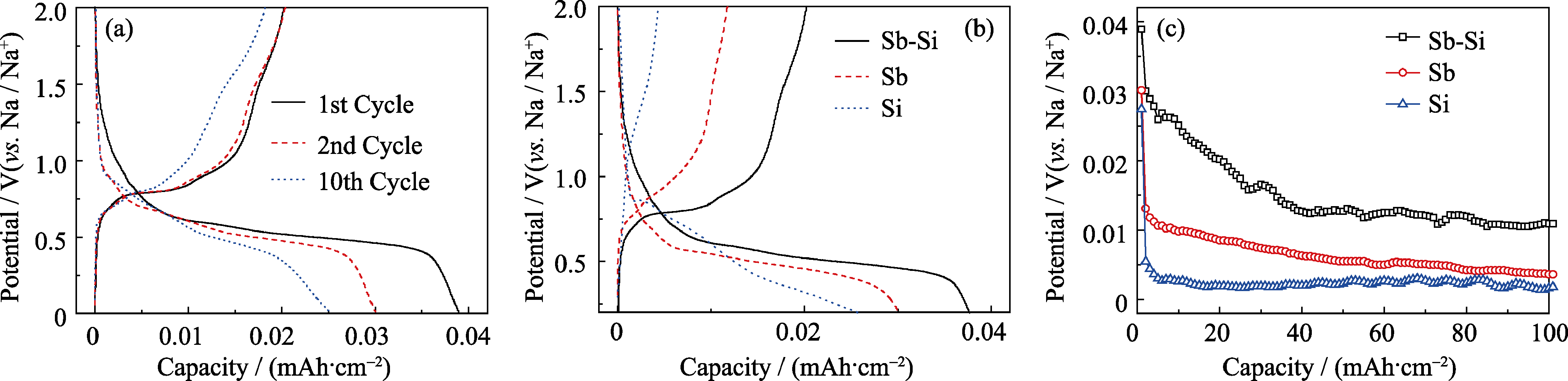
Fig. 1 Galvanostatic charge-discharge profiles of the as-deposited Sb-Si nanocomposite thin film at a current density of 10 μA/cm2 (a); The first galvanostatic charge-discharge profiles of the as-deposited Sb-Si nanocomposite thin film, pure Sb film and pure Si film (b); Cycling performance of Sb-Si nanocomposite thin film, pure Sb film and pure Si film (c)
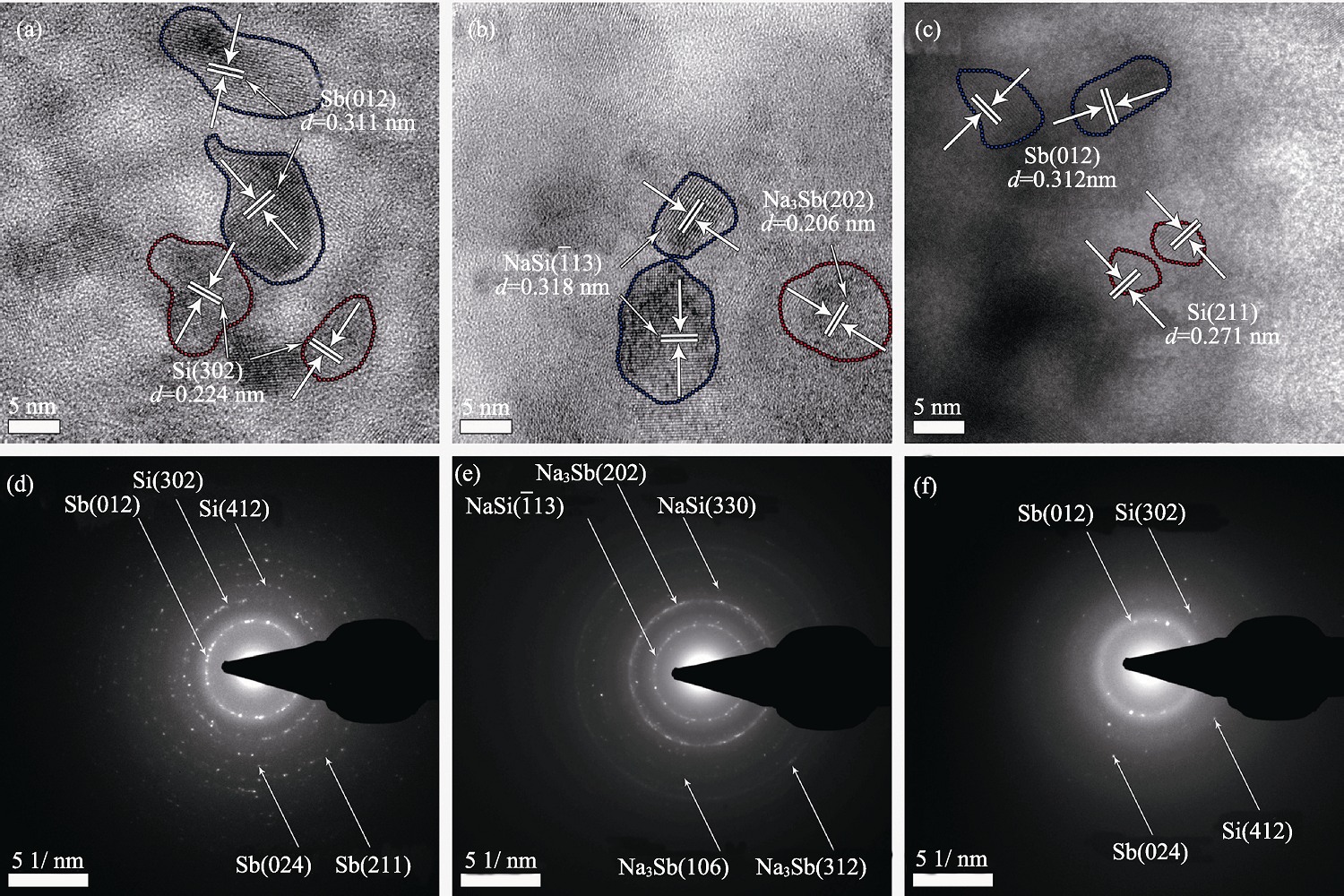
Fig. 4 HRTEM images (a)-(c) and SAED patterns (d)-(f) of Sb-Si nanocomposite thin film as-deposited, discharged to 0.1 V and recharged to 2.0 V, respectively
| Sb (R-3m) | Si (P41212) | |
|---|---|---|
| As-deposited | (No.85-1322) | (No.39-0973) |
| 0.311 | 0.311 (012) | |
| 0.226 | 0.228 (302) | |
| 0.181 | 0.183 (412) | |
| 0.158 | 0.156 (024) | |
| 0.139 | 0.139 (211) | |
| Na3Sb (P63/mmc) | NaSi (C2/c) | |
| Discharged | (No.74-1162) | (No.89-2625) |
| 0.320 | 0.319 (-113) | |
| 0.208 | 0.208 (202) | |
| 0.185 | 0.186 (330) | |
| 0.150 | 0.150 (106) | |
| 0.122 | 0.124 (312) | |
| Sb (R-3m) | Si (P41212) | |
| Recharged | (No.85-1322) | (No.39-0973) |
| 0.313 | 0.311 (012) | |
| 0.225 | 0.228 (302) | |
| 0.183 | 0.183 (412) | |
| 0.158 | 0.156 (024) |
Table 1 d-spacing (nm) derived from SAED analysis of as-deposited, first discharging to 0.1 V and recharging to 2.0 V of Sb-Si nanocomposite thin film electrode
| Sb (R-3m) | Si (P41212) | |
|---|---|---|
| As-deposited | (No.85-1322) | (No.39-0973) |
| 0.311 | 0.311 (012) | |
| 0.226 | 0.228 (302) | |
| 0.181 | 0.183 (412) | |
| 0.158 | 0.156 (024) | |
| 0.139 | 0.139 (211) | |
| Na3Sb (P63/mmc) | NaSi (C2/c) | |
| Discharged | (No.74-1162) | (No.89-2625) |
| 0.320 | 0.319 (-113) | |
| 0.208 | 0.208 (202) | |
| 0.185 | 0.186 (330) | |
| 0.150 | 0.150 (106) | |
| 0.122 | 0.124 (312) | |
| Sb (R-3m) | Si (P41212) | |
| Recharged | (No.85-1322) | (No.39-0973) |
| 0.313 | 0.311 (012) | |
| 0.225 | 0.228 (302) | |
| 0.183 | 0.183 (412) | |
| 0.158 | 0.156 (024) |
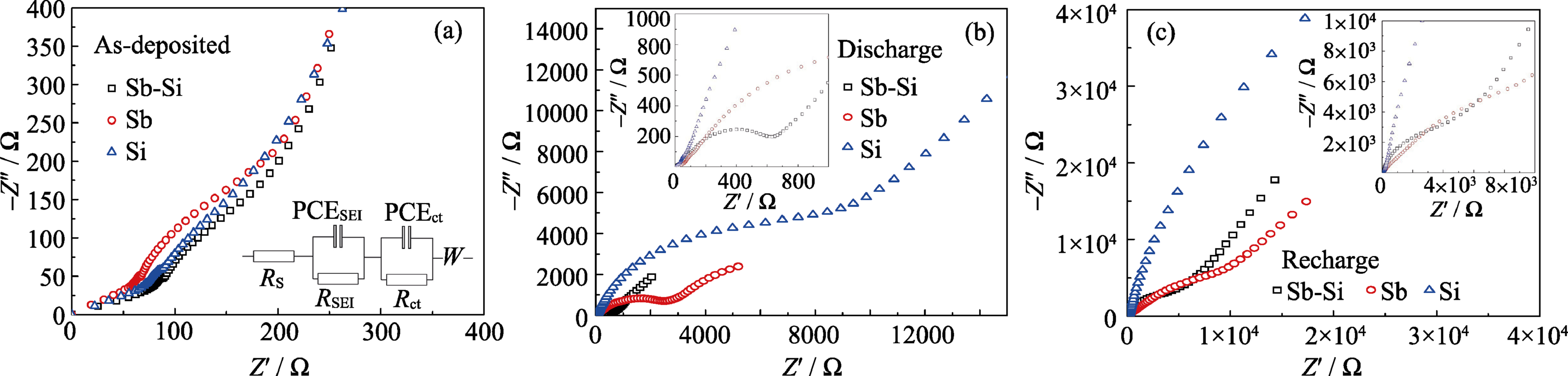
Fig. 5 Electrochemical impedance spectroscopy of Sb-Si nanocomposite thin film, Sb and Sn thin films at (a) as-deposited, (b) discharged and (c) charged states
| Rct /Ω | As-deposited | Discharged | Charged |
|---|---|---|---|
| Sb-Si | 132 | 762 | 791 |
| Sb | 116 | 3078 | 4985 |
| Si | 146 | 12521 | N/A |
Table 2 Impedance of as-deposited Sb-Si film, Sb film and Si film electrodes
| Rct /Ω | As-deposited | Discharged | Charged |
|---|---|---|---|
| Sb-Si | 132 | 762 | 791 |
| Sb | 116 | 3078 | 4985 |
| Si | 146 | 12521 | N/A |
| [1] | KIM S, SEO D, MA X, et al. Electrode materials for rechargeable sodium-ion batteries: potential alternatives to current lithium-ion batteries.Advanced Energy Materials. 2012, 2(7): 710-721. |
| [2] | PAN H, HU Y, CHEN L.Room-temperature stationary sodium- ion batteries for large-scale electric energy storage.Energy & Environmental Science, 2013, 6(8): 2338-2360. |
| [3] | ZHOU Y N, MA J, HU E,et al. Tuning charge-discharge induced unit cell breathing in layer-structured cathode materials for lithium- ion batteries. Nature Communications, 2014, 5(5): 5381. |
| [4] | XIE H.Comprehensive analysis on electrochemical energy storage mode and energy storage materials.Smart Grid, 2014(7): 4-8. |
| [5] | YABUUCHI N, KUBOTA K, DAHBI M,et al. Research development on sodium-ion batteries. Chemical Reviews, 2014, 114(23): 11636-11682. |
| [6] | ELLIS B L, NAZAR L F.Sodium and sodium-ion energy storage batteries.Current Opinion in Solid State & Materials Science, 2012, 16(4): 168-177. |
| [7] | WANG Y S, RONG X H, XU S Y, et al. Recent progress of electrode materials for room-temperature sodium-ion stationary batteries. Energy Storage Science and Technology, 2016, 5(3): 268-284. |
| [8] | LI Y, LU Y, ZHAO C,et al. Recent advances of electrode materials for low-cost sodium-ion batteries towards practical application for grid energy storage. Energy Storage Materials, 2017, 7: 130-151. |
| [9] | LI Y, HU Y, QI X,et al. Advanced sodium-ion batteries using superior low cost pyrolyzed anthracite anode: towards practical applications. Energy Storage Materials, 2016, 5: 191-197. |
| [10] | ZHOU Y N, SINA M, PEREIRA N,et al. FeO0.7F1.3/C nanocomposite as a high-capacity cathode material for sodium-ion batteries. Advanced Functional Materials, 2015, 25(5): 696-703. |
| [11] | YE F P, WANG L, LIAN F,et al. Advance in Na-ion batteries. Chemical Industry and Engineering Progress, 2013, 32(8): 1789-1795. |
| [12] | GE P, FOULETIER M.Electrochemical intercalation of sodium in graphite.Solid State Ionics, 1988, 28: 1172-1175. |
| [13] | SANGSTER J.C-Na (carbon-sodium) system.Journal of Phase Equilibria and Diffusion, 2007, 28(6): 571-579. |
| [14] | MORTAZAVI M, YE Q, BIRBILIS N,et al. High capacity group- 15 alloy anodes for Na-ion batteries: electrochemical and mechanical insights. Journal of Power Sources, 2015, 285: 29-36. |
| [15] | WANG M, YANG Z, WANG J,et al. Sb nanoparticles encapsulated in a reticular amorphous carbon network for enhanced sodium storage. Small, 2015, 11(40): 5381-5387. |
| [16] | WU L, HU X, QIAN J,et al. Sb-C nanofibers with long cycle life as an anode material for high-performance sodium-ion batteries. Energy & Environmental Science, 2013, 7(1): 323-328. |
| [17] | ZHANG N, LIU Y C, LU Y Y, et al. Spherical nano-Sb@C composite as a high-rate and ultra-stable anode material for sodium-ion batteries. Nano Research, 2015, 8(10): 3384-3393. |
| [18] | ALLAN P K, GRIFFIN J M, DARWICHE A,et al. Tracking sodium- antimonide phase transformations in sodium-ion anodes: insights from operando pair distribution function analysis and solid-state NMR spectroscopy. Journal of the American Chemical Society, 2016, 138(7): 2352-2365. |
| [19] | LI S, WANG Z, LIU J,et al. Yolk-shell Sn@C eggette-like nanostructure: application in lithium-ion and sodium-ion batteries. ACS Applied Materials & Interfaces, 2016, 8(30): 19438-19445. |
| [20] | NITHYADHARSENI P, REDDY M V, NALINI B,et al. Electrochemical studies of CNT/Si-SnSb nanoparticles for lithium ion batteries. Materials Research Bulletin, 2015, 70: 478-485. |
| [21] | WANG Y, ZHANG P, WANG J,et al. Lithium storage characteristics and electrochemical performance of Si-Sb-Ag composite anode materials. International Journal Electrochemical Science, 2015, 10: 9652-9665. |
| [22] | SZZECH J R, SONG J.Nanostructured silicon for high capacity lithium battery anodes.Energy & Environmental Science, 2010, 4(1): 56-72. |
| [23] | WANG J, WANG Y, ZHANG P,et al. Preparation and electrochemical properties of binary SixSb immiscible alloy for lithium ion batteries. Journal of Alloys & Compounds, 2014, 610(30): 308-314. |
| [24] | GUO H, ZHAO H, YIN C,et al. Si/SnSb alloy composite as high capacity anode materials for Li-ion batteries. Journal of Alloys and Compounds. 2006, 426(1/2): 277-280. |
| [25] | CHOU C, LEE M, HWANG G S.A comparative first-principles study on sodiation of silicon, germanium, and tin for sodium-ion |
| batteries. Journal of Physical Chemistry C, 2015, 119(27): 14843-14850. | |
| [26] | LIM C, HUANG T, SHAO P,et al. Experimental study on sodiation of amorphous silicon for use as sodium-ion battery anode. Electrochimica Acta, 2016, 211: 265-272. |
| [27] | XU Y, SWAANS E, BASAK S,et al. Reversible Na-ion uptake in Si nanoparticles. Advanced Energy Materials, 2016, 6(2): 1501436. |
| [28] | GORKA J, BAGGETTO L, KEUM J K,et al. The electrochemical reactions of SnO2 with Li and Na: a study using thin films and mesoporous carbons. Journal of Power Sources, 2015, 284: 1-9. |
| [29] | ZHOU Y N, ZHANG H, WU C L,et al. Electrochemical properties of GeO2 films fabricated by plused laser deposition. Chinese Journal of Inorganic Chemistry, 2007, 23(8): 1353-1357. |
| [30] | ELLIS L D, WILKES B N, HATCHARD T D,et al. In situ XRD study of silicon, lead and bismuth negative electrodes in nonaqueous sodium cells. Journal of the Electrochemical Society, 2013, 161(3): A416-A421. |
| [31] | KOMABA S, MATSUURA Y, ISHIKAWA T,et al. Redox reaction of Sn-polyacrylate electrodes in aprotic Na cell. Electrochemistry Communications, 2012, 21: 65-68. |
| [32] | LI G, WANG W, YANG W,et al. Epitaxial growth of group III-nitride films by pulsed laser deposition and their use in the development of LED devices. Surface Science Reports, 2015, 70(3): 380-423. |
| [33] | TANG P, LI B, FENG L, et al. Structural, electrical and optical properties of AlSb thin films deposited by pulsed laser deposition. Journal of Alloys and Compounds, 2017, 692: 22-25. |
| [34] | MOSCICKI T, RADZIEJEWSKA J, HOFFMAN J,et al. WB2 to WB3 phase change during reactive spark plasma sintering and pulsed laser ablation/deposition processes. Ceramics International, 2015, 41(7): 8273-8281. |
| [35] | ZHU X, ONG C S, XU X,et al. Direct observation of lithium-ion transport under an electrical field in LixCoO2 nanograins. Scientific Reports, 2013, 3(1): 1084. |
| [1] | BAO Ke, LI Xijun. Chemical Vapor Deposition of Vanadium Dioxide for Thermochromic Smart Window Applications [J]. Journal of Inorganic Materials, 2024, 39(3): 233-258. |
| [2] | LIU Song, ZHANG Faqiang, LUO Jin, LIU Zhifu. 0.9BaTiO3-0.1Bi(Mg1/2Ti1/2)O3 Ferroelectric Thin Films: Preparation and Energy Storage [J]. Journal of Inorganic Materials, 2024, 39(3): 291-298. |
| [3] | XU Xiangming, Husam N ALSHAREEF. Perspective of MXetronics [J]. Journal of Inorganic Materials, 2024, 39(2): 171-178. |
| [4] | HU Mengfei, HUANG Liping, LI He, ZHANG Guojun, WU Houzheng. Research Progress on Hard Carbon Anode for Li/Na-ion Batteries [J]. Journal of Inorganic Materials, 2024, 39(1): 32-44. |
| [5] | HU Ying, LI Ziqing, FANG Xiaosheng. Solution-prepared AgBi2I7 Thin Films and Their Photodetecting Properties [J]. Journal of Inorganic Materials, 2023, 38(9): 1055-1061. |
| [6] | CHEN Mingyue, YAN Zhichao, CHEN Jing, LI Minjuan, LIU Zhiyong, CAI Chuanbing. YBa2Cu3O7-δ Thin Film: Preparation by BaCl2/BaF2-MOD Method and Superconducting Property [J]. Journal of Inorganic Materials, 2023, 38(2): 199-204. |
| [7] | WANG Yutong, ZHANG Feifan, XU Naicai, WANG Chunxia, CUI Lishan, HUANG Guoyong. Research Progress of LiTi2(PO4)3 Anode for Aqueous Lithium-ion Batteries [J]. Journal of Inorganic Materials, 2022, 37(5): 481-492. |
| [8] | XIA Qiuying, SUN Shuo, ZAN Feng, XU Jing, XIA Hui. Amorphous LiSiON Thin Film Electrolyte for All-solid-state Thin Film Lithium Battery [J]. Journal of Inorganic Materials, 2022, 37(2): 230-236. |
| [9] | WANG Jing, XU Shoudong, LU Zhonghua, ZHAO Zhuangzhuang, CHEN Liang, ZHANG Ding, GUO Chunli. Hollow-structured CoSe2/C Anode Materials: Preparation and Sodium Storage Properties for Sodium-ion Batteries [J]. Journal of Inorganic Materials, 2022, 37(12): 1344-1350. |
| [10] | LI Kunru, HU Xinghui, ZHANG Zhengfu, GUO Yuzhong, HUANG Ruian. Three-dimensional Porous Biogenic Si/C Composite for High Performance Lithium-ion Battery Anode Derived from Equisetum Fluviatile [J]. Journal of Inorganic Materials, 2021, 36(9): 929-935. |
| [11] | ZHAO Changjiang,MA Chao,LIU Juncheng,LIU Zhigang,CHEN Yan. Sputtering Power on the Microstructure and Properties of MgF2 Thin Films Prepared with Magnetron Sputtering [J]. Journal of Inorganic Materials, 2020, 35(9): 1064-1070. |
| [12] | ZHAN Jing,XU Changfan,LONG Yiyu,LI Qihou. Bi2Mn4O10: Preparation by Polyacrylamide Gel Method and Electrochemical Performance [J]. Journal of Inorganic Materials, 2020, 35(7): 827-833. |
| [13] | XIA Tian, MENG Xie, LUO Ting, ZHAN Zhongliang. La 3+-substituted Sr2Fe1.5Ni0.1Mo0.4O6-δ as Anodes for Solid Oxide Fuel Cells [J]. Journal of Inorganic Materials, 2020, 35(5): 617-622. |
| [14] | ZHU Zeyang,WEI Jishi,HUANG Jianhang,DONG Xiangyang,ZHANG Peng,XIONG Huanming. Preparation of ZnO Nanorods with Lattice Vacancies and Their Application in Ni-Zn Battery [J]. Journal of Inorganic Materials, 2020, 35(4): 423-430. |
| [15] | ZHENG Shiyou, DONG Fei, PANG Yuepeng, HAN Pan, YANG Junhe. Research Progress on Nanostructured Metal Oxides as Anode Materials for Li-ion Battery [J]. Journal of Inorganic Materials, 2020, 35(12): 1295-1306. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||