无机材料学报 ›› 2023, Vol. 38 ›› Issue (5): 537-543.DOI: 10.15541/jim20220449 CSTR: 32189.14.10.15541/jim20220449
马晓森( ), 张丽晨, 刘砚超, 汪全华, 郑家军(
), 张丽晨, 刘砚超, 汪全华, 郑家军( ), 李瑞丰
), 李瑞丰
收稿日期:2022-08-01
修回日期:2022-11-02
出版日期:2022-11-16
网络出版日期:2022-11-16
通讯作者:
郑家军, 教授. E-mail: zhengjiajun@tyut.edu.cn作者简介:马晓森(1998-), 男, 硕士研究生. E-mail: 294945674@qq.com
基金资助:
MA Xiaosen( ), ZHANG Lichen, LIU Yanchao, WANG Quanhua, ZHENG Jiajun(
), ZHANG Lichen, LIU Yanchao, WANG Quanhua, ZHENG Jiajun( ), LI Ruifeng
), LI Ruifeng
Received:2022-08-01
Revised:2022-11-02
Published:2022-11-16
Online:2022-11-16
Contact:
ZHENG Jiajun, professor. E-mail: zhengjiajun@tyut.edu.cnAbout author:MA Xiaosen (1998-), male, Master candidate. E-mail: 294945674@qq.com
Supported by:摘要:
常见的吸附剂如13X等的硅铝比较低, 具有较强的亲水性, 但水和有机挥发份(VOCs)之间的竞争吸附, 常常会影响吸附剂对VOCs实际脱除效果。本研究利用CTABr为模板剂, 正硅酸乙酯为硅源, 对13X进行表面修饰, 制备了以13X为核, 介孔硅为壳的核壳复合材料13X@SiO2, 并以甲苯作为探针分子在穿透实验装置对改性前后沸石分别进行干/湿条件下的吸附性能测试。结果表明: 在干燥条件下, 13X@SiO2-2.6样品(制备中添加了2.6 mL正硅酸乙酯)相比13X原样的吸附量提升了18%左右。在30%和50%相对湿度下, 13X@SiO2的最优吸附容量分别提高了约53%和90%; 循环再生实验表明13X@SiO2-2.6样品经2次再生后仍保持初始样品90%的甲苯吸附量。
中图分类号:
马晓森, 张丽晨, 刘砚超, 汪全华, 郑家军, 李瑞丰. 13X@SiO2合成及其甲苯吸附性能[J]. 无机材料学报, 2023, 38(5): 537-543.
MA Xiaosen, ZHANG Lichen, LIU Yanchao, WANG Quanhua, ZHENG Jiajun, LI Ruifeng. 13X@SiO2: Synthesis and Toluene Adsorption[J]. Journal of Inorganic Materials, 2023, 38(5): 537-543.
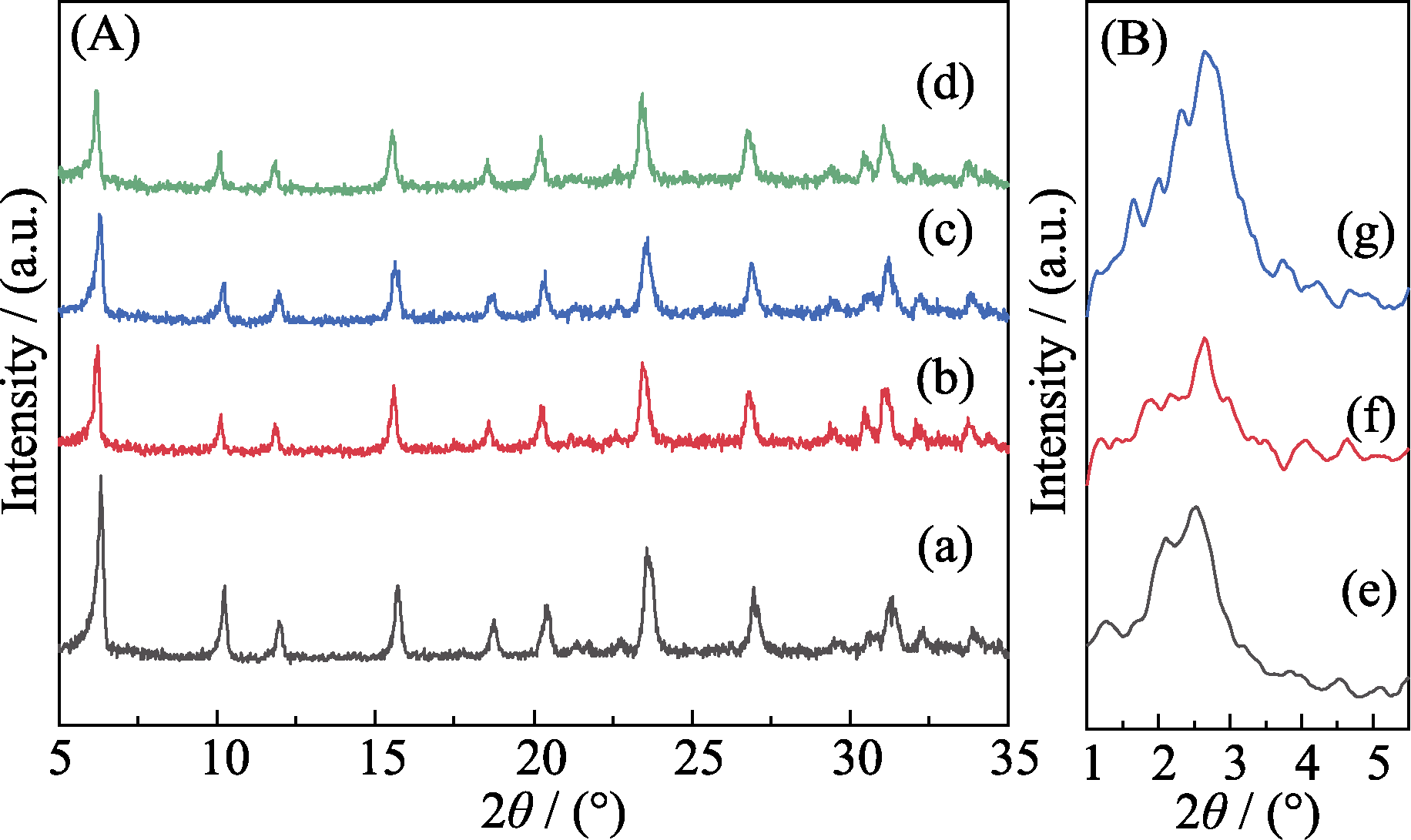
图1 样品(a)13X、(b, e)13X@SiO2-2.2、(c, f)13X@SiO2-2.6和(d, g)13X@SiO2-3.5的XRD图谱
Fig. 1 XRD patterns of the samples of (a) 13X, (b, e) 13X@SiO2-2.2,(c, f) 13X@SiO2-2.6, and (d, g) 13X@SiO2-3.5 (A) Large angle XRD patterns; (B) Small angle XRD patterns

图S3 样品的(A)氮气吸附脱附等温曲线与(B)DFT模型孔径分布图
Fig. S3 (A) Nitrogen adsorption-desorption isothermal curves and (B) the corresponding pore size distributions decided by a DFT model of samples
| Sample | SBET/(m2·g-1) | Sext/(m2·g-1) | Smic/(m2·g-1) | Vmic/(cm3·g-1) | Vmes/(cm3·g-1) |
|---|---|---|---|---|---|
| 13X | 314 | 14 | 299 | 0.11 | 0.02 |
| 13X@SiO2-2.2 | 324 | 95 | 229 | 0.09 | 0.07 |
| 13X@SiO2-2.6 | 337 | 130 | 207 | 0.08 | 0.09 |
| 13X@SiO2-3.5 | 444 | 259 | 184 | 0.07 | 0.18 |
表1 样品的比表面积及孔结构参数
Table 1 Textural properties of the samples
| Sample | SBET/(m2·g-1) | Sext/(m2·g-1) | Smic/(m2·g-1) | Vmic/(cm3·g-1) | Vmes/(cm3·g-1) |
|---|---|---|---|---|---|
| 13X | 314 | 14 | 299 | 0.11 | 0.02 |
| 13X@SiO2-2.2 | 324 | 95 | 229 | 0.09 | 0.07 |
| 13X@SiO2-2.6 | 337 | 130 | 207 | 0.08 | 0.09 |
| 13X@SiO2-3.5 | 444 | 259 | 184 | 0.07 | 0.18 |
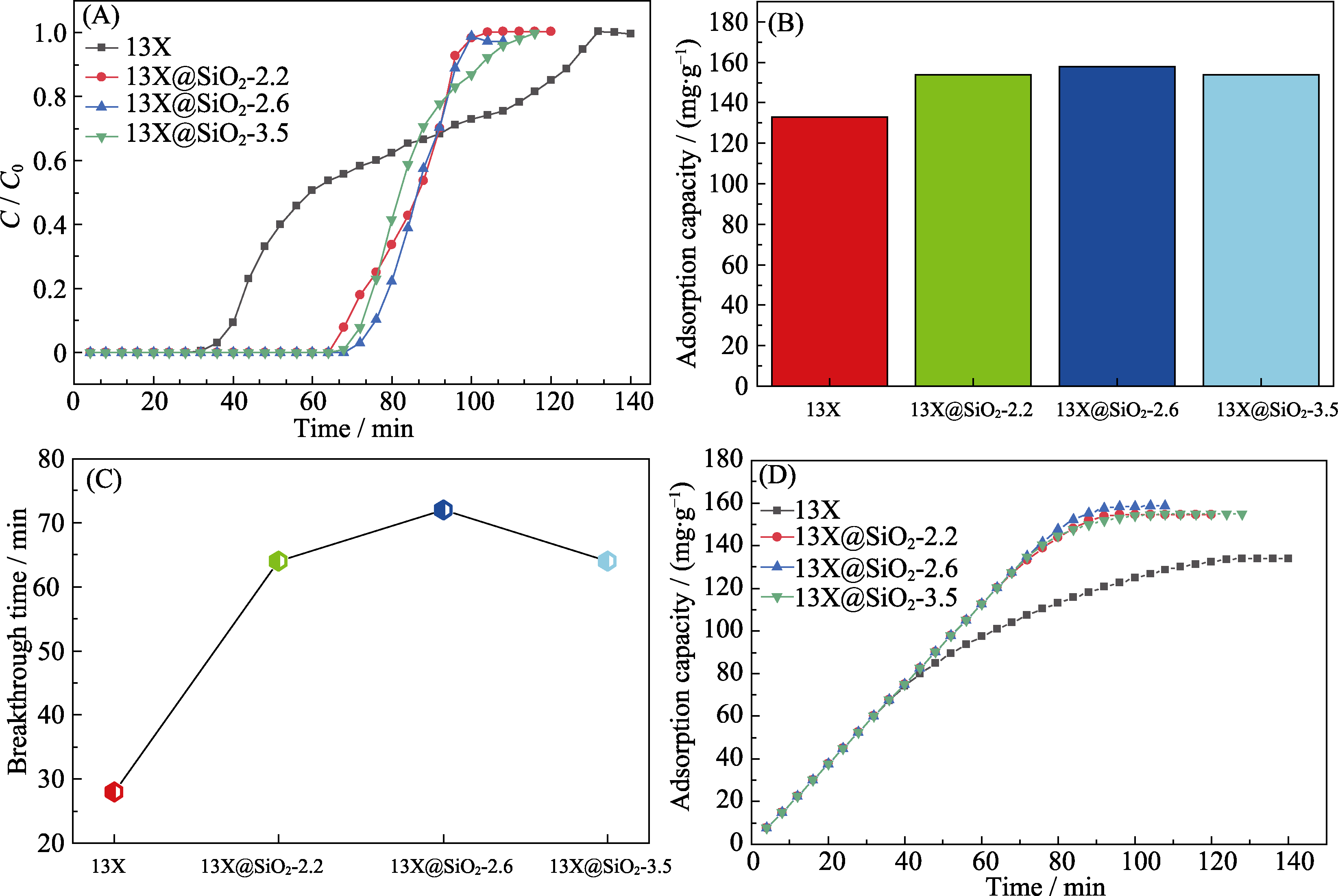
图4 干条件下不同吸附剂对甲苯的吸附实验
Fig. 4 Adsorption of toluene on the different adsorbents under dry condition (A) Adsorption breakthrough curves; (B) Saturated adsorption capacity; (C) Comparison of the breakthrough times; (D) Cumulative adsorption capacity of different adsorbents
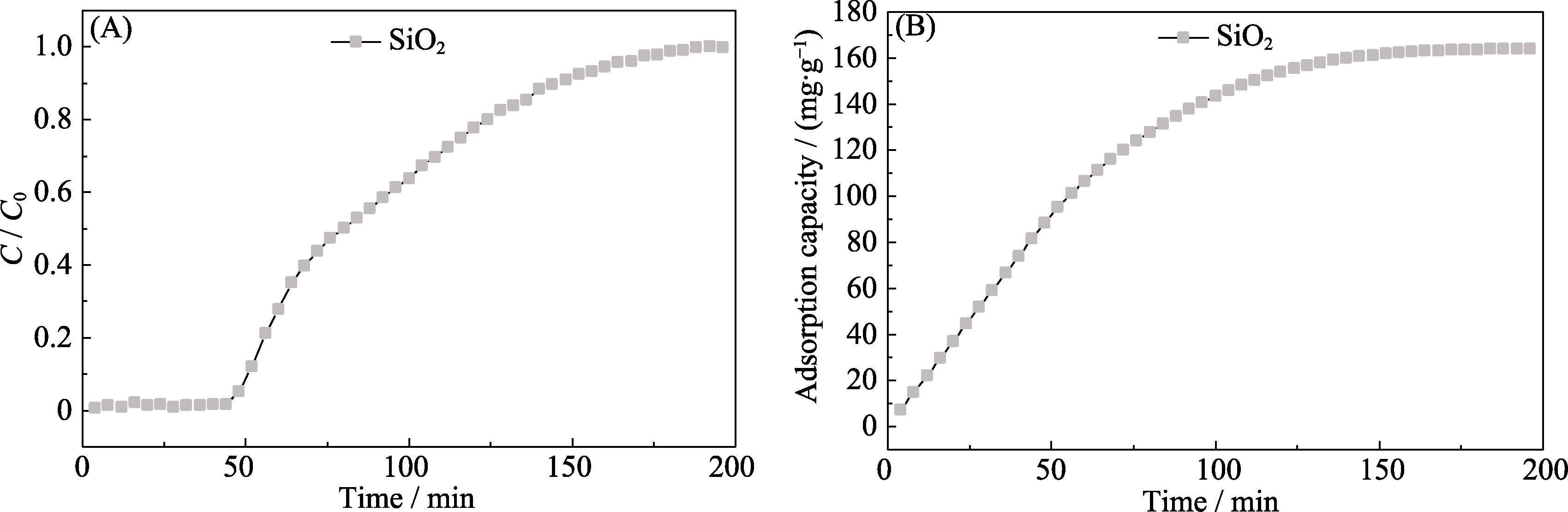
图S6 SiO2在干条件下对甲苯的(A)吸附穿透曲线和(B)累积吸附量
Fig. S6 (A) Toluene adsorption breakthrough curves and (B) cumulative toluene adsorption capacity of SiO2 under dry condition
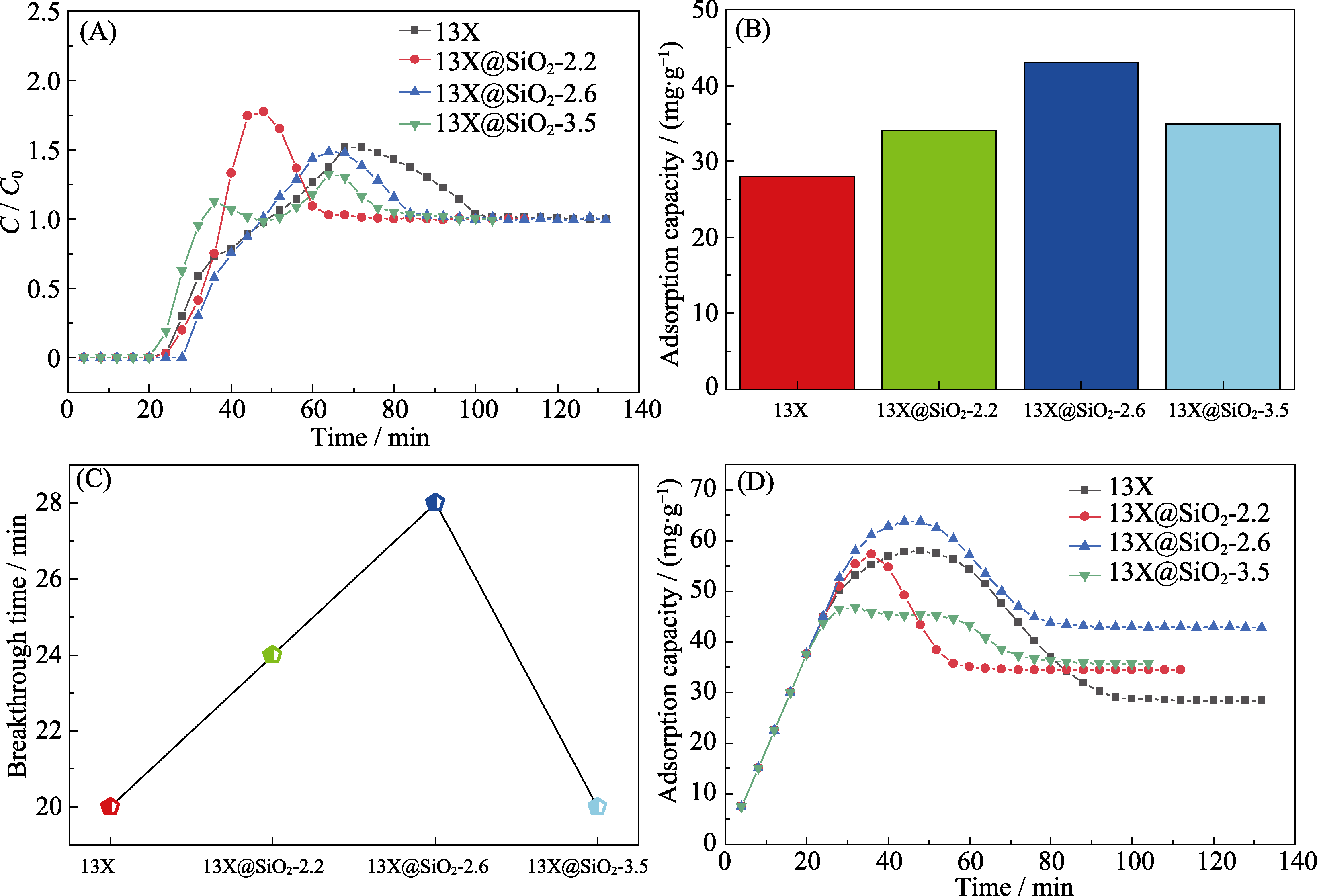
图5 不同吸附剂在相对湿度30%条件下对甲苯的吸附实验
Fig. 5 Adsorption of toluene on the different adsorbents under 30% relative humid conditions (A) Adsorption breakthrough curves; (B) Saturated adsorption capacity; (C) Comparison of the breakthrough time; (D) Cumulative adsorption capacities of different adsorbents of toluene

图6 不同吸附剂在相对湿度50%条件下对甲苯的吸附实验
Fig. 6 Adsorption of toluene on the different adsorbents under 50% relative humid conditions (A) Adsorption breakthrough curves; (B) Saturated adsorption capacity; (C) Comparison of the breakthrough time; (D) Cumulative adsorption capacity of different adsorbents of toluene
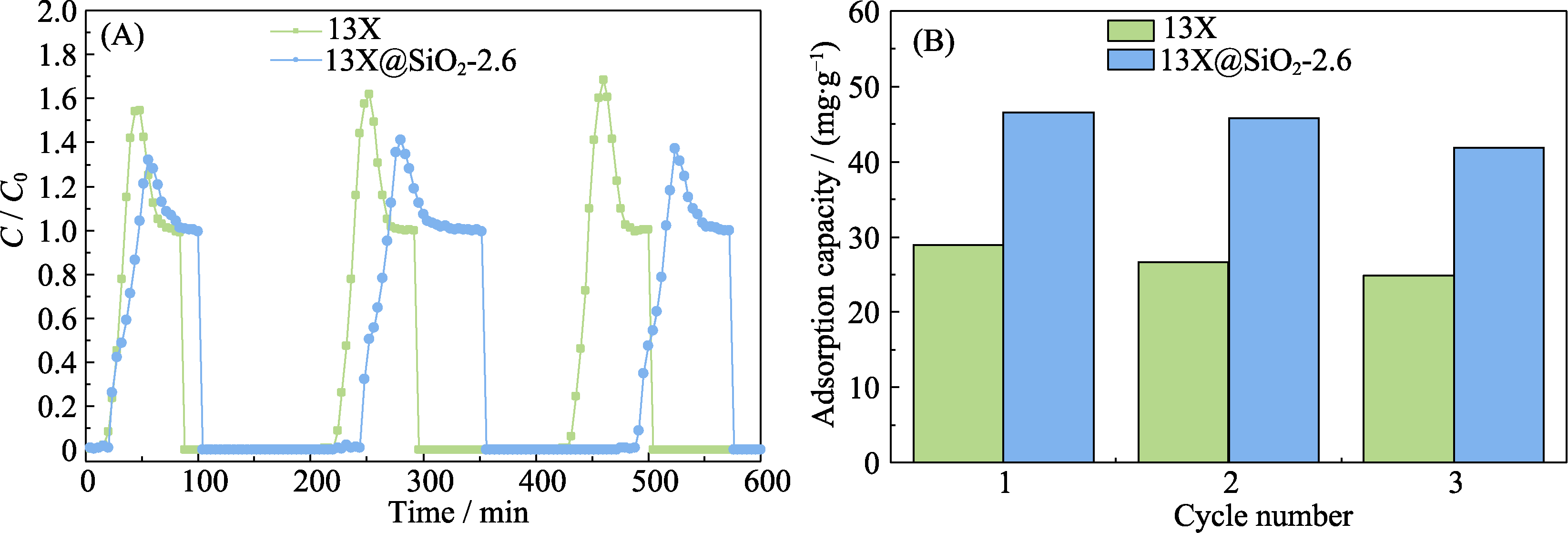
图S7 50% RH下, 13X和13X@SiO2-2.6的(A)三次吸附-脱附循环吸附穿透曲线与(B)饱和吸附量变化曲线
Fig. S7 (A) Adsorption of toluene on different adsorbents with triple adsorption-desorption cycle. Adsorption penetration curve and (B) saturated adsorption capacity under 50% relative humid conditions
| [1] |
KAMAL M S, RAZZAK S A, HOSSAIN M M. Catalytic oxidation of volatile organic compounds (VOCs)-a review. Atmospheric Environment, 2016, 140: 117.
DOI URL |
| [2] |
LI W B, WANG J X, GONG H. Catalytic combustion of VOCs on non-noble metal catalysts. Catalysis Today, 2009, 148(1/2): 81.
DOI URL |
| [3] |
DENG H, PAN T T, ZHANG Y, et al. Adsorptive removal of toluene and dichloromethane from humid exhaust on MFI, BEA and FAU zeolites: an experimental and theoretical study. Chemical Engineering Journal, 2020, 394: 124986.
DOI URL |
| [4] |
ZHANG X D, LV X T, SHI X Y, et al. Enhanced hydrophobic UiO-66 (University of Oslo 66) metal-organic framework with high capacity and selectivity for toluene capture from high humid air. Journal of Colloid and Interface Science, 2018, 539: 152.
DOI URL |
| [5] |
BAEK S, KIM J, IHM S. Design of dual functional adsorbent/ catalyst system for the control of VOC’s by using metal-loaded hydrophobic Y-zeolites. Catalysis Today, 2004, 93-95: 575.
DOI URL |
| [6] |
BENKHEDDA J, JAUBERT J N, BARTH D, et al. Experimental and modeled results describing the adsorption of toluene onto activated carbon. Journal of Chemical & Engineering Data, 2000, 45(4): 650.
DOI URL |
| [7] |
KARKA S, KODUKULA S, NANDURY S V, et al. Polyethylenimine- modified zeolite 13X for CO2 capture: adsorption and kinetic studies. ACS OMEGA, 2019, 4(15): 16441.
DOI URL |
| [8] |
HARLICK P J E, TEZEL F H. An experimental adsorbent screening study for CO2 removal from N2. Microporous and Mesoporous Materials, 2004, 76(1/2/3): 71.
DOI URL |
| [9] | SHEN C M, WOREK W M. Cosorption characteristics of solid adsorbents. International Journal of Heat & Mass Transfer, 1994, 37(14): 2123. |
| [10] |
LIU S, PENG Y, CHEN J J, et al. Engineering surface functional groups on mesoporous silica: towards a humidity-resistant hydrophobic adsorbent. Journal of Materials Chemistry A, 2018, 6(28): 13769.
DOI URL |
| [11] |
GUILLEMOT M, MIJOIN J, MIGNARD S, et al. Adsorption of tetrachloroethylene (PCE) in gas phase on zeolites of faujasite type: Influence of water vapour and of Si/Al ratio. Microporous and Mesoporous Materials, 2008, 111(1/2/3): 334.
DOI URL |
| [12] |
YIN T, MENG X, JIN L P, et al. Prepared hydrophobic Y zeolite for adsorbing toluene in humid environment. Microporous and Mesoporous Materials, 2020, 305: 110327.
DOI URL |
| [13] |
JIA L X, SUN X Y, YE X Q, et al. Core-shell composites of USY@mesosilica: synthesis and application in cracking heavy molecules with high liquid yield. Microporous and Mesoporous Materials, 2013, 176: 16-24.
DOI URL |
| [14] |
LI R N, XUE T S, LI Z, et al. Hierarchical structure ZSM-5/ SBA-15 composite with improved hydrophobicity for adsorption- desorption behavior of toluene. Chemical Engineering Journal, 2020, 392: 124861.
DOI URL |
| [15] |
LI R N, CHONG S J, ALTAF N, et al. Synthesis of ZSM-5/siliceous zeolite composites for improvement of hydrophobic adsorption of volatile organic compounds. Frontiers in chemistry, 2019, 7: 505.
DOI PMID |
| [16] |
LIU H J, WEI K Y, LONG C. Enhancing adsorption capacities of low-concentration VOCs under humid conditions using NaY@meso-SiO2 core-shell composite. Chemical Engineering Journal, 2022, 442: 136108.
DOI URL |
| [17] |
MIYAMOTO M, ONO S, KUSUKAMI K, et al. High water tolerance of a core-shell-structured Zeolite for CO2adsorptive separation under wet conditions. ChemSusChem, 2018, 11(11): 1756.
DOI URL |
| [18] |
LIU L Y, DU T, FANG X, et al. Preparation of hydrophobic zeolite 13X@SiO2and their adsorption properties of CO2and H2O. Advanced Materials Research, 2014, 1053: 311.
DOI URL |
| [19] |
LIU L Y, SINGH R, LI G, et al. Synthesis of hydrophobic zeolite X@SiO2 core-shell composites. Materials Chemistry and Physics, 2012, 133(2/3): 1144.
DOI URL |
| [20] | LI R N, XUE T S, BINGRE R, et al. Microporous zeolite@vertically aligned Mg-Al layered double hydroxide core@shell structures with improved hydrophobicity and toluene adsorption capacity under wet conditions. ACS Applied Materials & Interfaces, 2018, 10(41): 34834. |
| [21] | YI H, LI Z Y, REN C Q. Introduction to the standard relative humidity table for saturated salt solutions (international recommendation). The 7th National Conference on Humidity and Moisture and the 5th Conference on Gas-Humidity Sensitivity, Huhehaote, 1998: 70-72. |
| [22] |
LU S, LIU Q, HAN R, et al. Core-shell structured Y zeolite/ hydrophobic organic polymer with improved toluene adsorption capacity under dry and wet conditions. Chemical Engineering Journal, 2021, 409: 128194.
DOI URL |
| [23] |
LUO X, GUO J, CHANG P, et al. ZSM-5@MCM-41 composite porous materials with a core-shell structure: Adjustment of mesoporous orientation basing on interfacial electrostatic interactions and their application in selective aromatics transport. Separation and Purification Technology, 2020, 239: 116516.
DOI URL |
| [24] |
XIA H J, WANG J, CHEN G, et al. One-pot synthesis of SiO2@SiO2 core-shell microspheres with controllable mesopore size as a new stationary phase for fast HPLC separation of alkyl benzenes and β-agonists. Microchimica Acta, 2019, 186(2): 125.
DOI |
| [25] | 罗智恒. 疏水性13X沸石的制备及其在H2O/CO2吸附分离中的应用研究. 沈阳: 东北大学硕士学位论文, 2017. |
| [26] |
VELLINGIRI K, KUMAR P, DEEP A, et al. Metal-organic frameworks for the adsorption of gaseous toluene under ambient temperature and pressure. Chemical Engineering Journal, 2017, 307: 1116.
DOI URL |
| [27] |
KRAUS M, TROMMLER U, HOLZER F, et al. Competing adsorption of toluene and water on various zeolites. Chemical Engineering Journal, 2018, 351: 356.
DOI URL |
| [28] | LEE K M, KIM N S, NUMAN M, et al. Post synthetic modification of zeolite internal surface for sustainable capture of volatile organic compounds under humid conditions. ACS Applied Materials & Interfaces, 2021, 13(45): 53925. |
| [29] |
JACOBS J H, DEERING C E, LESAGE K L, et al. Rapid cycling thermal swing adsorption apparatus: commissioning and data analyses for water adsorption of zeolites 4A and 13X over 2000 cycles. Industrial & Engineering Chemistry Research, 2021, 60(19): 7487.
DOI URL |
| [30] | FISCHER F, LUTZ W, BUHL J C, et al. Insights into the hydrothermal stability of zeolite 13X. Microporous and Mesoporous Materials, 2018, 262: 258 |
| [1] | 魏建文, 张丽娟, 耿琳琳, 李誉, 廖雷, 王敦球. 以ZSM-5/MCM-48为载体制备新型高容量CO2吸附剂的性能及机理研究[J]. 无机材料学报, 2025, 40(7): 833-839. |
| [2] | 江宗玉, 黄红花, 清江, 王红宁, 姚超, 陈若愚. 铝离子掺杂MIL-101(Cr)的制备及其VOCs吸附性能研究[J]. 无机材料学报, 2025, 40(7): 747-753. |
| [3] | 陈义, 邱海鹏, 陈明伟, 徐昊, 崔恒. SiC/SiC复合材料基体硼改性方法及其力学性能研究[J]. 无机材料学报, 2025, 40(5): 504-510. |
| [4] | 洪培萍, 梁龙, 吴炼, 马颖康, 庞浩. ZIF-67结构调控及其对盐酸金霉素的吸附性能研究[J]. 无机材料学报, 2025, 40(4): 388-396. |
| [5] | 穆爽, 马沁, 张禹, 沈旭, 杨金山, 董绍明. Yb2Si2O7改性SiC/SiC复合材料的氧化行为研究[J]. 无机材料学报, 2025, 40(3): 323-328. |
| [6] | 杨舒琪, 杨存国, 牛慧祝, 石唯一, 舒珂维. GeP3/科琴黑复合材料作为钠离子电池高性能负极材料[J]. 无机材料学报, 2025, 40(3): 329-336. |
| [7] | 谌广昌, 段小明, 朱金荣, 龚情, 蔡德龙, 李宇航, 杨东雷, 陈彪, 李新民, 邓旭东, 余瑾, 刘博雅, 何培刚, 贾德昌, 周玉. 直升机特定结构先进陶瓷材料研究进展与应用展望[J]. 无机材料学报, 2025, 40(3): 225-244. |
| [8] | 栾新刚, 何典蔚, 涂建勇, 成来飞. 2D平纹和3D针刺C/SiC复合材料的低速冲击破坏行为和失效机理[J]. 无机材料学报, 2025, 40(2): 205-214. |
| [9] | 王文婷, 徐敬军, 马科, 李美栓, 李兴超, 李同起. 原位反应/热压合成Ti2AlC-20TiB2复合材料在1000~1300 ℃空气中的高温氧化行为[J]. 无机材料学报, 2025, 40(1): 31-38. |
| [10] | 全文心, 余艺平, 方冰, 李伟, 王松. 管状C/SiC复合材料高温空气氧化行为与宏细观建模研究[J]. 无机材料学报, 2024, 39(8): 920-928. |
| [11] | 何思哲, 王俊舟, 张勇, 费嘉维, 吴爱民, 陈意峰, 李强, 周晟, 黄昊. 高频低损耗的Fe/亚微米FeNi软磁复合材料[J]. 无机材料学报, 2024, 39(8): 871-878. |
| [12] | 孙海洋, 季伟, 王为民, 傅正义. TiB-Ti周期序构复合材料设计、制备及性能研究[J]. 无机材料学报, 2024, 39(6): 662-670. |
| [13] | 吴晓晨, 郑瑞晓, 李露, 马浩林, 赵培航, 马朝利. SiCf/SiC陶瓷基复合材料高温环境损伤原位监测研究进展[J]. 无机材料学报, 2024, 39(6): 609-622. |
| [14] | 粟毅, 史扬帆, 贾成兰, 迟蓬涛, 高扬, 马青松, 陈思安. 浆料浸渍辅助PIP工艺制备C/HfC-SiC复合材料的微观结构及性能研究[J]. 无机材料学报, 2024, 39(6): 726-732. |
| [15] | 赵日达, 汤素芳. 多孔碳陶瓷化改进反应熔渗法制备陶瓷基复合材料研究进展[J]. 无机材料学报, 2024, 39(6): 623-633. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||