
Journal of Inorganic Materials ›› 2023, Vol. 38 ›› Issue (11): 1309-1315.DOI: 10.15541/jim20230277
Special Issue: 【能源环境】燃料电池(202506)
• RESEARCH ARTICLE • Previous Articles Next Articles
YANG Daihui1( ), SUN Tian2, TIAN Hexin1, SHI Xiaofei1, MA Dongwei1
), SUN Tian2, TIAN Hexin1, SHI Xiaofei1, MA Dongwei1
Received:2023-06-12
Revised:2023-07-21
Published:2023-08-21
Online:2023-08-21
About author:YANG Daihui (1993-), male, Master. E-mail: 841671731@qq.com
Supported by:CLC Number:
YANG Daihui, SUN Tian, TIAN Hexin, SHI Xiaofei, MA Dongwei. Iron-nitrogen-codoped Mesoporous Carbon: Facile Synthesis and Catalytic Performance of Oxygen Reduction Reaction[J]. Journal of Inorganic Materials, 2023, 38(11): 1309-1315.
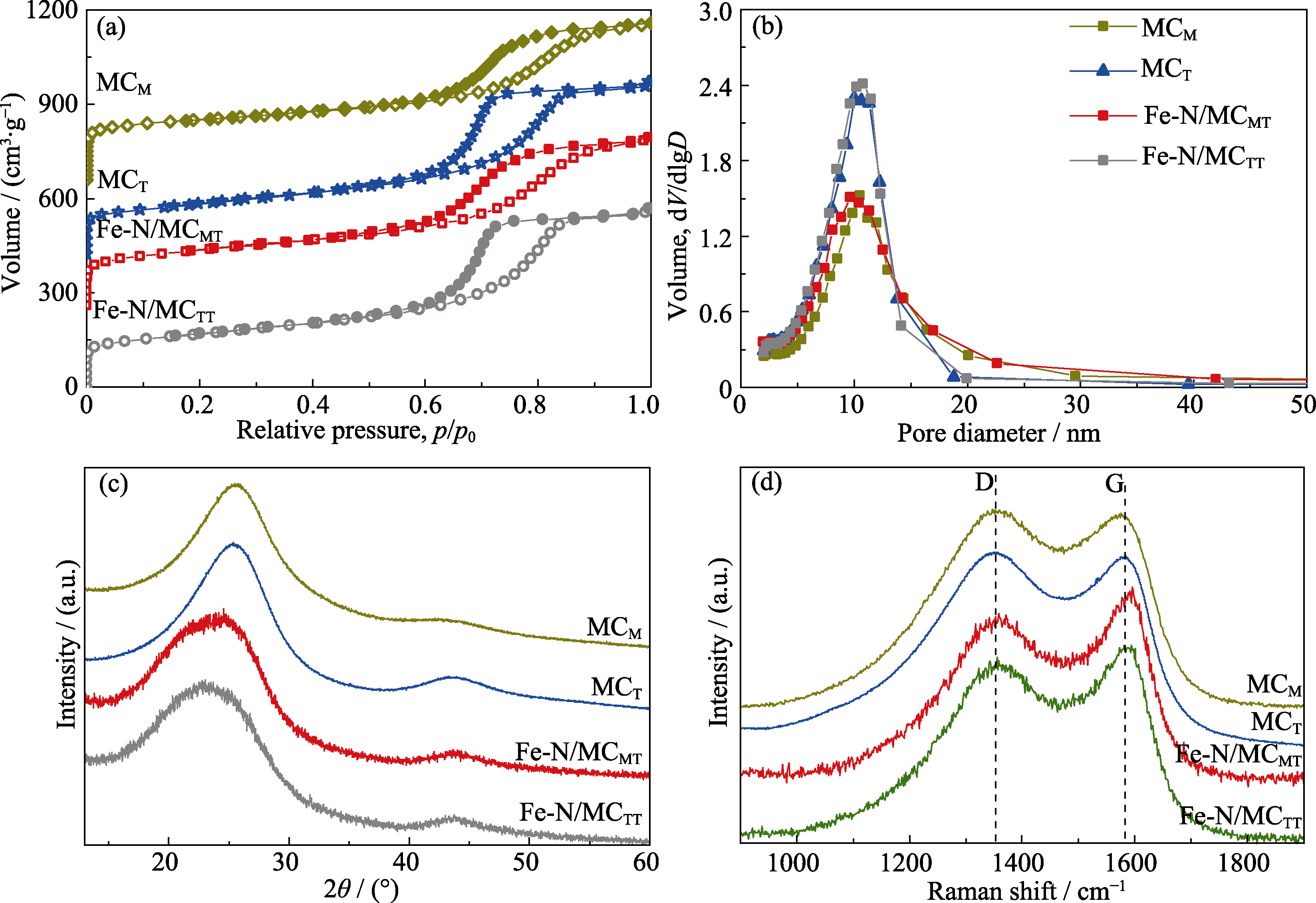
Fig. 2 (a) N2 adsorption-desorption isotherms, (b) pore-size distributions, (c) XRD patterns, and (d) Raman spectra of MCM, MCT, Fe-N/MCMT and Fe-N/MCTT

Fig. 3 HAADF-STEM images of (a-c) Fe-N/MCMT and (d-f) Fe-N/MCTT Single Fe atoms and Fe atom clusters are highlighted by white circles, respectively. Colorful figures are available on website
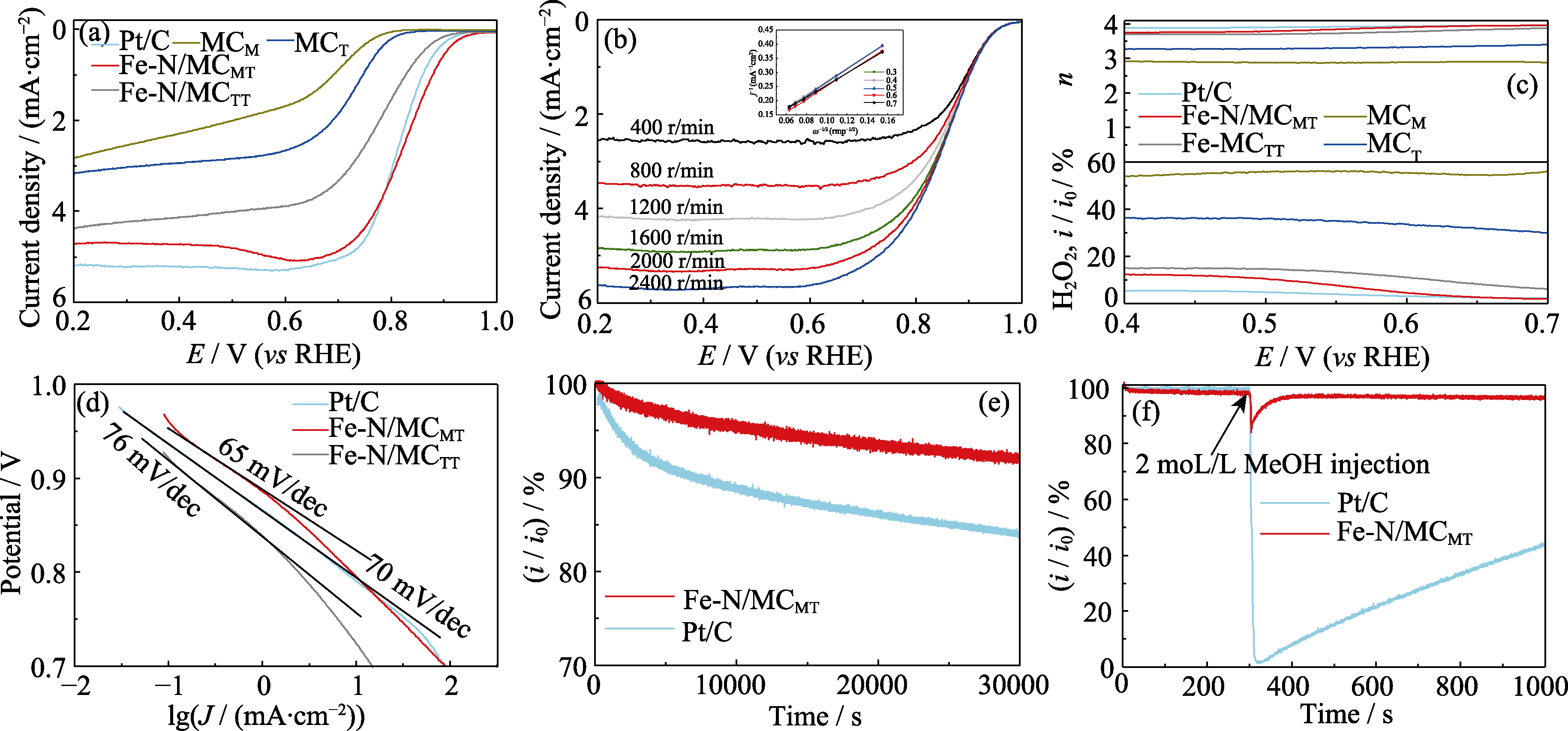
Fig. 5 ORR performance of catalysts (a) LSV curves of different catalysts in O2-saturated 0.1 mol/L KOH at a scan rate of 10 mV/s and a rotation rate of 1600 r/min; (b) LSV curves of Fe-N/MCMT at different rotation rates with inset showing K-L plots obtained from polarization curves; (c) Plots of number of electron transfer and H2O2 yield with different catalysts at the rotation speed of 1600 r/min; (d) Tafel plots derived from Fig. 6(a); (e, f) Chronoamperometric responses of Fe-N/MCMT and Pt/C in (e) presence or (f) absence of methanol at 0.7 V (vs RHE). Colorful figures are available on website
| Samples | N/%(in atomic) | Binding energy of relative nitrogen content/eV | ||||
|---|---|---|---|---|---|---|
| Pyridinic N | Fe-Nx | Pyrollic N | Graphitic N | Oxygenated N | ||
| Fe-N/MCMT | 5.92 | 0.2 (398.2) | 0.11 ( 399.3) | 0.58 (401) | 0.11 (403) | |
| Fe-N/MCTT | 5.12 | 0.21 (398.1) | 0.07 ( 399.2) | 0.68 (401) | 0.04 (403) | |
| MCM | 16.48 | 0.41 (398.3) | 0.39 (400) | 0.16 (401) | 0.04 (403) | |
| MCT | 10.00 | 0.26 (398.3) | 0.28 (400) | 0.42 (400.98) | 0.04 (403) | |
Table 1 Nitrogen content of each sample
| Samples | N/%(in atomic) | Binding energy of relative nitrogen content/eV | ||||
|---|---|---|---|---|---|---|
| Pyridinic N | Fe-Nx | Pyrollic N | Graphitic N | Oxygenated N | ||
| Fe-N/MCMT | 5.92 | 0.2 (398.2) | 0.11 ( 399.3) | 0.58 (401) | 0.11 (403) | |
| Fe-N/MCTT | 5.12 | 0.21 (398.1) | 0.07 ( 399.2) | 0.68 (401) | 0.04 (403) | |
| MCM | 16.48 | 0.41 (398.3) | 0.39 (400) | 0.16 (401) | 0.04 (403) | |
| MCT | 10.00 | 0.26 (398.3) | 0.28 (400) | 0.42 (400.98) | 0.04 (403) | |
| Sample | XPS | ||||
|---|---|---|---|---|---|
| Fe/% | N/% | S/% | O/% | C/% | |
| Fe-N/MCMT | 0.49 | 5.92 | 0.32 | 8.41 | 84.86 |
| Fe-N/MCTT | 0.64 | 5.12 | 0.57 | 9.34 | 84.33 |
| MCM | 0 | 16.48 | 1.27 | 7.92 | 74.33 |
| MCT | 0 | 10 | 0.9 | 5 | 84.1 |
Table 2 Elemental percentages in atom of each sample
| Sample | XPS | ||||
|---|---|---|---|---|---|
| Fe/% | N/% | S/% | O/% | C/% | |
| Fe-N/MCMT | 0.49 | 5.92 | 0.32 | 8.41 | 84.86 |
| Fe-N/MCTT | 0.64 | 5.12 | 0.57 | 9.34 | 84.33 |
| MCM | 0 | 16.48 | 1.27 | 7.92 | 74.33 |
| MCT | 0 | 10 | 0.9 | 5 | 84.1 |
| [1] |
JIANG L, XU S, XIA B, et al. Defect engineering of graphene hybrid catalysts for oxygen reduction reactions. J. Inorg. Mater., 2022, 37(2): 215.
DOI |
| [2] |
KIM D, ZUSSBLATT N P, CHUNG H T, et al. Highly graphitic mesoporous Fe, N-doped carbon materials for oxygen reduction electrochemical catalysts. ACS Appl. Mater. Interfaces, 2018, 10(30): 25337.
DOI URL |
| [3] |
SUN Y T, DING S, XU S S, et al. Metallic two-dimensional metal-organic framework arrays for ultrafast water splitting. J. Power Sources. 2021, 494: 229733.
DOI URL |
| [4] |
RAMASWAMY N, TYLUS U, JIA Q Y, et al. Activity descriptor identification for oxygen reduction on nonprecious electrocatalysts: linking surface science to coordination chemistry. J. Am. Chem. Soc., 2013, 135(41): 15443.
DOI PMID |
| [5] |
LEE S H, KIM J, CHUNG D Y, et al. Design principle of Fe-N-C electrocatalysts: how to optimize multimodal porous structures? J. Am. Chem. Soc., 2019, 141(5): 2035.
DOI PMID |
| [6] |
KONG A G, ZHU X F, HAN Z, et al. Ordered hierarchically micro- and mesoporous Fe-Nx-embedded graphitic architectures as efficient electrocatalysts for oxygen reduction reaction. ACS Catal., 2014, 4(6): 1793.
DOI URL |
| [7] |
NISHIHARA H, KYOTANI T. Templated nanocarbons for energy storage. Adv. Mater., 2012, 24(33): 4473.
DOI URL |
| [8] |
PENG Y, LU B Z, CHEN S W. Carbon-supported single atom catalysts for electrochemical energy conversion and storage. Adv. Mater., 2018, 30(48): 1801995.
DOI URL |
| [9] |
LEE J S, PARK G, KIM S T, et al. A highly efficient electrocatalyst for the oxygen reduction reaction: N-doped ketjenblack incorporated into Fe/Fe3C-functionalized melamine foam. Angew. Chem. Int. Ed., 2013, 52(3): 1026.
DOI URL |
| [10] |
YANG L, CHENG D J, ZENG X F, et al. Unveiling the high- activity origin of single-atom iron catalysts for oxygen reduction reaction. Proc. Natl. Acad. Sci. U.S.A., 2018, 115(26): 6626.
DOI URL |
| [11] |
LU X, YIM W L, SURYANTO B H R, et al. Electrocatalytic oxygen evolution at surface-oxidized multiwall carbon nanotubes. J. Am. Chem. Soc., 2015, 137(8): 2901.
DOI PMID |
| [12] |
LIU J, JIAO M G, MEI B B, et al. Carbon-supported divacancy- anchored platinum single-atom electrocatalysts with superhigh Pt utilization for the oxygen reduction reaction. Angew. Chem. Int. Ed., 2019, 131(4): 1175.
DOI URL |
| [13] |
ZHANG S G, MANDAI T, UENO K, et al. Hydrogen-bonding spramolecular protic salt as an “all-in-one” precursor for nitrogen- doped mesoporous carbons for CO2 adsorption. Nano Energy, 2015, 13: 376.
DOI URL |
| [14] |
XING C, YANG D H, ZHANG Y T, et al. Semi-closed synthesis of nitrogen and oxygen co-doped mesoporous carbon for selective aqueous oxidation. Green Energy Environ., 2022, 7(1): 43.
DOI URL |
| [15] |
SING K S. Reporting physisorption data for gas/solid systems with special reference to the determination of surface area and porosity. Pure Appl. Chem., 1985, 57(4): 603.
DOI URL |
| [16] | DAS A, CHAKRABORTY B, SOOD A K B. Raman spectroscopy of graphene on different substrates and influence of defects. Mater. Sci., 2008, 31: 579. |
| [17] |
WU Z Y, XU X X, HU B C, et al. Iron carbide nanoparticles encapsulated in mesoporous Fe-N-doped carbon nanofibers for efficient electrocatalysis. Angew. Chem. Int. Ed., 2015, 54(28): 8179.
DOI URL |
| [18] |
SUN M, DAVENPORT D, LIU H J, et al. Highly efficient and sustainable non-precious-metal Fe-N-C electrocatalysts for the oxygen reduction reaction. J. Mater. Chem. A, 2018, 6(6): 2527.
DOI URL |
| [19] |
SEROV A, ARTYUSHKOVA K, ATANASSOV P. Fe-N-C oxygen reduction fuel cell catalyst derived from carbendazim: synthesis, structure, and reactivity. Adv. Energy Mater., 2014, 4(10): 1301735.
DOI URL |
| [20] |
ZHAO Y X, LAI Q X, WANG Y, et al. Interconnected hierarchically porous Fe, N-codoped carbon nanofibers as efficient oxygen reduction catalysts for Zn-air batteries. ACS Appl. Mater. Interfaces, 2017, 9(19): 16178.
DOI URL |
| [21] |
DING Y J, NIU Y C, YANG J, et al. A metal-amino acid complex- derived bifunctional oxygen electrocatalyst for rechargeable zinc-air batteries. Small, 2016, 12(39): 5414.
DOI URL |
| [22] |
LEFEVRE M, PROIETTI E, JAOUEN F, et al. Iron-based catalysts with improved oxygen reduction activity in polymer electrolyte fuel cells. Science, 2009, 324(5923): 71.
DOI PMID |
| [23] |
LI J, SONG Y J, ZHANG G X, et al. Multicolor printing using electric-field-responsive and photocurable photonic crystals. Adv. Funct. Mater., 2017, 27(43): 1702825.
DOI URL |
| [24] |
CAO L, LI Z H, GU Y, et al. Rational design of n-doped carbon nanobox-supported Fe/Fe2N/Fe3C nanoparticles as efficient oxygen reduction catalysts for Zn-air batteries. J. Mater. Chem. A, 2017, 5(22): 11340.
DOI URL |
| [1] | GUO Ziyu, ZHU Yunzhou, WANG Li, CHEN Jian, LI Hong, HUANG Zhengren. Effect of Zn2+ Catalyst on Microporous Structure of Porous Carbon Prepared from Phenolic Resin/Ethylene Glycol [J]. Journal of Inorganic Materials, 2025, 40(5): 466-472. |
| [2] | LI Jianjun, CHEN Fangming, ZHANG Lili, WANG Lei, ZHANG Liting, CHEN Huiwen, XUE Changguo, XU Liangji. Peroxymonosulfate Activation by CoFe2O4/MgAl-LDH Catalyst for the Boosted Degradation of Antibiotic [J]. Journal of Inorganic Materials, 2025, 40(4): 440-448. |
| [3] | XIN Zhenyu, GUO Ruihua, WUREN Tuoya, WANG Yan, AN Shengli, ZHANG Guofang, GUAN Lili. Pt-Fe/GO Nanocatalysts: Preparation and Electrocatalytic Performance on Ethanol Oxidation [J]. Journal of Inorganic Materials, 2025, 40(4): 379-387. |
| [4] | SUN Shujuan, ZHENG Nannan, PAN Haokun, MA Meng, CHEN Jun, HUANG Xiubing. Research Progress on Preparation Methods of Single-atom Catalysts [J]. Journal of Inorganic Materials, 2025, 40(2): 113-127. |
| [5] | LI Na, CAO Ruixiao, WEI Jin, ZHOU Han, XIAO Hongmei. Performance and Influencing Factors of Iron-based Catalyst for Ortho to Para Hydrogen Conversion [J]. Journal of Inorganic Materials, 2025, 40(1): 47-52. |
| [6] | LIAN Minli, SU Jiaxin, HUANG Hongyang, JI Yuyin, DENG Haifan, ZHANG Tong, CHEN Chongqi, LI Dalin. Supported Ni Catalysts from Ni-Mg-Al Hydrotalcite-like Compounds:Preparation and Catalytic Performance for Ammonia Decomposition [J]. Journal of Inorganic Materials, 2025, 40(1): 53-60. |
| [7] | LIU Lei, GUO Ruihua, WANG Li, WANG Yan, ZHANG Guofang, GUAN Lili. Oxygen Reduction Reaction on Pt3Co High-index Facets by Density Functional Theory [J]. Journal of Inorganic Materials, 2025, 40(1): 39-46. |
| [8] | JIN Yuxiang, SONG Erhong, ZHU Yongfu. First-principles Investigation of Single 3d Transition Metals Doping Graphene Vacancies for CO2 Electroreduction [J]. Journal of Inorganic Materials, 2024, 39(7): 845-852. |
| [9] | YE Zibin, ZOU Gaochang, WU Qiwen, YAN Xiaomin, ZHOU Mingyang, LIU Jiang. Preparation and Performances of Tubular Cone-shaped Anode-supported Segmented-in-series Direct Carbon Solid Oxide Fuel Cell [J]. Journal of Inorganic Materials, 2024, 39(7): 819-827. |
| [10] | ZHANG Wenyu, GUO Ruihua, YUE Quanxin, HUANG Yarong, ZHANG Guofang, GUAN Lili. High-entropy Phosphide Bifunctional Catalyst: Preparation and Performance of Efficient Water Splitting [J]. Journal of Inorganic Materials, 2024, 39(11): 1265-1274. |
| [11] | XIE Tian, SONG Erhong. Effect of Elastic Strains on Adsorption Energies of C, H and O on Transition Metal Oxides [J]. Journal of Inorganic Materials, 2024, 39(11): 1292-1300. |
| [12] | HE Qian, TANG Wanlan, HAN Bingkun, WEI Jiayuan, LÜ Wenxuan, TANG Zhaomin. pH Responsive Copper-Doped Mesoporous Silica Nanocatalyst for Enhanced Chemo-Chemodynamic Tumor Therapy [J]. Journal of Inorganic Materials, 2024, 39(1): 90-98. |
| [13] | WANG Lei, LI Jianjun, NING Jun, HU Tianyu, WANG Hongyang, ZHANG Zhanqun, WU Linxin. Enhanced Degradation of Methyl Orange with CoFe2O4@Zeolite Catalyst as Peroxymonosulfate Activator: Performance and Mechanism [J]. Journal of Inorganic Materials, 2023, 38(4): 469-476. |
| [14] | YAO Yishuai, GUO Ruihua, AN Shengli, ZHANG Jieyu, CHOU Kuochih, ZHANG Guofang, HUANG Yarong, PAN Gaofei. In-situ Loaded Pt-Co High Index Facets Catalysts: Preparation and Electrocatalytic Performance [J]. Journal of Inorganic Materials, 2023, 38(1): 71-78. |
| [15] | WANG Ruyi, XU Guoliang, YANG Lei, DENG Chonghai, CHU Delin, ZHANG Miao, SUN Zhaoqi. p-n Heterostructured BiVO4/g-C3N4 Photoanode: Construction and Its Photoelectrochemical Water Splitting Performance [J]. Journal of Inorganic Materials, 2023, 38(1): 87-96. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||