
Journal of Inorganic Materials ›› 2017, Vol. 32 ›› Issue (5): 495-501.DOI: 10.15541/jim20160419
• Orginal Article • Previous Articles Next Articles
SONG Yun-Fei, WANG Shao-Hua, DENG Cheng-Ji, ZHU Hong-Xi, LIU Jian-Peng, DING Jun, YU Chao
Received:2016-07-18
Revised:2016-09-09
Published:2017-05-20
Online:2017-05-02
About author:SONG Yun-Fei. E-mail: 1054827854@qq.com
SONG Yun-Fei, WANG Shao-Hua, DENG Cheng-Ji, ZHU Hong-Xi, LIU Jian-Peng, DING Jun, YU Chao. Effects and Oxidation Kinetics of Holding Time on the β-Sialon Bonded MgAl2O4-C Composites[J]. Journal of Inorganic Materials, 2017, 32(5): 495-501.
| Materials | Size/mm | wt/% |
|---|---|---|
| Fused spinel | 1-3 | 40 |
| ≤1 | 30 | |
| ≤0.088 | 10 | |
| Flake graphite | ≤0.088 | 10 |
| Silicon powder | ≤0.074 | 10 |
| Calcium lignosulfonate | 1.25 g/mL | 4 |
Table 1 Experimental formula of MgAl2O4-C materials
| Materials | Size/mm | wt/% |
|---|---|---|
| Fused spinel | 1-3 | 40 |
| ≤1 | 30 | |
| ≤0.088 | 10 | |
| Flake graphite | ≤0.088 | 10 |
| Silicon powder | ≤0.074 | 10 |
| Calcium lignosulfonate | 1.25 g/mL | 4 |
| Crystal face symbols | d/nm | |||
|---|---|---|---|---|
| XRD results | Theoretical results | |||
| Z=2 | Z=3 | Z=4 | ||
| (2 0 0) | 0.332011 | 0.331674 | 0.33284 | 0.33401 |
| (1 0 1) | 0.270064 | 0.270081 | 0.272096 | 0.27411 |
| (2 1 0) | 0.251264 | 0.250722 | 0.251604 | 0.25249 |
Table 2 Relationship between Z and d values from the theoretical and experimental results
| Crystal face symbols | d/nm | |||
|---|---|---|---|---|
| XRD results | Theoretical results | |||
| Z=2 | Z=3 | Z=4 | ||
| (2 0 0) | 0.332011 | 0.331674 | 0.33284 | 0.33401 |
| (1 0 1) | 0.270064 | 0.270081 | 0.272096 | 0.27411 |
| (2 1 0) | 0.251264 | 0.250722 | 0.251604 | 0.25249 |
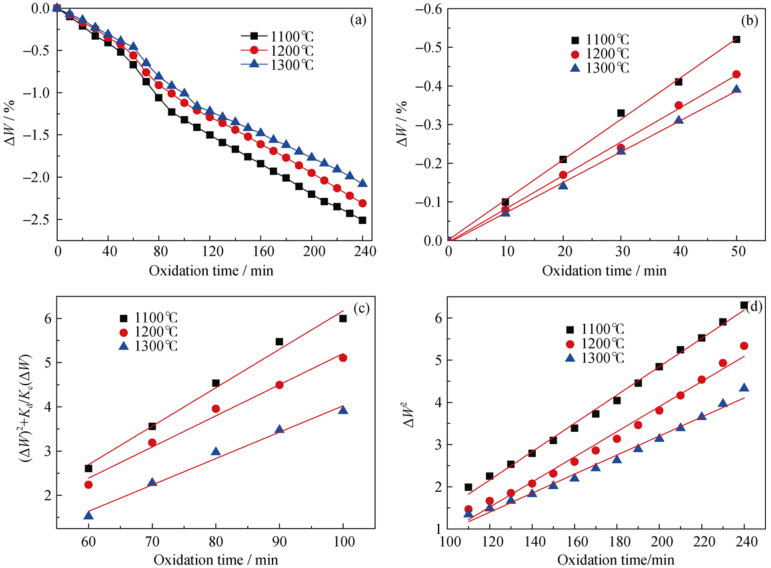
Fig. 6 (a) Oxidation kinetics of curves at constant temperatures, (b) oxidizing kinetic curves controlled by chemical reaction rate, (c) oxidizing kinetic curves controlled by interfacial chemical reaction and diffusion rate and (d) oxidizing kinetic curves controlled by diffusion rate of specimens SMAC
| [1] | LEIlA T, MOSTAFA M A, ZOHREH B. Instant synthesis of nano crystalline MgAl2O4 spinel powder.Journal of Inorganic Materials, 2011, 26(5): 550-554. |
| [2] | BRAULIO M A L, BITTENCOURT L R M, PANDOLFELLI V C. Selection of binders for in situ spinel refractory castables. Journal of the European Ceramic Society, 2009, 29(13): 2727-2735. |
| [3] | JING GUI-HUA, XIAO GUO-QING.Recent research development of magnesia-alumina spinel refractories.Refractories, 2004, 38(5): 347-349. |
| [4] | CHEN H, LIANG F, FANG W, et al.Fabrication of microporous corundum-spinel refractory castables by foam-gelcasting methods. Journal of Materials Engineering & Performance, 2015, 24(8): 1-7. |
| [5] | VANESA M, PILAR P, ANALIA G T M. Physical, chemical and thermal characterization of alumina-magnesia-carbon refractories.Ceramics International, 2014, 40(7): 9133-9149. |
| [6] | MUNOZ V, MARTINEZ A G T. Thermal evolution of Al2O3-MgO-C refractories. Procedia Materials Science, 2012, 1: 410-417. |
| [7] | KASHCHEEV I D, ZEMLYSNOI K G, VISLOGUZOVA É A, et al.Study of carbon-containing refractories for steel smelting production units.Refractories & Industrial Ceramics, 2007, 48(5): 313-316. |
| [8] | WEI C, ANATOLY R.A tough sialon ceramics based on α-Si3N4 With a whisker-like microstructure.Nature, 1997, 389(6652): 701-704. |
| [9] | PENG NAI, DENG CHENG-JI, ZHU HONG-XI, et al.Effect of keeping warm time on the properties of in-situ synthesis Si3N4 bonded MgO-C refractory. Science & Technology Review, 2013, 31(5): 37-39. |
| [10] | PENG N, DENG C J, ZHU H X, et al.Effects of alumina sources on the microstructure and properties of nitrided Al2O3-C refractories. Ceramics International, 2014, 41(4): 5513-5524. |
| [11] | DENG CHENG-JI, HONG YAN-RUO, SUN JIA-LIN, et al.Approach to the formation of MgAlON spinel in Al2O3-MgO-C refractory.Journal of the Chinese Ceramic Society, 2000, 28(5): 453-457. |
| [12] | ZHANG HAI-JUN, LIU ZHAN-JIE, ZHONG XIANG-CHONG, et al.Thermodynamic study and reduction-nitridation synthesis of O’-Sialon from coal gangue.Journal of Inorganic Materials, 2004, 19(5): 1129-1137. |
| [13] | ZHANG HAI-JUN, LIU ZHAN-JIE, ZHONG XIANG-CHONG.Properties of β-Sialon bonded corundum composites. Journal of the Chinese Ceramic Society, 2005, 33(11): 1308-1313. |
| [14] | ZHANG HAI-JUN, LI WEN-CHAO, YAO XI, et al.Friction and wear properties of O′-Sialon-ZrO-SiC composite.Journal of Inorganic Materials, 2000, 15(3): 480-486. |
| [15] | IZHEVSKIV V A, GENOVA L A, BRESSIANI J C, et al.Progress in SiAlON ceramics.Journal of the European Ceramic Society, 2000, 20(13): 2275-2295. |
| [16] | QIU XIONG-ER, PAN WEI, ZHOU YI-CHUN.Microwave synthesis of β-Sialon.Rare Metal Material and Engineering, 2007, 36(2): 354-358. |
| [17] | SOPICKA L M, TERPSTRA R A, METSELAAR R.Carbothermal production of β′-sialon from alumina, silica and carbon mixture.Journal of Materials Science, 1994, 30(24): 6363-6369. |
| [18] | HUANG JUN-TONG, FANG MING-HAO, WANG XIN-YING, et al.Study on synthesizing diphase sialon from lower Al2O3 bauxite by carbothermal reduction-nitridation.Rare Metal Material and Engineering, 2007, 36(s1): 373-375. |
| [19] | WANG CHUN-MEI, CHEN GUO-XIANG, CHEN XIAO-CHUN.Research and application of MgAl2O3-C refractories for 160t steel ladle.Refractories, 2005, 39(3): 230-231. |
| [20] | VAN R A, NEUROCK M, SHETTY S G.Reactivity theory of transition-metal surfaces: a Brønsted-Evans-Polanyi linear activation energy-free-energy analysis.Chemical Reviews, 2010, 110(4): 2005-2048. |
| [21] | HOFMANN S, CSANYI G, FERRARI A C, et al.Surface diffusion: the low activation energy path for nanotube growth.Physical Review Letters, 2005, 95(3): 1-4. |
| [1] | WANG Kunpeng, LIU Zhaolin, LIN Cunsheng, WANG Zhiyu. Development of Quasi-solid-state Na-ion Battery Based on Water-minimal Prussian Blue Cathode [J]. Journal of Inorganic Materials, 2024, 39(9): 1005-1012. |
| [2] | KONG Jianfeng, HUANG Jiecheng, LIU Zhaolin, LIN Cunsheng, WANG Zhiyu. Development of Quasi-solid-state Na-ion Battery Based on DPEPA-derived Gel Polymer Electrolyte [J]. Journal of Inorganic Materials, 2024, 39(12): 1331-1338. |
| [3] | LIU Suolan, LUAN Fuyuan, WU Zihua, SHOU Chunhui, XIE Huaqing, YANG Songwang. In-situ Growth of Conformal SnO2 Layers for Efficient Perovskite Solar Cells [J]. Journal of Inorganic Materials, 2024, 39(12): 1397-1403. |
| [4] | YAO Yishuai, GUO Ruihua, AN Shengli, ZHANG Jieyu, CHOU Kuochih, ZHANG Guofang, HUANG Yarong, PAN Gaofei. In-situ Loaded Pt-Co High Index Facets Catalysts: Preparation and Electrocatalytic Performance [J]. Journal of Inorganic Materials, 2023, 38(1): 71-78. |
| [5] | XUE Hongyun, WANG Congyu, MAHMOOD Asad, YU Jiajun, WANG Yan, XIE Xiaofeng, SUN Jing. Two-dimensional g-C3N4 Compositing with Ag-TiO2 as Deactivation Resistant Photocatalyst for Degradation of Gaseous Acetaldehyde [J]. Journal of Inorganic Materials, 2022, 37(8): 865-872. |
| [6] | WEN Zicong, NIU Dechao, LI Yongsheng. Silver Clusters-loaded Silica-based Hybrid Nanoparticles: Synthesis and SERS Performance [J]. Journal of Inorganic Materials, 2021, 36(12): 1297-1304. |
| [7] | YU Shouwu, ZHAO Zewen, ZHAO Jinjin, XIAO Shujuan, SHI Yan, GAO Cunfa, SU Xiao, HU Yuxiang, ZHAO Zhisheng, WANG Jie, WANG Lianzhou. Research Progress in Novel In-situ Integrative Photovoltaic-storage Tandem Cells [J]. Journal of Inorganic Materials, 2020, 35(6): 623-632. |
| [8] | XIA Tian, MENG Xie, LUO Ting, ZHAN Zhongliang. La 3+-substituted Sr2Fe1.5Ni0.1Mo0.4O6-δ as Anodes for Solid Oxide Fuel Cells [J]. Journal of Inorganic Materials, 2020, 35(5): 617-622. |
| [9] | YAN Yiyuan, JU Jiangwei, YU Meiyan, CHEN Shougang, CUI Guanglei. In-situ Polymerization Integrating 3D Ceramic Framework in All Solid-state Lithium Battery [J]. Journal of Inorganic Materials, 2020, 35(12): 1357-1364. |
| [10] | LIU Xiao-Yuan, LIU Bao-Dan, JIANG Ya-Nan, WANG Ke, ZHOU Yang, YANG Bing, ZHANG Xing-Lai, JIANG Xin. In-situ Synthesis of Perovskite SrTiO3 Nanostructures with Modified Morphology and Tunable Optical Absorption Property [J]. Journal of Inorganic Materials, 2019, 34(1): 65-71. |
| [11] | LIU Huan-Long, ZHAO Wei, LI Rui-Zhe, HUANG Xie-Yi, TANG Yu-Feng, LI Dong-Mei, HUANG Fu-Qiang. Facile Synthesis of Reduced Graphene Oxide In-situ Wrapped MnTiO3 Nanoparticles for Excellent Lithium Storage [J]. Journal of Inorganic Materials, 2018, 33(9): 1022-1028. |
| [12] | LIU Can-Jun, CHEN Shu, LI Jie. CdS/TiO2 Nanocrystalline Films: In-situ Synthesis and Photoelectrochemical Performance [J]. Journal of Inorganic Materials, 2018, 33(12): 1343-1348. |
| [13] | CHEN Cheng, LI Chen-Hui, KE Wen-Ming, SHI Yu-Sheng, HE Zhi-Yong, ZHANG Qi-Fu. Prediction of Sintering Process for ZrO2 Ceramics Based on Master Sintering Curve Theory and Its Expansion [J]. Journal of Inorganic Materials, 2016, 31(1): 53-57. |
| [14] | GUO Mi-Lan, TU Heng-Yong, LI Si-Lin, YU Qing-Chun. Fabrication and Characterization of Functionally Graded Cathodes Based on in-situ Formed La0.6Sr0.4CoO3-δ for Intermediate Temperature SOFCs [J]. Journal of Inorganic Materials, 2014, 29(6): 621-626. |
| [15] | YAN Zhi-Qiao,XIONG Xiang,XIAO Peng,HUANG Bo-Yun. Oxidation Kinetics and Mechanism of C/SiC Composites Fabricated by MSI Process [J]. Journal of Inorganic Materials, 2007, 22(6): 1151-1158. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||