
Journal of Inorganic Materials ›› 2016, Vol. 31 ›› Issue (8): 819-826.DOI: 10.15541/jim20150624
• Orginal Article • Previous Articles Next Articles
MAO Qi-Nan, LI He, JI Zhen-Guo, XI Jun-Hua, ZHANG Jun, KONG Zhe
Received:2015-12-14
Revised:2016-01-20
Published:2016-08-20
Online:2016-07-20
About author:MAO Qi-Nan. E-mail: maoqinan@hdu.edu.cn
Supported by:CLC Number:
MAO Qi-Nan, LI He, JI Zhen-Guo, XI Jun-Hua, ZHANG Jun, KONG Zhe. Influence of Eu2+ and Dy3+ Concentrations on Fluorescence and Phosphorescence of Sr2MgSi2O7 Phosphors[J]. Journal of Inorganic Materials, 2016, 31(8): 819-826.
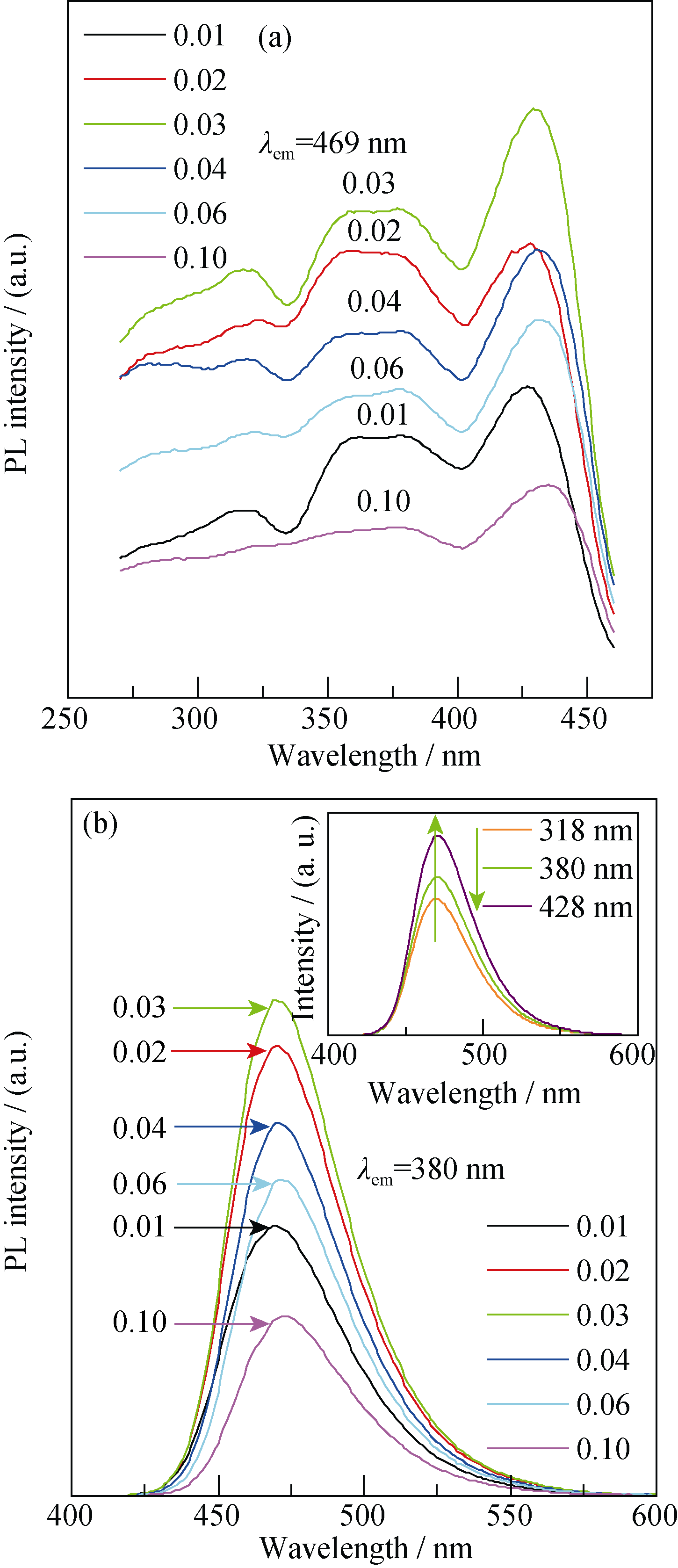
Fig. 2 Excitation and emission spectra of SMS: Eux with various Eu2+ concentrationsThe inset in Fig. 2 is the emission spectra of SMS: Eu0.03 under different excitation wavelengths
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 | |
|---|---|---|---|---|---|---|---|
| Intensity/(a.u.) | SMS: Eux | 387 | 645 | 710 | 536 | 453 | 257 |
| SMS: Eux, Dy0.02 | 317 | 494 | 621 | 419 | 365 | 165 | |
| Wavelength /nm | SMS: Eux | 470 | 470 | 471 | 471 | 472 | 473 |
| SMS: Eux, Dy0.02 | 470 | 470 | 471 | 471 | 472 | 473 | |
Table 1 Emission intensity and wavelength of SMS: Eux and SMS: Eux, Dy0.02 with varied Eu2+concentrations
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 | |
|---|---|---|---|---|---|---|---|
| Intensity/(a.u.) | SMS: Eux | 387 | 645 | 710 | 536 | 453 | 257 |
| SMS: Eux, Dy0.02 | 317 | 494 | 621 | 419 | 365 | 165 | |
| Wavelength /nm | SMS: Eux | 470 | 470 | 471 | 471 | 472 | 473 |
| SMS: Eux, Dy0.02 | 470 | 470 | 471 | 471 | 472 | 473 | |
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 |
|---|---|---|---|---|---|---|
| Centroid /cm-1 | 29550 | 30190 | 30130 | 30150 | 30140 | 30130 |
| Crystal-field splitting /cm-1 | 12820 | 12950 | 12960 | 13080 | 13120 | 13200 |
| Stokes shift /cm-1 | 1900 | 1790 | 1790 | 1690 | 1680 | 1670 |
Table 2 Centroid of 5d level of Eu2+, crystal-field splitting and Stokes shift in SMS: Eux phosphors
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 |
|---|---|---|---|---|---|---|
| Centroid /cm-1 | 29550 | 30190 | 30130 | 30150 | 30140 | 30130 |
| Crystal-field splitting /cm-1 | 12820 | 12950 | 12960 | 13080 | 13120 | 13200 |
| Stokes shift /cm-1 | 1900 | 1790 | 1790 | 1690 | 1680 | 1670 |
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 |
|---|---|---|---|---|---|---|
| τ1 /s | 10.7 | 11.4 | 4.5 | 5.1 | 3.6 | 3.1 |
| τ2 /s | 90.6 | 98.4 | 41.9 | 49.3 | 33.6 | 19.1 |
Table 3 Variations of decay constants, τ1 and τ2, with Eu2+ concentration in SMS: Eux, Dy0.02
| Concentration (x) | 0.01 | 0.02 | 0.03 | 0.04 | 0.06 | 0.10 |
|---|---|---|---|---|---|---|
| τ1 /s | 10.7 | 11.4 | 4.5 | 5.1 | 3.6 | 3.1 |
| τ2 /s | 90.6 | 98.4 | 41.9 | 49.3 | 33.6 | 19.1 |
| Concentration (y) | 0 | 0.01 | 0.02 | 0.04 | 0.06 | 0.10 | 0.20 |
|---|---|---|---|---|---|---|---|
| τ1 /s | 6.4 | 10.3 | 11.4 | 13.3 | 13.8 | 13.7 | 12.5 |
| τ2 /s | 48.3 | 94.1 | 98.4 | 129.7 | 141.1 | 164.5 | 138.4 |
Table 4 Variations of decay constants, τ1 and τ2, with Dy3+ concentration in SMS: Eu0.02, Dyy
| Concentration (y) | 0 | 0.01 | 0.02 | 0.04 | 0.06 | 0.10 | 0.20 |
|---|---|---|---|---|---|---|---|
| τ1 /s | 6.4 | 10.3 | 11.4 | 13.3 | 13.8 | 13.7 | 12.5 |
| τ2 /s | 48.3 | 94.1 | 98.4 | 129.7 | 141.1 | 164.5 | 138.4 |
| [1] | VAN DEN EECKHOUT K, SMET P F, POELMAN D. Persistent luminescence in Eu2+-doped compounds: a review.Materials, 2010, 3(4): 2536-2566. |
| [2] | MATSUZAWA T, AOKI Y, TAKEUCHI N, et al.A new long phosphorescent phosphor with high brightness, SrAl2O4: Eu2+, Dy3+.Journal of the Electrochemical Society, 1996, 143(8): 2670-2673. |
| [3] | KATSUMATA T, NABAE T, SASAJIMA K, et al.Effects of composition on the long phosphorescent SrAl2O4: Eu2+, Dy3+ phosphor crystals.Journal of the Electrochemical Society, 1997, 144(9): L243-L245. |
| [4] | QIU TAO, JI ZHEN-GUO, KONG ZHE, et al.Preparation and optimization of long persistent luminescent Sr4Al14O25: (Eu, Dy) phosphor materials.Journal of Inorganic Materials, 2012, 27(12): 1341-1344. |
| [5] | JI ZHENGUO, TIAN SHUN, CHEN WANKE, et al.Enhanced long lasting persistent luminescent SrAl2O4: Eu, Dy ceramics prepared by electron beam bombardment.Radiation Measurement, 2013, 59(59): 210-213. |
| [6] | WU HAOYI, HU YIHUA, CHEN LI, et al.Enhancement on the afterglow properties of Sr2MgSi2O7: Eu2+ by Er3+ codoping.Materials Letters, 2011, 65(17): 2676-2679. |
| [7] | YU SHENG-FEI, PI PI-HUI, WEN XIU-FANG et al. Coordination encapsulation on SrAl2O4: Eu2+, Dy3+ phosphor and its characterization.Journal of Inorganic Materials, 2007, 22(4): 642-646. |
| [8] | PUNG S Y, TAN C H, FAUZI M N A, et al. Luminescence of Sr2MgSi2O7 phosphor prepared by solid state reaction.Advanced Materials Research, 2014, 1024: 344-347. |
| [9] | SAKAI M, SHIRAISHI S, ZUKAWA T, et al.Phosphor, Light-emitting Device, and Plasma Display Panel. US Patent, 7902756, 2011.3.8. |
| [10] | SHRIVASTAVA R, KAUR J.Characterisation and mechanoluminescence studies of Sr2MgSi2O7: Eu2+, Dy3+.Journal of Radiation Research and Applied Sciences, 2015, 8(2): 201-207. |
| [11] | ZHANG Y, WEI W, DAS G K, et al.Engineering lanthanide-based materials for nanomedicine.Journal of Photochemistry and Photobiology C, 2014, 20(9): 71-96. |
| [12] | POORT S H M, REIJNHOUDT H M, VAN DER KUIP H O T, et al. Luminescence of Eu2+ in silicate host lattices with alkaline earth ions in a row.Journal of Alloys and Compounds, 1996, 241(1/2): 75-81. |
| [13] | LIN YUANHUA, TANG ZILONG, ZHANG ZHONGTAI, et al.Preparation of a new long afterglow blue-emitting Sr2MgSi2O7- based photoluminescent phosphor.Journal of Materials Science Letters, 2001, 20(16): 1505-1506. |
| [14] | WU HAOYI, HU YIHUA, WANG YINHAI, et al.Influence on luminescent properties of the Sr2MgSi2O7: Eu2+ by Dy3+, Nd3+ co-doping.Journal of Alloys and Compounds, 2009, 486(1): 549-553. |
| [15] | ZHANG XINGUO, TANG XUEPING, ZHANG JILIN, et al.Luminescent properties of Sr2MgSi2O7: Eu2+ as blue phosphor for NUV light-emitting diodes.Powder Technology, 2010, 204(2): 263-267. |
| [16] | ALVANI A A S, MOZTARZADEH F, SARABI A A. Preparation and properties of long afterglow in alkaline earth silicate phosphors co-doped by Eu2O3 and Dy2O3.Journal of Luminescence, 2005, 115(3): 147-150. |
| [17] | SHI CHAOSHU, FU YIBING, LIU BO, et al. The roles of Eu2+ and Dy3+ in the blue long-lasting phosphor Sr2MgSi2O7: Eu2+, Dy3+. Journal of Luminescence, 2007, 122-123: 11-13. |
| [18] | SAHU I P, BISEN D P, TAMRAKAR R K, et al.Enhancement of the photoluminescence and long afterglow properties of Sr2MgSi2O7: Eu2+ phosphor by Dy3+ co-doping.Luminescence, 2015, 30(8): 1318-1325. |
| [19] | WU HAOYI, HU YIHUA, WANG YINHAI, et al.Influence on the luminescence properties of the lattice defects in Sr2MgSi2O7: Eu2+, M (M = Dy3+, La3+ or Na1+).Journal of Alloys and Compounds, 2010, 497(1): 330-335. |
| [20] | ZHU YANAN, CHEN ZHI, GE MINGQIAO.Preparation of Sr2MgSi2O7: Eu2+, Dy3+ nanofiber by electrospinning assisted solid-state reaction.Journal of Materials Science: Materials in Electronics, 2014, 25(12): 5512-5517. |
| [21] | DEXTER D L.A theory of sensitized luminescence in solids.Journal of Chemical Physics, 1953, 21(5): 836-850. |
| [22] | JIANG LING, CHANG CHENGKANG, MAO DALI, et al.Concentration quenching of Eu2+ in Ca2MgSi2O7: Eu2+ phosphor.Materials Science and Engineering B, 2003, 103(3): 271-275. |
| [23] | BLASSE G, GRABMAIER B C. Luminescent Materials, 1st edition. Berlin: Springer, 1994: 10-32. |
| [24] | ZHANG MEI, HE XIN, LUO JIANYI, et al.Dependence of optical properties on the composition of (Ba1-x-ySrxEuy)Si2O2N2 phosphors for white light emitting diodes.Materials Chemistry and Physics, 2014, 147(3): 968-973. |
| [25] | SONG X F, FU R L, AGATHOPOULOS S, et al.Photoluminescence properties of Eu2+-activated CaSi2O2N2: redshift and concentration quenching.Journal of Applied Physics, 2009, 106(3): 033103. |
| [26] | SONG FENGLAN, CHEN DONGHUA, YUAN YUHONG.Synthesis of Sr2MgSi2O7: Eu, Dy and Sr2MgSi2O7: Eu, Dy, Nd by a modified solid-state reaction and their luminescent properties.Journal of Alloys and Compounds, 2008, 458(1): 564-568. |
| [27] | ROH H S, CHO I S, AN J S, et al.Enhanced photoluminescence property of Dy3+ co-doped BaAl2O4: Eu2+ green phosphors.Ceramics International, 2012, 38(1): 443-447. |
| [28] | SHIN H, ULLAH S, CHUNG K.Effect of nominal substitution of Dy3+ for host cations in SrAl2O4: Eu2+ phosphor on phase evolution and long afterglow luminescence.Journal of Alloys and Compounds, 2012, 544: 181-187. |
| [29] | HARANATH D, SHARMA P, CHANDER H.Optimization of boric acid content in developing efficient blue emitting, long persistent phosphor.Journal of Physics D, 2005, 38(3): 371-375. |
| [30] | GUPTA B K, KUMAR A, KUMAR P, et al.Probing on green long persistent Eu2+/Dy3+ doped Sr3SiAl4O11 emerging phosphor for security applications.Journal of Applied Physics, 2015, 117(24): 243104. |
| [31] | JU GIUFANG, HU YIHUA, CHEN LI, et al.Concentration quenching of persistent luminescence.Physica B, 2013, 415: 1-4. |
| [1] | JIANG Zongyu, HUANG Honghua, QING Jiang, WANG Hongning, YAO Chao, CHEN Ruoyu. Aluminum Ion Doped MIL-101(Cr): Preparation and VOCs Adsorption Performance [J]. Journal of Inorganic Materials, 2025, 40(7): 747-753. |
| [2] | ZHOU Yangyang, ZHANG Yanyan, YU Ziyi, FU Zhengqian, XU Fangfang, LIANG Ruihong, ZHOU Zhiyong. Enhancement of Piezoelectric Properties in CaBi4Ti4O15-based Ceramics through Bi3+ Self-doping Strategy [J]. Journal of Inorganic Materials, 2025, 40(6): 719-728. |
| [3] | SUN Yuxuan, WANG Zheng, SHI Xue, SHI Ying, DU Wentong, MAN Zhenyong, ZHENG Liaoying, LI Guorong. Defect Dipole Thermal-stability to the Electro-mechanical Properties of Fe Doped PZT Ceramics [J]. Journal of Inorganic Materials, 2025, 40(5): 545-551. |
| [4] | AN Ran, LIN Si, GUO Shigang, ZHANG Chong, ZHU Shun, HAN Yingchao. Iron-doped Nano-hydroxyapatite: Preparation and Ultraviolet Absorption Performance [J]. Journal of Inorganic Materials, 2025, 40(5): 457-465. |
| [5] | PAN Yuzhou, HE Fajian, XU Lulu, DAI Shixun. Broadband 3 μm Mid-infrared Emission in Dy3+/Yb3+ Co-doped Tellurite Glass under 980 nm LD Excitation [J]. Journal of Inorganic Materials, 2025, 40(5): 521-528. |
| [6] | QU Jifa, WANG Xu, ZHANG Weixuan, ZHANG Kangzhe, XIONG Yongheng, TAN Wenyi. Enhanced Sulfur-resistance for Solid Oxide Fuel Cells Anode via Doping Modification of NaYTiO4 [J]. Journal of Inorganic Materials, 2025, 40(5): 489-496. |
| [7] | MU Haojie, ZHANG Yuanjiang, YU Bin, FU Xiumei, ZHOU Shibin, LI Xiaodong. Preparation and Properties of ZrO2 Doped Y2O3-MgO Nanocomposite Ceramics [J]. Journal of Inorganic Materials, 2025, 40(3): 281-289. |
| [8] | SHEN Hao, CHEN Qianqian, ZHOU Boxiang, TANG Xiaodong, ZHANG Yuanyuan. Preparation and Energy Storage Properties of A-site La/Sr Co-doped PbZrO3 Thin Films [J]. Journal of Inorganic Materials, 2024, 39(9): 1022-1028. |
| [9] | CHENG Jun, ZHANG Jiawei, QIU Pengfei, CHEN Lidong, SHI Xun. Preparation and Thermoelectric Transport Properties of P-doped β-FeSi2 [J]. Journal of Inorganic Materials, 2024, 39(8): 895-902. |
| [10] | ZHAO Zhihan, GUO Peng, WEI Jing, CUI Li, LIU Shanze, ZHANG Wenlong, CHEN Rende, WANG Aiying. Ti Doped Diamond Like Carbon Films: Piezoresistive Properties and Carrier Transport Behavior [J]. Journal of Inorganic Materials, 2024, 39(8): 879-886. |
| [11] | LI Jiaqi, LI Xiaosong, LI Xuanhe, ZHU Xiaobing, ZHU Aimin. Transition Metal-doped Manganese Oxide: Synthesis by Warm Plasma and Electrocatalytic Performance for Oxygen Evolution Reaction [J]. Journal of Inorganic Materials, 2024, 39(7): 835-844. |
| [12] | TAM YU Puy Mang, XU Yu, GAO Quanhao, ZHOU Haiqiong, ZHANG Zhen, YIN Hao, LI Zhen, LÜ Qitao, CHEN Zhenqiang, MA Fengkai, SU Liangbi. Spectroscopic Properties and Optical Clusters in Erbium-doped CaF2, SrF2 and PbF2 Crystals [J]. Journal of Inorganic Materials, 2024, 39(3): 330-336. |
| [13] | LI Qiushi, YIN Guangming, LÜ Weichao, WANG Huaiyao, LI Jinglin, YANG Hongguang, GUAN Fangfang. Preparation of Na+/g-C3N4 Materials and Their Photocatalytic Degradation Mechanism on Methylene Blue [J]. Journal of Inorganic Materials, 2024, 39(10): 1143-1150. |
| [14] | DAI Le, LIU Yang, GAO Xuan, WANG Shuhao, SONG Yating, TANG Mingmeng, DMITRY V Karpinsky, LIU Lisha, WANG Yaojin. Self-polarization Achieved by Compositionally Gradient Doping in BiFeO3 Thin Films [J]. Journal of Inorganic Materials, 2024, 39(1): 99-106. |
| [15] | LI Guanglan, WANG Tianyu, LIU Yichen, LU Zhongfa. Layered NiFeCo-LDH-Ti6C3.75 Catalyst: Preparation and Performance for Oxygen Evolution Reaction [J]. Journal of Inorganic Materials, 2023, 38(7): 823-829. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||