无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 519-526.DOI: 10.15541/jim20250265 CSTR: 32189.14.10.15541/jim20250265
所属专题: 【能源环境】金属有机框架材料MOF(202512)
朱开煌1,2( ), 杨世杰2,3, 李欣格2,3, 宋贯卿2,3, 史淦升2, 王焱2, 任小孟4, 陆遥1, 徐新宏4(
), 杨世杰2,3, 李欣格2,3, 宋贯卿2,3, 史淦升2, 王焱2, 任小孟4, 陆遥1, 徐新宏4( ), 孙静2(
), 孙静2( )
)
收稿日期:2025-06-23
修回日期:2025-09-04
出版日期:2026-04-20
网络出版日期:2025-09-11
通讯作者:
孙 静, 研究员. E-mail: jingsun@mail.sic.ac.cn;作者简介:朱开煌(2000-), 男, 硕士研究生. E-mail: ZHUkaihuang_11@outlook.com
ZHU Kaihuang1,2( ), YANG Shijie2,3, LI Xinge2,3, SONG Guanqing2,3, SHI Gansheng2, WANG Yan2, REN Xiaomeng4, LU Yao1, XU Xinhong4(
), YANG Shijie2,3, LI Xinge2,3, SONG Guanqing2,3, SHI Gansheng2, WANG Yan2, REN Xiaomeng4, LU Yao1, XU Xinhong4( ), SUN Jing2(
), SUN Jing2( )
)
Received:2025-06-23
Revised:2025-09-04
Published:2026-04-20
Online:2025-09-11
Contact:
SUN Jing, professor. E-mail: jingsun@mail.sic.ac.cn;About author:ZHU Kaihuang (2000-), male, Master candidate. E-mail: ZHUkaihuang_11@outlook.com
摘要:
挥发性有机化合物(VOCs), 尤其是芳香烃类物质如甲苯, 由于其高挥发性和生物毒性, 严重威胁环境与人类健康。传统的金属有机框架(MOFs)材料多以微孔结构为主, 但在吸附容量与分子传质之间存在矛盾, 这促使研究者探索更优的材料体系。本研究以UiO-66为主体, 复合氧化石墨烯(GO)进行改性, 构建了GO复合的金属有机框架凝胶(MOGs)。GO的π共轭结构增强了与甲苯分子的π-π相互作用, 丰富的含氧官能团则促进了与金属节点的配位, 形成更多Lewis酸性位点, 协同增强了金属-π相互作用。实验结果显示, UG-1样品(GO与ZrCl4质量比为1 : 100)在动态穿透实验中的平衡吸附容量达77.4 mg/g, 静态吸附实验中的饱和吸附容量高达1245.5 mg/g, 优于UiO66 MOF和UiO66 MOG, 表现出卓越的吸附性能。本工作揭示了GO改性对材料孔结构及主-客体相互作用的多重调控机理, 为设计高效可再生VOCs吸附剂提供了新的理论基础和实践支撑。
中图分类号:
朱开煌, 杨世杰, 李欣格, 宋贯卿, 史淦升, 王焱, 任小孟, 陆遥, 徐新宏, 孙静. 基于UiO-66骨架的氧化石墨烯改性金属有机框架凝胶的制备及其对甲苯的高效吸附性能[J]. 无机材料学报, 2026, 41(4): 519-526.
ZHU Kaihuang, YANG Shijie, LI Xinge, SONG Guanqing, SHI Gansheng, WANG Yan, REN Xiaomeng, LU Yao, XU Xinhong, SUN Jing. Graphene Oxide Modified UiO-66 Based Metal Organic Framework Gel: Preparation and Efficient Toluene Adsorption Performance[J]. Journal of Inorganic Materials, 2026, 41(4): 519-526.
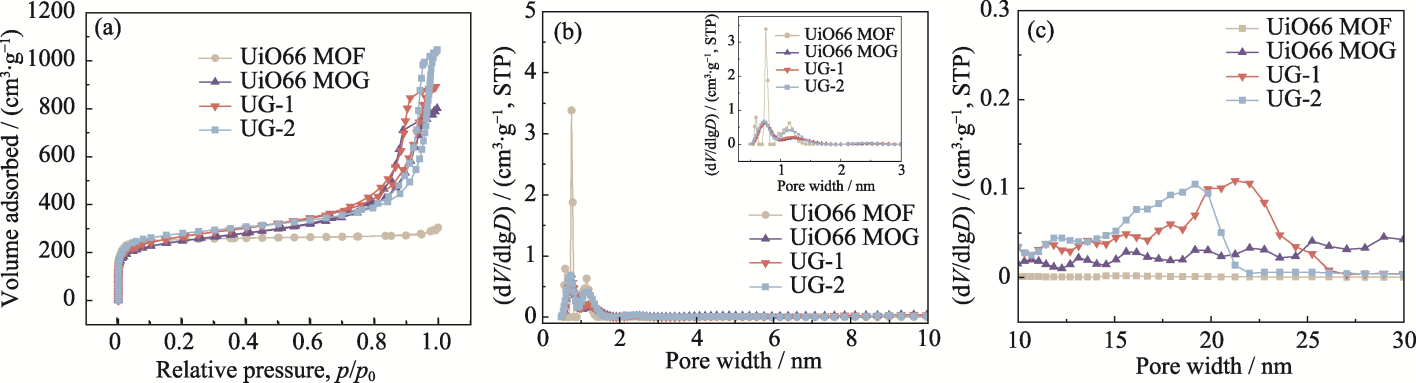
图3 UiO66 MOF、UiO66 MOG、UG-1和UG-2的(a) N2吸-脱附等温曲线、(b) 0~10 nm的孔径分布图(插图为0.5~3 nm的孔径放大图)和(c) 10~30 nm的孔径分布图
Fig. 3 (a) N2 adsorption-desorption isotherms, and pore size distribution curves in the ranges of (b) 0-10 nm and (c) 10-30 nm of UiO66 MOF, UiO66 MOG, UG-1, and UG-2 with inset in (b) showing magnified view for the 0.5-3 nm range
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) |
|---|---|---|---|
| UiO66 MOF | 1020.9 | 945.2 | 75.7 |
| UiO66 MOG | 909.8 | 511.1 | 398.7 |
| UG-1 | 973.8 | 536.8 | 437.0 |
| UG-2 | 1062.4 | 749.5 | 312.9 |
表1 UiO66 MOF、UiO66 MOG和UG-x的比表面积以及孔结构参数
Table 1 Specific surface area and pore structure parameters of UiO66 MOF, UiO66 MOG and UG-x
| Sample | SBET/(m2·g-1) | Smicro/(m2·g-1) | Smeso/(m2·g-1) |
|---|---|---|---|
| UiO66 MOF | 1020.9 | 945.2 | 75.7 |
| UiO66 MOG | 909.8 | 511.1 | 398.7 |
| UG-1 | 973.8 | 536.8 | 437.0 |
| UG-2 | 1062.4 | 749.5 | 312.9 |
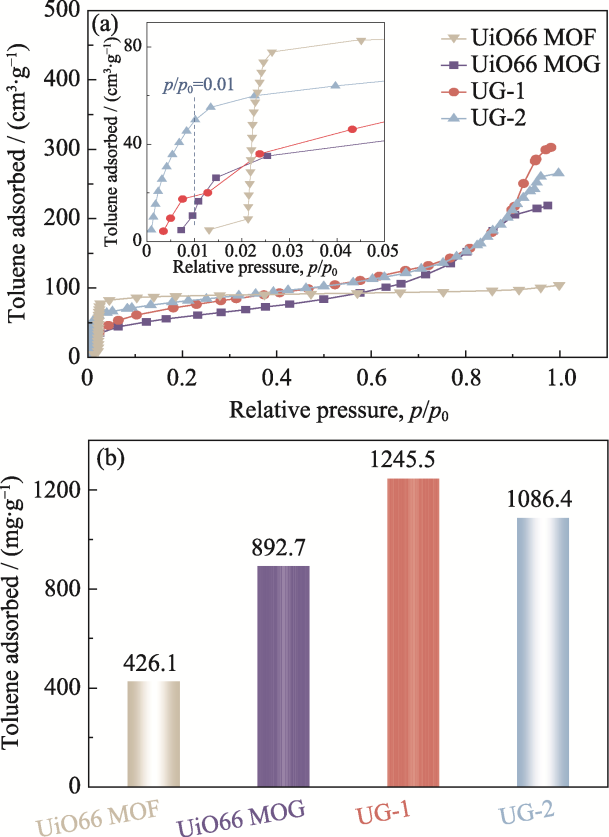
图4 UiO66 MOF、UiO66 MOG、UG-1和UG-2的(a)甲苯静态吸附曲线(插图是吸附剂在低相对压力区间p/p0=0~0.05的甲苯静态吸附曲线)和(b)甲苯饱和吸附容量
Fig. 4 (a) Toluene vapor adsorption isotherms and (b) saturated toluene adsorption capacities of UiO66 MOF, UiO66 MOG, UG-1, and UG-2 with inset in (a) showing adsorption isotherms in a low-pressure region p/p0=0-0.05
| Adsorbent | p/p0 | Temperature/K | q/(mg·g-1) |
|---|---|---|---|
| AC[ | 1.0 | 298 | 530.4 |
| Silica gel[ | 1.0 | 298 | 437.4 |
| UiO-66-NH2@ABP[ | 0.9 | 298 | 178.9 |
| MIL-125-NH2[ | 0.94 | 298 | 293 |
| MIL-100(Fe)_A2[ | 1.0 | 298 | 523.3 |
| HKUST-1[ | 0.9 | 298 | 516 |
| MSN-100[ | 0.94 | 298 | 461 |
| UG-1 | 1.0 | 298 | 1245.5 |
表2 不同吸附剂的甲苯饱和吸附容量[2,18 -22]
Table 2 Toluene saturation adsorption capacities of various adsorbents[2,18 -22]
| Adsorbent | p/p0 | Temperature/K | q/(mg·g-1) |
|---|---|---|---|
| AC[ | 1.0 | 298 | 530.4 |
| Silica gel[ | 1.0 | 298 | 437.4 |
| UiO-66-NH2@ABP[ | 0.9 | 298 | 178.9 |
| MIL-125-NH2[ | 0.94 | 298 | 293 |
| MIL-100(Fe)_A2[ | 1.0 | 298 | 523.3 |
| HKUST-1[ | 0.9 | 298 | 516 |
| MSN-100[ | 0.94 | 298 | 461 |
| UG-1 | 1.0 | 298 | 1245.5 |
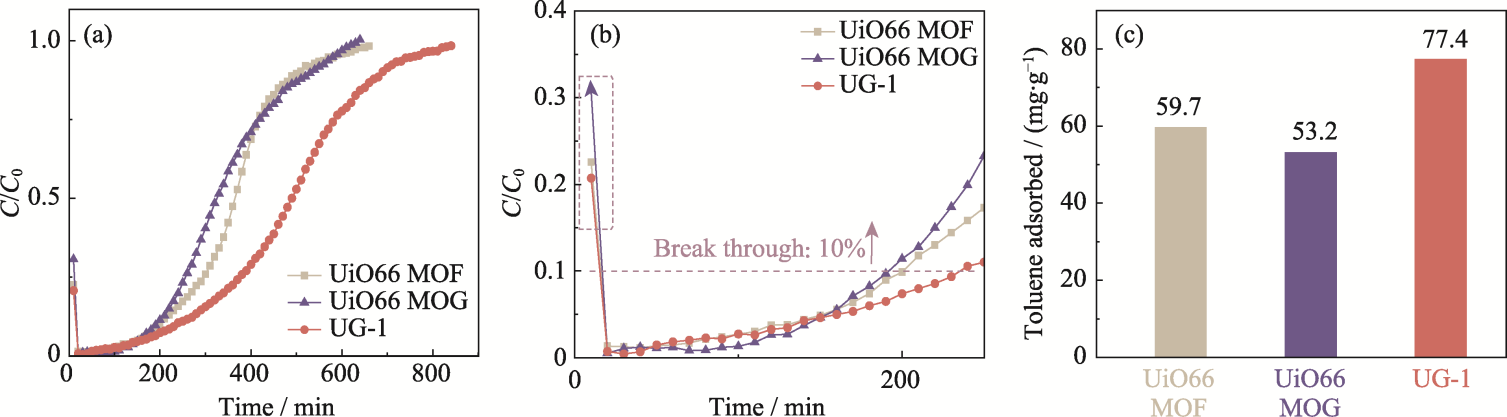
图5 UiO66 MOF、UiO66 MOG、UG-1 (a) 800和(b) 250 min内的甲苯动态穿透曲线和(c)甲苯平衡吸附容量
Fig. 5 Dynamic breakthrough curves for toluene adsorption within (a) 800 and (b) 250 min, and (c) equilibrium toluene adsorption capacities of UiO66 MOF, UiO66 MOG and UG-1
| Sample | tb/min | te/min | q/(mg·g-1) |
|---|---|---|---|
| UiO66 MOF | 200 | 580 | 59.7 |
| UiO66 MOG | 200 | 590 | 53.2 |
| UG-1 | 240 | 750 | 77.4 |
表3 UiO66 MOF、UiO66 MOG和UG-1的突破时间、平衡时间以及平衡吸附量
Table 3 Breakthrough time, equilibrium time and equilibrium adsorption capacities of UiO66 MOF, UiO66 MOG and UG-1
| Sample | tb/min | te/min | q/(mg·g-1) |
|---|---|---|---|
| UiO66 MOF | 200 | 580 | 59.7 |
| UiO66 MOG | 200 | 590 | 53.2 |
| UG-1 | 240 | 750 | 77.4 |
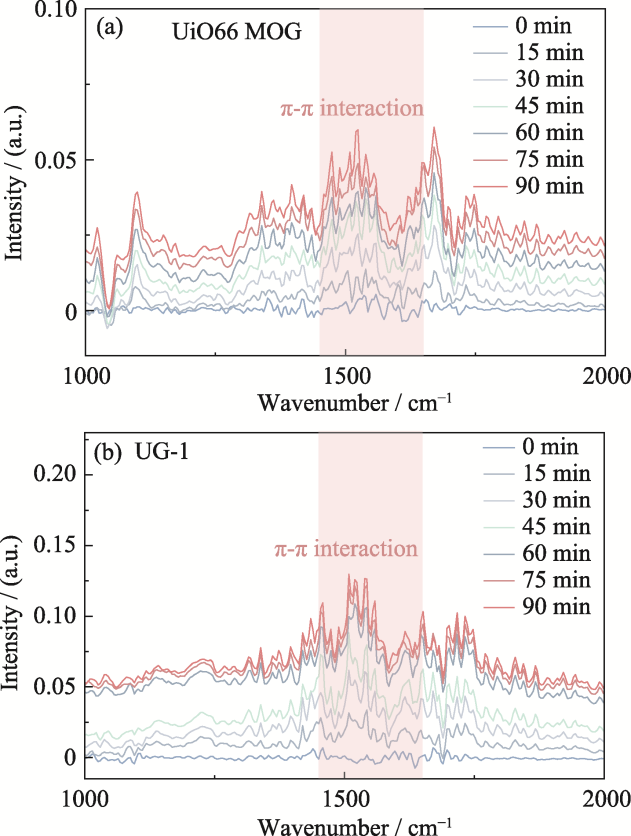
图7 (a) UiO66 MOG和(b) UG-1对甲苯吸附的原位DRIFTS图谱
Fig. 7 In-situ DRIFTS spectra of toluene adsorption for (a) UiO66 MOG and (b) UG-1 Colorful figures are available on website
| [1] |
QU F, ZHU L Z, YANG K. Adsorption behaviors of volatile organic compounds (VOCs) on porous clay heterostructures (PCH). Journal of Hazardous Materials, 2009, 170(1): 7.
DOI PMID |
| [2] |
KIM B, LEE Y R, KIM H Y, et al. Adsorption of volatile organic compounds over MIL-125-NH2. Polyhedron, 2018, 154: 343.
DOI URL |
| [3] |
ZHANG X Y, GAO B, CREAMER A E, et al. Adsorption of VOCs onto engineered carbon materials: a review. Journal of Hazardous Materials, 2017, 338: 102.
DOI PMID |
| [4] |
LI X Q, ZHANG L, YANG Z Q, et al. Adsorption materials for volatile organic compounds (VOCs) and the key factors for VOCs adsorption process: a review. Separation and Purification Technology, 2020, 235: 116213.
DOI URL |
| [5] |
YANG S J, WANG X, SONG G Q, et al. Ti-O-Mo bond-bridged PMA@MIL-125-NH2 photocatalyst for gas acetone photocatalytic degradation. Applied Catalysis B: Environment and Energy, 2025, 367: 125112.
DOI URL |
| [6] |
FU Y, ZHANG P Y, WANG X J. Feasible defect and hydrophobic modification of MIL-100(Cr) for efficient toluene removal from air. Chemical Engineering Journal, 2024, 482: 149163.
DOI URL |
| [7] |
VALENZANO L, CIVALLERI B, CHAVAN S, et al. Disclosing the complex structure of UiO-66 metal organic framework: a synergic combination of experiment and theory. Chemistry of Materials, 2011, 23(7): 1700.
DOI URL |
| [8] |
ZHANG X D, LV X T, SHI X Y, et al. Enhanced hydrophobic UiO-66 (University of Oslo 66) metal-organic framework with high capacity and selectivity for toluene capture from high humid air. Journal of Colloid and Interface Science, 2019, 539: 152.
DOI PMID |
| [9] |
LI Y J, MIAO J P, SUN X J, et al. Mechanochemical synthesis of Cu-BTC@GO with enhanced water stability and toluene adsorption capacity. Chemical Engineering Journal, 2016, 298: 191.
DOI URL |
| [10] |
HOU J W, SAPNIK A F, BENNETT T D. Metal-organic framework gels and monoliths. Chemical Science, 2019, 11(2): 310.
DOI URL |
| [11] |
CHEN L, WANG X, SHI G S, et al. The regulation of Lewis acid/ basic sites in NaFe bimetal MOXs for the controllable photocatalytic degradation of electron-rich/deficient VOCs. Applied Catalysis B: Environmental, 2023, 334: 122850.
DOI URL |
| [12] |
QIN H J, SUN J W, YANG X B, et al. Defective UiO-66 metal- organic gels for optimizing gaseous toluene capture. Journal of Colloid and Interface Science, 2024, 655: 23.
DOI URL |
| [13] |
TIAN T, ZENG Z X, VULPE D, et al. A Sol-Gel monolithic metal-organic framework with enhanced methane uptake. Nature Materials, 2018, 17(2): 174.
DOI PMID |
| [14] |
YU J J, WANG X, CHEN L, et al. Enhanced adsorption and visible-light photocatalytic degradation of toluene by CQDs/UiO- 66 MOG with hierarchical pores. Chemical Engineering Journal, 2022, 435: 135033.
DOI URL |
| [15] |
YOST B T, GIBBONS B, WILSON A, et al. Vibrational spectroscopy investigation of defects in Zr- and Hf-UiO-66. RSC Advances, 2022, 12(35): 22440.
DOI PMID |
| [16] |
JI W L, LI W W, WANG Y, et al. Zr-doped MIL-101(Fe)/ graphene oxide nanocomposites: an efficient and water-stable MOF-based adsorbent for As(V) adsorption in aqueous solution. Separation and Purification Technology, 2024, 339: 126681.
DOI URL |
| [17] |
CONNOLLY B M, ARAGONES-ANGLADA M, GANDARA- LOE J, et al. Tuning porosity in macroscopic monolithic metal- organic frameworks for exceptional natural gas storage. Nature Communications, 2019, 10: 2345.
DOI |
| [18] |
SUI H, LIU H X, AN P, et al. Application of silica gel in removing high concentrations toluene vapor by adsorption and desorption process. Journal of the Taiwan Institute of Chemical Engineers, 2017, 74: 218.
DOI URL |
| [19] |
XIAO Y T, WU Y, SUN C, et al. UiO-66-NH2 incorporated nanofibrous membranes by direct electrospinning/in-situ growth for toluene adsorption. Journal of Environmental Chemical Engineering, 2025, 13(1): 115198.
DOI URL |
| [20] |
DUAN C X, YU Y, YANG P F, et al. Engineering new defects in MIL-100(Fe) via a mixed-ligand approach to effect enhanced volatile organic compound adsorption capacity. Industrial & Engineering Chemistry Research, 2020, 59(2): 774.
DOI URL |
| [21] | XU F, XIAN S K, XIA Q B, et al. Effect of textural properties on the adsorption and desorption of toluene on the metal-organic frameworks HKUST-1 and MIL-101. Adsorption Science & Technology, 2013, 31(4): 325. |
| [22] |
HU Q, DOU B J, TIAN H, et al. Mesoporous silicalite-1 nanospheres and their properties of adsorption and hydrophobicity. Microporous and Mesoporous Materials, 2010, 129(1/2): 30.
DOI URL |
| [23] |
DU Y K, CHEN H Y, XU X, et al. Surface modification of biomass derived toluene adsorbent: hierarchically porous characterization and heteroatom doped effect. Microporous and Mesoporous Materials, 2020, 293: 109831.
DOI URL |
| [24] |
ALIVAND M S, TEHRANI N H M H, ASKARIEH M, et al. Defect engineering-induced porosity in graphene quantum dots embedded metal-organic frameworks for enhanced benzene and toluene adsorption. Journal of Hazardous Materials, 2021, 416: 125973.
DOI URL |
| [25] |
CAO J W, LI Y X, MA X B, et al. Constructing binuclear sites to modulate the charge distribution of MIL-101 for enhanced toluene adsorption performance: experimental and theoretical studies. Separation and Purification Technology, 2025, 354: 129400.
DOI URL |
| [26] |
CHEN L, WANG X, RAO Z P, et al. One-pot synthesis of the MIL-100 (Fe) MOF/MOX homojunctions with tunable hierarchical pores for the photocatalytic removal of BTXS. Applied Catalysis B: Environmental, 2022, 303: 120885.
DOI URL |
| [27] |
YE L L, CHEN X, CHEN Y, et al. Improvement of membrane- forming ability and separation performance of hydroxylated BN membrane by GO via π-π interaction. Desalination, 2023, 565: 116824.
DOI URL |
| [1] | 王浩宇, 柯学, 关世伟, 钱仕, 刘宣勇. Fe掺杂Ti-MOFs用于牙周炎抗菌声动力治疗[J]. 无机材料学报, 2026, 41(4): 527-535. |
| [2] | 范雨竹, 王媛, 王林燕, 向美玲, 鄢雨婷, 黎本慧, 李敏, 文志东, 王海超, 陈永福, 邱会东, 赵波, 周成裕. 氧化石墨烯基吸附材料去除水体中Pb(II): 制备、性能及机理[J]. 无机材料学报, 2026, 41(1): 12-26. |
| [3] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [4] | 朱文杰, 唐璐, 陆继长, 刘江平, 罗永明. 钙钛矿型氧化物催化氧化挥发性有机化合物的研究进展[J]. 无机材料学报, 2025, 40(7): 735-746. |
| [5] | 江宗玉, 黄红花, 清江, 王红宁, 姚超, 陈若愚. 铝离子掺杂MIL-101(Cr)的制备及其VOCs吸附性能研究[J]. 无机材料学报, 2025, 40(7): 747-753. |
| [6] | 王悦, 王欣, 于显利. 室温铁磁性还原氧化石墨烯基全碳膜[J]. 无机材料学报, 2025, 40(3): 305-313. |
| [7] | 瞿牡静, 张淑兰, 朱梦梦, 丁浩杰, 段嘉欣, 代恒龙, 周国红, 李会利. CsPbBr3@MIL-53纳米复合荧光粉的合成、性能及其白光LEDs应用[J]. 无机材料学报, 2024, 39(9): 1035-1043. |
| [8] | 董怡曼, 谭占鳌. 宽带隙钙钛矿基二端叠层太阳电池复合层的研究进展[J]. 无机材料学报, 2023, 38(9): 1031-1043. |
| [9] | 张笑宇, 刘永盛, 李然, 李耀刚, 张青红, 侯成义, 李克睿, 王宏志. 基于Cu3(HHTP)2薄膜的离子液体电致变色电极[J]. 无机材料学报, 2022, 37(8): 883-890. |
| [10] | 洪佳辉, 马冉, 仵云超, 文涛, 艾玥洁. MOFs自牺牲模板法制备CoNx/g-C3N4纳米材料用作高效光催化还原U(VI)[J]. 无机材料学报, 2022, 37(7): 741-749. |
| [11] | 孙铭, 邵溥真, 孙凯, 黄建华, 张强, 修子扬, 肖海英, 武高辉. RGO/Al复合材料界面性质第一性原理研究[J]. 无机材料学报, 2022, 37(6): 651-659. |
| [12] | 董淑蕊, 赵笛, 赵静, 金万勤. 离子化氨基酸对氧化石墨烯膜渗透汽化过程中水选择性渗透的影响[J]. 无机材料学报, 2022, 37(4): 387-394. |
| [13] | 王晶, 徐守冬, 卢中华, 赵壮壮, 陈良, 张鼎, 郭春丽. 钠离子电池中空结构CoSe2/C负极材料的制备及储钠性能研究[J]. 无机材料学报, 2022, 37(12): 1344-1350. |
| [14] | 李豪, 唐志红, 卓尚军, 钱荣. 基于ZIF8/rGO的高性能NO2室温传感器[J]. 无机材料学报, 2021, 36(12): 1277-1282. |
| [15] | 王玉伟, 陈佳杰, 田正芳, 朱敏, 朱钰方. 卟啉基金属有机框架负载高铁酸钾: 光-化学动力学联合治疗肿瘤性能研究[J]. 无机材料学报, 2021, 36(12): 1305-1315. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||