
Journal of Inorganic Materials ›› 2017, Vol. 32 ›› Issue (9): 997-1003.DOI: 10.15541/jim20170001
• Orginal Article • Previous Articles Next Articles
NAN Hui1, WANG Wen-Li2, HAN Jian-Hua1, YIN Xue-Wen1, ZHOU Yu1, ZHAO Xiao-Chong3, LIN Hong1
Received:2017-01-03
Published:2017-09-30
Online:2017-08-29
About author:NAN Hui (1984–), male, candidate of PhD. E-mail: 865615800@qq.com
Supported by:CLC Number:
NAN Hui, WANG Wen-Li, HAN Jian-Hua, YIN Xue-Wen, ZHOU Yu, ZHAO Xiao-Chong, LIN Hong. Low-cost Preparation of Graphene Papers from Chemical Reduction with FeI2/Ni2+ for Conductivity and Catalytic Propert[J]. Journal of Inorganic Materials, 2017, 32(9): 997-1003.
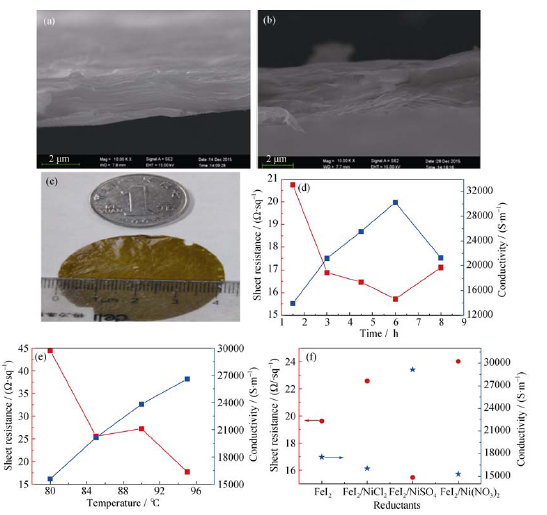
Fig. 1 Cross section SEM images of the original GO film (a) and RGO films (b) after reduction; Sheet resistance and conductivity of as-prepared RGO (c) reduced with different time (d), temperature (e), reductant (f)
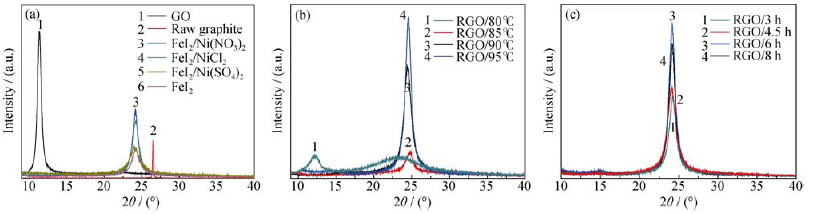
Fig. 2 XRD patterns of raw graphite, GO, and RGO reduced with different reductant (a); by FeI2/Ni(NO3)2 with different temperatures (b) and for different time (c)
| Sample | Reductant | ID/(a.u.) | IG/(a.u.) | ID/IG |
|---|---|---|---|---|
| GO | 499.7 | 462.5 | 1.08 | |
| RGO | FeI2 | 377.3 | 237.3 | 1.59 |
| RGO | FeI2/NiCl2 | 402.2 | 251.7 | 1.60 |
| RGO | FeI2/Ni(NO3)2 | 389.6 | 250.2 | 1.66 |
| RGO | FeI2/NiSO4 | 564.8 | 389.2 | 1.45 |
Table 1 ID/IG of GO and RGO reduced with different solutions
| Sample | Reductant | ID/(a.u.) | IG/(a.u.) | ID/IG |
|---|---|---|---|---|
| GO | 499.7 | 462.5 | 1.08 | |
| RGO | FeI2 | 377.3 | 237.3 | 1.59 |
| RGO | FeI2/NiCl2 | 402.2 | 251.7 | 1.60 |
| RGO | FeI2/Ni(NO3)2 | 389.6 | 250.2 | 1.66 |
| RGO | FeI2/NiSO4 | 564.8 | 389.2 | 1.45 |
| [1] | ROY-MAYHEW J D, BOZYM D J, PUNCKT C,et al.Functionalized graphene as a catalytic counter electrode in dye-sensitized solar cell. ACS Nano, 2010, 4(10): 6203-6211. |
| [2] | HU Y H, WANG H, HU B.Thinnest two-dimensional nano material-graphene for solar energ.Chemsuschem, 2010, 3(7): 782-796. |
| [3] | XU Z W, LI H J, CAO G X,et al.Synthesis of hybrid graphene carbon-coated nanocatalyst. J. Mater. Chem., 2010, 20(38): 8230-8232. |
| [4] | VIET H P, TRAN V C, HUR S H,et al.Chemical fictionalization of graphene sheets by solvothermal reduction of a graphene oxide suspension in nmethyl-2-pyrrolidon. J. Mater. Chem., 2011, 21(10): 3371-3377. |
| [5] | ZHOU X F, WANG F, ZHU Y M, et al. Graphene modified LiFePO4 cathode materials for high power lithium ion batteries.J. Mater. Chem., 2011, 21(10): 3353-3358. |
| [6] | HIRALAL P, IMAIZUMI S, UNALAN H E,et al. Nanomaterial enhanced all-solid flexible zinc-carbon batterie. ACS Nano, 2010, 4(5): 2730-2734. |
| [7] | SU F Y, YOU C H, HE YB, et al.Pore diameter control in anodic titanium and aluminums oxides.J. Mater. Chem., 2010, 20(2): 9644-9650. |
| [8] | ZHAO X, TIAN H, ZHU M Y,et al.Carbon nanosheets as the electrode material in superapacitor. J. Power Sources, 2009, 194(2): 1208-1212. |
| [9] | BAE J, SONG M K, PARK Y J,et al.Fiber super capacitors made of nanowire-fiber hybrid structures for wearable/flexible energy storag. Angew Chem. Int. Edit, 2011, 50(7): 1683-1687. |
| [10] | LI Z P, WANG J Q, LIU X H,et al.Electro static layer-by-layer self-assembly multilayer films based on graphene and manganese dioxide sheets as novel electrode materials for super capacitor. J. Mater. Chem., 2011, 21(10): 3397-3403. |
| [11] | GAO Y J, MA D, WANG C L, et al.Reduced graphene oxide as a catalyst for hydrogenation of nitrobenzene at room temperature.Chem. Commun., 2011, 47(8): 2432-2434. |
| [12] | MOHANTY N, NAGARAJA A, ARMESTO J,et al.High-through put ultrafast synthesis of solution-dispersed graphene via facile hydride chemistry. Small, 2010, 6: 226-231. |
| [13] | ZHANG H, LI Y, WANG Y, et al. P25-graphene composite as a high performance photocatalyst. ACS Nano, 2010, 4: 380-386. |
| [14] | LI Q, GUO B, YU J,et al.Highly efficient visible-light-driven photo catalytic hydrogen production of CdS-cluster-decorated grapheme nanosheet. J. Am. Chem. Soc., 2011, 133: 10878-10884. |
| [15] | XIANG Q, YU J, JARONIEC M.Graphene-based semiconductor photo catalyst.. Chem. Soc. Rev., 2012, 41: 782-796. |
| [16] | ZHAO X C, LIN H, LI J F,et al.Low-cost preparation of a conductive and catalytic grapheme film from chemical reduction with AlI3. Carbon, 2012, 50: 3497-3502. |
| [17] | KIM K S, ZHAO Y, JANG H,et al.Large-scale pattern growth of graphene films for stretchable transparent electrode. Nature, 2009, 457(7230): 706-710. |
| [18] | SHEN J H, ZHU Y H, CHEN C,et al.Facile preparation and up conversion luminescence of graphene quantum dot. Chem. Common., 2011, 47(9): 2580-2582. |
| [19] | JIANG B J, TIAN C G, WANG L,et al.Facile fabrication of high quality graphene from expands able graphite: simultaneous exfoliation and reductio. Chem. Common., 2010, 46(27): 4920-4922. |
| [20] | ZUBAREV D Y, YOU X Q, CLEAN J,et al.Patterns of local aromaticity in grapheme ox radical. J. Mater. Chem., 2011, 21(10): 3404-3409. |
| [21] | DREYER D R, MURALI S, ZHU Y W,et al.Reduction of graphite oxide using alcohol. J. Mater. Chem., 2011, 21(10): 3443-3447. |
| [22] | HE F A, FAN J T, MA D,et al.The attachment of Fe3O4 nanoparticles to graphene oxide by covalent bonding. Carbon, 2010, 48(11): 3139-3144. |
| [23] | PEI S F, ZHAO J P, DU J H,et al.Direct reduction of graphene oxide films into highly conductive and flexible grapheme films by hydrohalicacid. Carbon, 2010, 48(15): 4466-4474. |
| [24] | LIU C Y, HAO F, ZHAO X C,et al.Low temperature reduction of free-standing graphene oxide papers with metal iodides for ultrahigh bulk conductivit. Scientific Reports, 2014, 4: 3965. |
| [25] | SHEN J F, YAN B, SHI M,et al.One step hydrothermal synthesis of TiO2-reduced graphene oxide sheets. J. Mater. Chem., 2011, 21(10): 3415-3421. |
| [26] | BAGRI A, MATTEVI C, ACIK M,et al.Structural evolution during the reduction of chemically derived graphene oxid. Nat. Chem., 2010, 2(7): 581-587. |
| [27] | CANCADO L G, JORIO A, FERREIRA E H,et al.Quantizing defects in graphene via Raman spectroscopy at different excitation energies. Nano Letters, 2011, 11(8): 3190-3196. |
| [28] | PANSARE S V, PAUL E K.Organ catalytic symmetric direct vinylogous aldo lreactions of gamma-crotonolactone with aromatic aldehyde.. ChemCommun, 2011, 47(3): 1027-1029. |
| [29] | SAITO Y, KUBO W, KITAMURA T,et al.I-/I3- (-) redox reaction behavioronpoly (3, 4-Ethylenedioxythiophene) counter electrode in dye-sensitized solar cells. J. Photochem. Photobio. A, 2004, 164(3): 153-157. |
| [1] | ZHU Wenjie, TANG Lu, LU Jichang, LIU Jiangping, LUO Yongming. Research Progress on Catalytic Oxidation of Volatile Organic Compounds by Perovskite Oxides [J]. Journal of Inorganic Materials, 2025, 40(7): 735-746. |
| [2] | CHEN Libo, SHENG Ying, WU Ming, SONG Jiling, JIAN Jian, SONG Erhong. Na and O Co-doped Carbon Nitride for Efficient Photocatalytic Hydrogen Evolution [J]. Journal of Inorganic Materials, 2025, 40(5): 552-562. |
| [3] | GUO Ziyu, ZHU Yunzhou, WANG Li, CHEN Jian, LI Hong, HUANG Zhengren. Effect of Zn2+ Catalyst on Microporous Structure of Porous Carbon Prepared from Phenolic Resin/Ethylene Glycol [J]. Journal of Inorganic Materials, 2025, 40(5): 466-472. |
| [4] | GAO Chenguang, SUN Xiaoliang, CHEN Jun, LI Daxin, CHEN Qingqing, JIA Dechang, ZHOU Yu. SiBCN-rGO Ceramic Fibers Based on Wet Spinning Technology: Microstructure, Mechanical and Microwave-absorbing Properties [J]. Journal of Inorganic Materials, 2025, 40(3): 290-296. |
| [5] | WANG Yue, WANG Xin, YU Xianli. Room-temperature Ferromagnetic All-carbon Films Based on Reduced Graphene Oxide [J]. Journal of Inorganic Materials, 2025, 40(3): 305-313. |
| [6] | LI Na, CAO Ruixiao, WEI Jin, ZHOU Han, XIAO Hongmei. Performance and Influencing Factors of Iron-based Catalyst for Ortho to Para Hydrogen Conversion [J]. Journal of Inorganic Materials, 2025, 40(1): 47-52. |
| [7] | SHI Tong, GAN Qiaowei, LIU Dong, ZHANG Ying, FENG Hao, LI Qiang. Boost Electrochemical Reduction of CO2 to Formate Using a Self-supporting Bi@Cu Nanotree Electrode [J]. Journal of Inorganic Materials, 2024, 39(7): 810-818. |
| [8] | YANG Bo, LÜ Gongxuan, MA Jiantai. Electrocatalytic Water Splitting over Nickel Iron Hydroxide-cobalt Phosphide Composite Electrode [J]. Journal of Inorganic Materials, 2024, 39(4): 374-382. |
| [9] | YE Maosen, WANG Yao, XU Bing, WANG Kangkang, ZHANG Shengnan, FENG Jianqing. II/Z-type Bi2MoO6/Ag2O/Bi2O3 Heterojunction for Photocatalytic Degradation of Tetracycline under Visible Light Irradiation [J]. Journal of Inorganic Materials, 2024, 39(3): 321-329. |
| [10] | ZHANG Zhimin, GE Min, LIN Han, SHI Jianlin. Novel Magnetoelectric Catalytic Nanoparticles: RNS Release and Antibacterial Efficiency [J]. Journal of Inorganic Materials, 2024, 39(10): 1114-1124. |
| [11] | LI Qiushi, YIN Guangming, LÜ Weichao, WANG Huaiyao, LI Jinglin, YANG Hongguang, GUAN Fangfang. Preparation of Na+/g-C3N4 Materials and Their Photocatalytic Degradation Mechanism on Methylene Blue [J]. Journal of Inorganic Materials, 2024, 39(10): 1143-1150. |
| [12] | WANG Yanli, QIAN Xinyi, SHEN Chunyin, ZHAN Liang. Graphene Based Mesoporous Manganese-Cerium Oxides Catalysts: Preparation and Low-temperature Catalytic Reduction of NO [J]. Journal of Inorganic Materials, 2024, 39(1): 81-89. |
| [13] | DONG Yiman, TAN Zhan’ao. Research Progress of Recombination Layers in Two-terminal Tandem Solar Cells Based on Wide Bandgap Perovskite [J]. Journal of Inorganic Materials, 2023, 38(9): 1031-1043. |
| [14] | LI Yuejun, CAO Tieping, SUN Dawei. Bi4O5Br2/CeO2 Composite with S-scheme Heterojunction: Construction and CO2 Reduction Performance [J]. Journal of Inorganic Materials, 2023, 38(8): 963-970. |
| [15] | TUERHONG Munire, ZHAO Honggang, MA Yuhua, QI Xianhui, LI Yuchen, YAN Chenxiang, LI Jiawen, CHEN Ping. Construction and Photocatalytic Activity of Monoclinic Tungsten Oxide/Red Phosphorus Step-scheme Heterojunction [J]. Journal of Inorganic Materials, 2023, 38(6): 701-707. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||