无机材料学报 ›› 2026, Vol. 41 ›› Issue (4): 509-518.DOI: 10.15541/jim20250250 CSTR: 32189.14.10.15541/jim20250250
张梦婕1( ), 李智博1,2, 黄瑞楠1, 吕向菲1, 王伟1(
), 李智博1,2, 黄瑞楠1, 吕向菲1, 王伟1( )
)
收稿日期:2025-06-13
修回日期:2025-10-10
出版日期:2026-04-20
网络出版日期:2025-10-17
通讯作者:
王 伟, 教授. E-mail: wwchem@chd.edu.cn作者简介:张梦婕(2000-), 女, 硕士研究生. E-mail: zhangmengjie0120@163.com
基金资助:
ZHANG Mengjie1( ), LI Zhibo1,2, HUANG Ruinan1, LÜ Xiangfei1, WANG Wei1(
), LI Zhibo1,2, HUANG Ruinan1, LÜ Xiangfei1, WANG Wei1( )
)
Received:2025-06-13
Revised:2025-10-10
Published:2026-04-20
Online:2025-10-17
Contact:
WANG Wei, professor. E-mail: wwchem@chd.edu.cnAbout author:ZHANG Mengjie (2000-), female, Master candidate. E-mail: zhangmengjie0120@163.com
Supported by:摘要:
随着尾气排放法规的日益严格, 对柴油机尾气颗粒物过滤器(DPF)的过滤精度提出了更高的要求, 传统DPF难以满足尾气中纳米颗粒精密过滤的需求。本研究以堇青石蜂窝陶瓷为基体, 采用十六烷基二甲基溴化铵(CTAB)辅助共沉淀法在硼酸铝晶须上负载Co0.8FexCe0.2-xCr2O4系列尖晶石型催化剂。独特的“纤毛状晶须增强过滤-双金属掺杂催化氧化”分级结构设计实现了高效过滤性能和低温催化性能的结合, 可在较低的温度下催化氧化碳烟颗粒, Fe与Ce共掺杂产生了显著的协同效应, 提高了催化剂表面氧物种(氧空位)、Co3+、Cr6+的浓度, 增强了反应活性。负载C0.8F0.15C0.05C2催化剂(Z/C0.8F0.15C0.05C2-CT)使碳烟颗粒转化率达到50%对应的温度(T50=446 ℃)显著优于原始堇青石(T50=567 ℃)。5次循环测试表明该催化剂具有良好的稳定性和CO2选择性(86%~94%)。这种独特的分级结构有效实现了高效颗粒过滤与低温催化燃烧性能的统一, 在DPF领域具有潜在的应用前景。
中图分类号:
张梦婕, 李智博, 黄瑞楠, 吕向菲, 王伟. 堇青石/硼酸铝晶须/Co0.8FexCe0.2-xCr2O4催化剂的制备及其碳烟过滤-催化燃烧性能[J]. 无机材料学报, 2026, 41(4): 509-518.
ZHANG Mengjie, LI Zhibo, HUANG Ruinan, LÜ Xiangfei, WANG Wei. Cordierite/Aluminum Borate Whiskers/Co0.8FexCe0.2-xCr2O4 Catalysts: Preparation and Filtration Catalytic Performance for Carbon Soot[J]. Journal of Inorganic Materials, 2026, 41(4): 509-518.
| Sample | Calcination temperature/℃ | Holding time/h |
|---|---|---|
| A900-3 | 900 | 3 |
| A1000-3 | 1000 | 3 |
| A1050-3 | 1050 | 3 |
| A1100-3 | 1100 | 3 |
| A1200-3 | 1200 | 3 |
| A1050-1 | 1050 | 1 |
| A1050-2 | 1050 | 2 |
| A1050-4 | 1050 | 4 |
表1 不同加热温度和保温时间下制备的样品
Table 1 Samples preparation with different heating temperatures and holding time
| Sample | Calcination temperature/℃ | Holding time/h |
|---|---|---|
| A900-3 | 900 | 3 |
| A1000-3 | 1000 | 3 |
| A1050-3 | 1050 | 3 |
| A1100-3 | 1100 | 3 |
| A1200-3 | 1200 | 3 |
| A1050-1 | 1050 | 1 |
| A1050-2 | 1050 | 2 |
| A1050-4 | 1050 | 4 |

图1 样品的SEM照片
Fig. 1 SEM images of samples (a) Cordierite honeycomb ceramics; (b) A900-3; (c) A1000-3; (d) A1050-3; (e) A1100-3; (f) A1200-3; (g) A1050-1; (h) A1050-2; (i) A1050-4

图2 整体式催化剂的SEM照片
Fig. 2 SEM images of integral catalysts (a) Z/CoCr2O4-CT; (b) Z/C0.8F0.2C0C2-CT; (c) Z/C0.8F0.15C0.05C2-CT; (d) Z/C0.8F0.1C0.1C2-CT; (e) Z/C0.8F0.05C0.15C2-CT; (f) Z/C0.8F0C0.2C2-CT
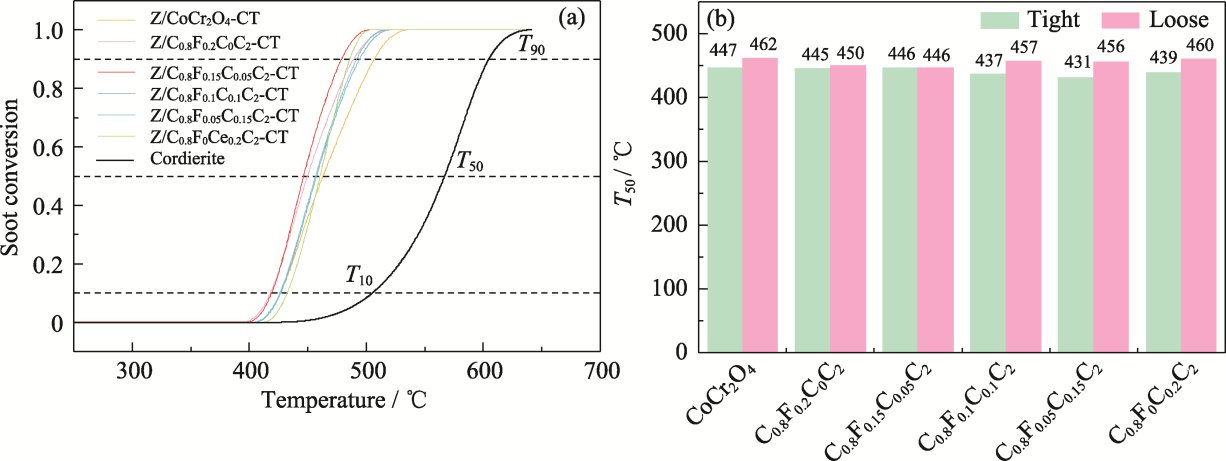
图8 (a) Z/C0.8FxC0.2-xC2-CT的碳烟催化燃烧活性; (b)尖晶石催化剂在紧密和松散接触条件下的催化性能
Fig. 8 (a) Soot catalytic combustion activity of Z/C0.8FxC0.2-xC2-CT; (b) Catalytic performance of spinel catalysts under tight and loose contact conditions Colorful figures are available on website
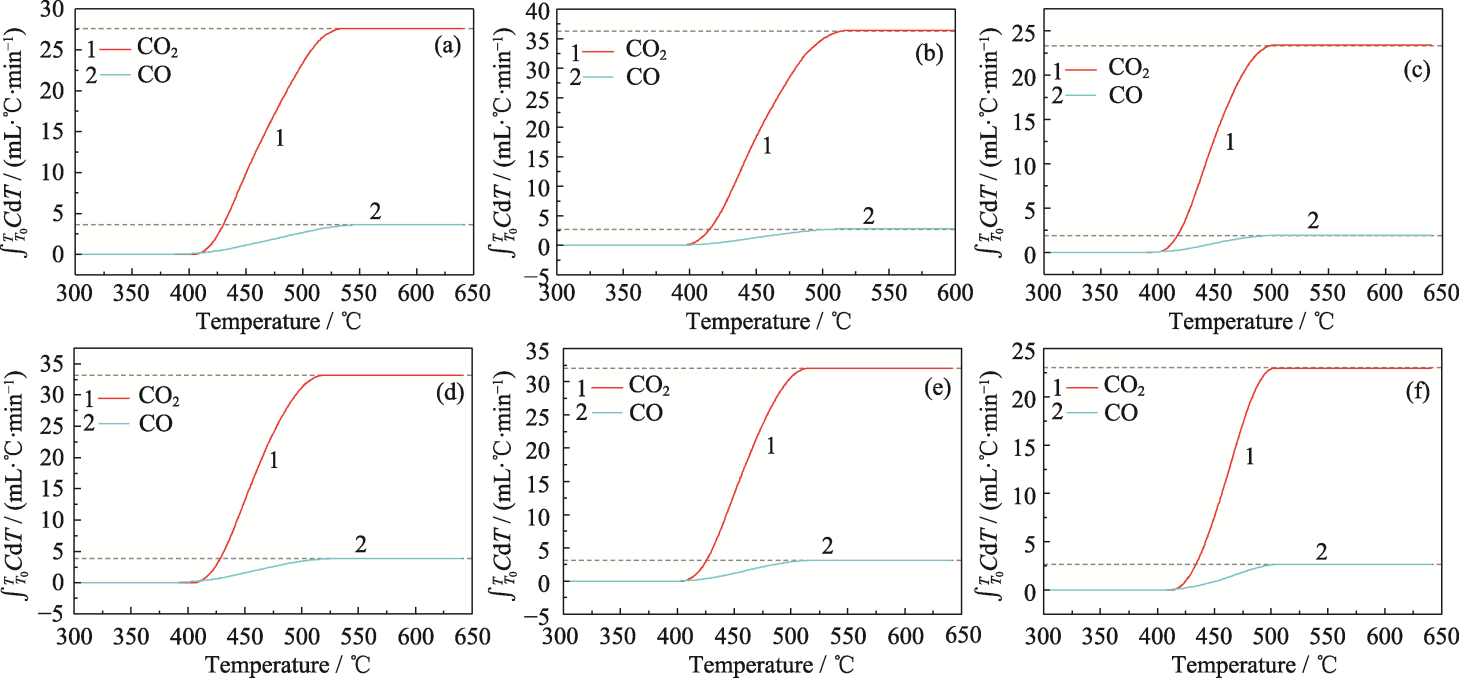
图10 Z/C0.8FxC0.2-xC2-CT催化碳烟燃烧生成的CO和CO2积分曲线
Fig. 10 Integral curves of CO and CO2 generated from combustion of soot catalyzed by Z/C0.8FxC0.2-xC2-CT (a) Z/CoCrO4-CT; (b) Z/C0.8F0.2C0C2-CT; (c) Z/C0.8F0.15C0.05C2-CT; (d) Z/C0.8F0.1C0.1C2-CT; (e) Z/C0.8F0.05C0.15C2-CT; (f) Z/C0.8F0C0.2C2-CT
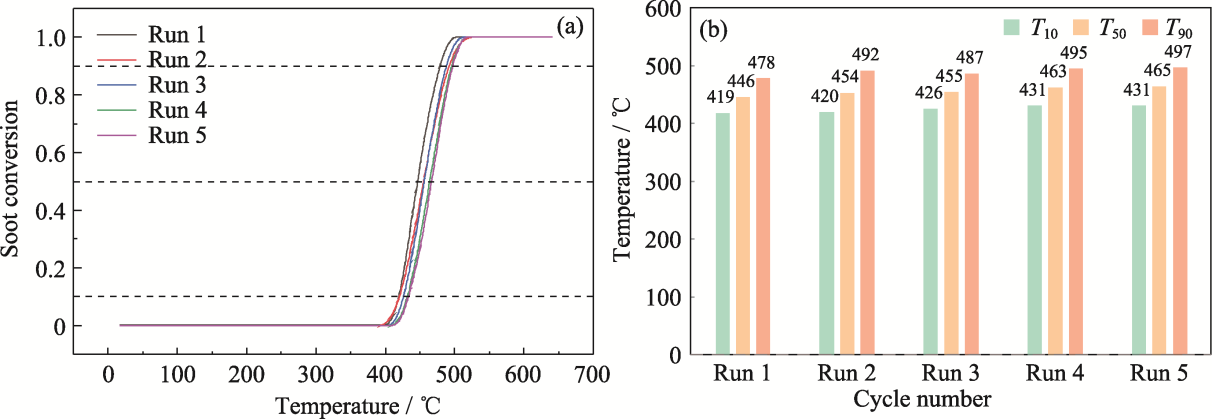
图11 Z/C0.8F0.15C0.05C2-CT的循环稳定性能
Fig. 11 Cyclic stability performance of Z/C0.8F0.15C0.05C2-CT (a) Soot conversion rate; (b) T10, T50 and T90. Colorful figures are available on website
| Sample | Lmean/μm | Dmean/μm | Lmean/Dmean |
|---|---|---|---|
| A900-3 | 8.69 | 0.66 | 13.17 |
| A1000-3 | 37.86 | 1.93 | 19.62 |
| A1050-3 | 48.42 | 0.71 | 68.20 |
| A1100-3 | 42.79 | 1.64 | 26.09 |
| A1050-1 | 15.70 | 0.62 | 25.32 |
| A1050-2 | 36.33 | 1.36 | 26.71 |
| A1050-4 | 53.49 | 2.07 | 25.84 |
表S1 硼酸铝晶须的各种参数
Table S1 Various parameters of aluminum borate whiskers
| Sample | Lmean/μm | Dmean/μm | Lmean/Dmean |
|---|---|---|---|
| A900-3 | 8.69 | 0.66 | 13.17 |
| A1000-3 | 37.86 | 1.93 | 19.62 |
| A1050-3 | 48.42 | 0.71 | 68.20 |
| A1100-3 | 42.79 | 1.64 | 26.09 |
| A1050-1 | 15.70 | 0.62 | 25.32 |
| A1050-2 | 36.33 | 1.36 | 26.71 |
| A1050-4 | 53.49 | 2.07 | 25.84 |
| Element | O | Na | Mg | Al | Si | Co | Ce | Fe | Cr |
|---|---|---|---|---|---|---|---|---|---|
| Atomic/% | 54.01 | 11.22 | 1.67 | 27.90 | 3.03 | 0.52 | 0.07 | 0.23 | 1.35 |
表S2 Z/C0.8F0.15C0.05C2-CT的表面元素含量
Table S2 Surface element contents of Z/C0.8F0.15C0.05C2-CT
| Element | O | Na | Mg | Al | Si | Co | Ce | Fe | Cr |
|---|---|---|---|---|---|---|---|---|---|
| Atomic/% | 54.01 | 11.22 | 1.67 | 27.90 | 3.03 | 0.52 | 0.07 | 0.23 | 1.35 |
| Sample | 2θ/(°) | FWHM/(°) | D/nm | d/nm | a |
|---|---|---|---|---|---|
| P-CoCr2O4-CT | 35.7881 | 0.8329 | 9.9079 | 0.2506 | 0.8312 |
| P-C0.8F0.2C0C2-CT | 35.7934 | 1.0052 | 8.2100 | 0.2506 | 0.8310 |
| P-C0.8F0.15C0.05C2-CT | 35.8655 | 1.2531 | 6.5869 | 0.2500 | 0.8294 |
| P-C0.8F0.1C0.1C2-CT | 35.8792 | 1.4346 | 5.7536 | 0.2500 | 0.8291 |
| P-C0.8F0.05C0.15C2-CT | 35.8928 | 1.4480 | 5.7009 | 0.2499 | 0.8288 |
| P-C0.8F0C0.2C2-CT | 35.9450 | 1.2704 | 6.4985 | 0.2496 | 0.8277 |
表S3 催化剂的晶格参数
Table S3 Crystal parameters of catalysts
| Sample | 2θ/(°) | FWHM/(°) | D/nm | d/nm | a |
|---|---|---|---|---|---|
| P-CoCr2O4-CT | 35.7881 | 0.8329 | 9.9079 | 0.2506 | 0.8312 |
| P-C0.8F0.2C0C2-CT | 35.7934 | 1.0052 | 8.2100 | 0.2506 | 0.8310 |
| P-C0.8F0.15C0.05C2-CT | 35.8655 | 1.2531 | 6.5869 | 0.2500 | 0.8294 |
| P-C0.8F0.1C0.1C2-CT | 35.8792 | 1.4346 | 5.7536 | 0.2500 | 0.8291 |
| P-C0.8F0.05C0.15C2-CT | 35.8928 | 1.4480 | 5.7009 | 0.2499 | 0.8288 |
| P-C0.8F0C0.2C2-CT | 35.9450 | 1.2704 | 6.4985 | 0.2496 | 0.8277 |
| Sample | Relative atomic ratio | ||||
|---|---|---|---|---|---|
| Osur/O | Co3+/Co | Fe3+/Fe | Ce4+/Ce | Cr6+/Cr | |
| Z/CoCr2O4-CT | 0.78 | 0.53 | / | / | 0.41 |
| Z/C0.8F0.2C0C2-CT | 0.81 | 0.55 | 0.36 | 0.75 | 0.50 |
| Z/C0.8F0.15C0.05C2-CT | 0.82 | 0.56 | 0.36 | 0.77 | 0.50 |
| Z/C0.8F0.1C0.1C2-CT | 0.80 | 0.54 | 0.33 | 0.73 | 0.49 |
| Z/C0.8F0.05C0.15C2-CT | 0.81 | 0.55 | 0.35 | 0.74 | 0.49 |
| Z/C0.8F0C0.2C2-CT | 0.79 | 0.53 | 0.34 | 0.72 | 0.44 |
表S4 表面物种的相对原子比
Table S4 Relative atomic ratios of surface species
| Sample | Relative atomic ratio | ||||
|---|---|---|---|---|---|
| Osur/O | Co3+/Co | Fe3+/Fe | Ce4+/Ce | Cr6+/Cr | |
| Z/CoCr2O4-CT | 0.78 | 0.53 | / | / | 0.41 |
| Z/C0.8F0.2C0C2-CT | 0.81 | 0.55 | 0.36 | 0.75 | 0.50 |
| Z/C0.8F0.15C0.05C2-CT | 0.82 | 0.56 | 0.36 | 0.77 | 0.50 |
| Z/C0.8F0.1C0.1C2-CT | 0.80 | 0.54 | 0.33 | 0.73 | 0.49 |
| Z/C0.8F0.05C0.15C2-CT | 0.81 | 0.55 | 0.35 | 0.74 | 0.49 |
| Z/C0.8F0C0.2C2-CT | 0.79 | 0.53 | 0.34 | 0.72 | 0.44 |
| Sample | Amount of carbon dioxide formed/(a.u.) |
|---|---|
| Z/CoCr2O4-CT | 85.312 |
| Z/C0.8F0.2C0C2-CT | 141.973 |
| Z/C0.8F0.15C0.05C2-CT | 145.109 |
| Z/C0.8F0.1C0.1C2-CT | 137.191 |
| Z/C0.8F0.05C0.15C2-CT | 138.278 |
| Z/C0.8F0C0.2C2-CT | 107.788 |
表S5 Soot-TPR曲线的定量结果
Table S5 Quantitative results of Soot-TPR profiles
| Sample | Amount of carbon dioxide formed/(a.u.) |
|---|---|
| Z/CoCr2O4-CT | 85.312 |
| Z/C0.8F0.2C0C2-CT | 141.973 |
| Z/C0.8F0.15C0.05C2-CT | 145.109 |
| Z/C0.8F0.1C0.1C2-CT | 137.191 |
| Z/C0.8F0.05C0.15C2-CT | 138.278 |
| Z/C0.8F0C0.2C2-CT | 107.788 |
| Sample | T10/℃ | T50/℃ | T90/℃ |
|---|---|---|---|
| Cordierite | 505 | 567 | 605 |
| Z/CoCr2O4-CT | 426 | 462 | 506 |
| Z/C0.8F0.2C0C2-CT | 418 | 450 | 489 |
| Z/C0.8F0.15C0.05C2-CT | 418 | 446 | 478 |
| Z/C0.8F0.1C0.1C2-CT | 426 | 457 | 494 |
| Z/C0.8F0.05C0.15C2-CT | 426 | 456 | 491 |
| Z/C0.8F0C0.2C2-CT | 432 | 460 | 486 |
表S6 在79% N2、21% O2气氛下的碳烟燃烧性能
Table S6 Soot combustion performance in 79% N2 and 21% O2 atmosphere
| Sample | T10/℃ | T50/℃ | T90/℃ |
|---|---|---|---|
| Cordierite | 505 | 567 | 605 |
| Z/CoCr2O4-CT | 426 | 462 | 506 |
| Z/C0.8F0.2C0C2-CT | 418 | 450 | 489 |
| Z/C0.8F0.15C0.05C2-CT | 418 | 446 | 478 |
| Z/C0.8F0.1C0.1C2-CT | 426 | 457 | 494 |
| Z/C0.8F0.05C0.15C2-CT | 426 | 456 | 491 |
| Z/C0.8F0C0.2C2-CT | 432 | 460 | 486 |
| Sample | |||
|---|---|---|---|
| Z/CoCr2O4-CT | 27.6 | 3.6 | 88.5% |
| Z/C0.8F0.2C0C2-CT | 36.4 | 2.8 | 92.9% |
| Z/C0.8F0.15C0.05C2-CT | 23.4 | 1.9 | 92.5% |
| Z/C0.8F0.1C0.1C2-CT | 33.2 | 3.9 | 89.5% |
| Z/C0.8F0.05C0.15C2-CT | 32.0 | 3.1 | 91.2% |
| Z/C0.8F0C0.2C2-CT | 23.0 | 2.6 | 89.8% |
表S7 整体式催化剂的CO2选择性
Table S7 CO2 selectivity of integral catalysts
| Sample | |||
|---|---|---|---|
| Z/CoCr2O4-CT | 27.6 | 3.6 | 88.5% |
| Z/C0.8F0.2C0C2-CT | 36.4 | 2.8 | 92.9% |
| Z/C0.8F0.15C0.05C2-CT | 23.4 | 1.9 | 92.5% |
| Z/C0.8F0.1C0.1C2-CT | 33.2 | 3.9 | 89.5% |
| Z/C0.8F0.05C0.15C2-CT | 32.0 | 3.1 | 91.2% |
| Z/C0.8F0C0.2C2-CT | 23.0 | 2.6 | 89.8% |
| Cycle | |||
|---|---|---|---|
| 1st | 23.4 | 1.9 | 92.5% |
| 2nd | 47.3 | 4.7 | 91.0% |
| 3rd | 31.9 | 2.2 | 93.5% |
| 4th | 32.0 | 5.0 | 86.5% |
| 5th | 29.4 | 4.3 | 87.2% |
表S8 循环样品的CO2选择性
Table S8 CO2 selectivity of circulated samples
| Cycle | |||
|---|---|---|---|
| 1st | 23.4 | 1.9 | 92.5% |
| 2nd | 47.3 | 4.7 | 91.0% |
| 3rd | 31.9 | 2.2 | 93.5% |
| 4th | 32.0 | 5.0 | 86.5% |
| 5th | 29.4 | 4.3 | 87.2% |
| Sample | M0/g | (M0-∆M)/g | η/% |
|---|---|---|---|
| Cordierite | 0.0157 | 0.0079 | 50.32 |
| Cordierite/aluminum borate | 0.0139 | 88.54 | |
| Z/C0.8F0.15C0.05C2-CT | 0.0141 | 89.81 |
表S9 整体式催化剂的过滤性能
Table S9 Filtration performance of monolithic catalysts
| Sample | M0/g | (M0-∆M)/g | η/% |
|---|---|---|---|
| Cordierite | 0.0157 | 0.0079 | 50.32 |
| Cordierite/aluminum borate | 0.0139 | 88.54 | |
| Z/C0.8F0.15C0.05C2-CT | 0.0141 | 89.81 |
| Catalyzer | Maximum combustion rate temperature/℃ | Ref. |
|---|---|---|
| Pt/Al2O3 | 464 | [ |
| CuxZn1-xAl2O4 | 630 | [ |
| CoAl2O4 | 510 | [ |
| CoCr2O4 | 420 | [ |
| CuCo2O4 | 574 | [ |
| Z/C0.8F0.15C0.05C2-CT | 446 | This work |
表S10 过滤催化性能对比
Table S10 Comparison of filtration catalytic performance of different catalyzers
| Catalyzer | Maximum combustion rate temperature/℃ | Ref. |
|---|---|---|
| Pt/Al2O3 | 464 | [ |
| CuxZn1-xAl2O4 | 630 | [ |
| CoAl2O4 | 510 | [ |
| CoCr2O4 | 420 | [ |
| CuCo2O4 | 574 | [ |
| Z/C0.8F0.15C0.05C2-CT | 446 | This work |
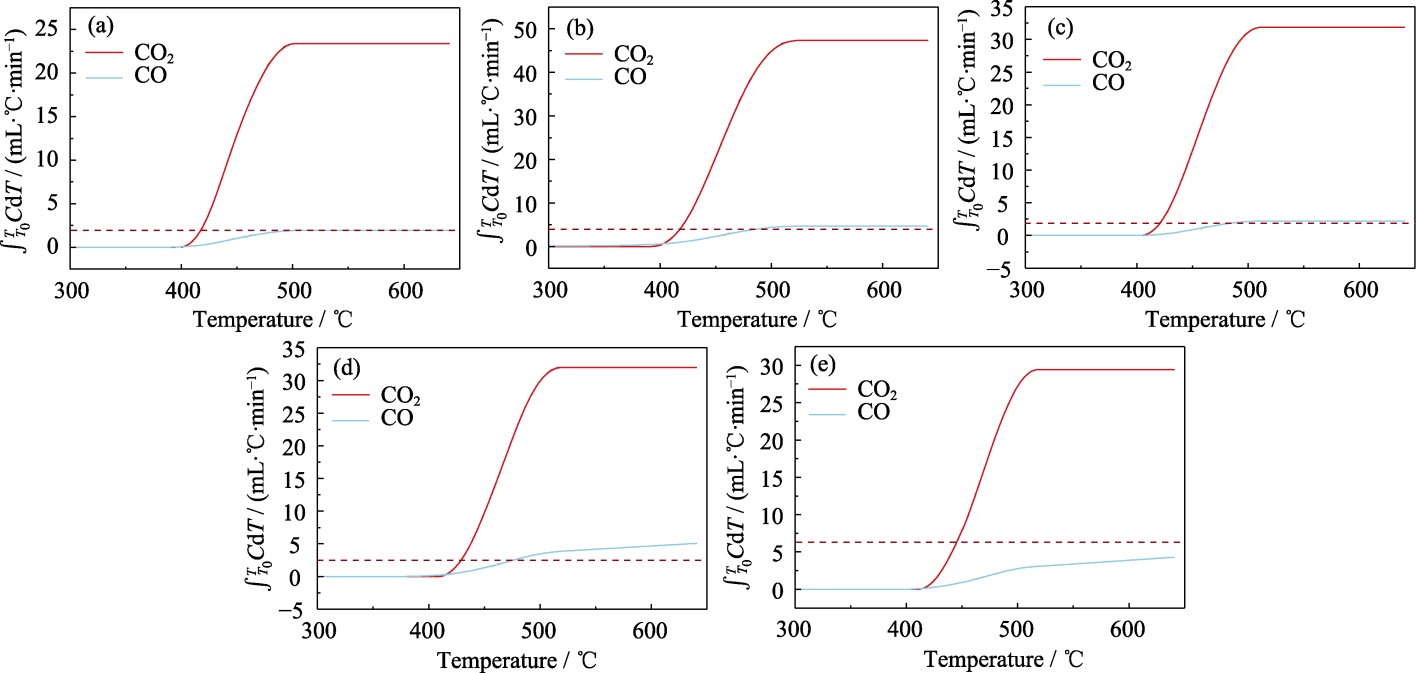
图S1 Z/C0.8F0.15C0.05C2-CT循环催化碳烟燃烧生成的CO和CO2积分曲线
Fig. S1 Integral curves of CO and CO2 generated from cycle catalytic soot combustion by Z/C0.8FxC0.2-xC2-CT (a) Cycle 1; (b) Cycle 2; (c) Cycle 3; (d) Cycle 4; (e) Cycle 5
| [1] | KO A, KIM J, CHOI K, et al. Experimental study of particle emission characteristics of a heavy-duty diesel engine and effects of after-treatment systems: selective catalytic reduction, diesel particulate filter, and diesel particulate and NOx reduction. Proceedings of the Institution of Mechanical Engineers, Part D: Journal of Automobile Engineering, 2012, 226(12): 1689. |
| [2] |
ZHANG S H, FU M L, ZHANG H F, et al. Emission control status and future perspectives of diesel trucks in China. Journal of Environmental Sciences, 2025, 148: 702.
DOI PMID |
| [3] |
CHUNG I C, AN Y K, PARK J, et al. Analysis of engine efficiency of diesel vehicle in transient operating conditions. International Journal of Automotive Technology, 2021, 22(4): 941.
DOI |
| [4] |
LI X D, XUE C F, GENG J W, et al. Study on regenerative exothermic process in ash containing DPF structure based on lattice Boltzmann method. Case Studies in Thermal Engineering, 2024, 55: 104105.
DOI URL |
| [5] |
THIERINGER J R D, HAFEN N, MEYER J, et al. Investigation of the rearrangement of reactive-inert particulate structures in a single channel of a wall-flow filter. Separations, 2022, 9(8): 195.
DOI URL |
| [6] |
YAMAMOTO K, NAKAMURA M, YANE H, et al. Simulation on catalytic reaction in diesel particulate filter. Catalysis Today, 2010, 153(3/4): 118.
DOI URL |
| [7] |
NIE W M, MEI X Y, XIE W P, et al. Electricity-driven rapid regeneration of ceramic paper-based soot filters with conductive potassium-supported antimony-doped tin oxide catalyst. Chemical Engineering Journal, 2024, 494: 152967.
DOI URL |
| [8] |
ZHANG C L, GAO S Y, ZHOU S R, et al. New method for the preparation of hierarchical nanotube K0.3xMnxCe1-xOδ catalysts and their excellent catalytic performance for soot combustion. Applied Catalysis B: Environment and Energy, 2024, 355: 124169.
DOI URL |
| [9] |
JAMPAIAH D, VELISOJU V K, DEVAIAH D, et al. Flower-like Mn3O4/CeO2 microspheres as an efficient catalyst for diesel soot and CO oxidation: synergistic effects for enhanced catalytic performance. Applied Surface Science, 2019, 473: 209.
DOI URL |
| [10] |
XU K, ZHANG Y, SHAN W P, et al. Promotional effects of Sm/Ce/La doping on soot oxidation over MnCo2O4 spinel catalysts. The Journal of Physical Chemistry C, 2021, 125(48): 26484.
DOI URL |
| [11] |
ZHAO H, ZHOU X X, PAN L Y, et al. Facile synthesis of spinel Cu1.5Mn1.5O4 microspheres with high activity for the catalytic combustion of diesel soot. RSC Advances, 2017, 7(33): 20451.
DOI URL |
| [12] |
杨华, 杨幸川, 徐丽, 等. 不同A位取代Co基尖晶石纳米线阵列的合成及其催化碳烟消除的研究. 现代化工, 2023, 43(3): 98.
DOI |
| [13] | 杨华. 泡沫镍基底上钴基尖晶石型整体式碳烟催化剂的研究. 郑州: 郑州大学硕士学位论文, 2022. |
| [14] | 宗鑫, 李新令. NO2-O2氛围下碳黑颗粒氧化特性研究. 小型内燃机与车辆技术, 2018, 47(5): 1. |
| [15] |
ATRIBAK I, BUENO-LÓPEZ A, GARCÍA-GARCÍA A. Uncatalysed and catalysed soot combustion under NOx + O2: real diesel versus model soots. Combustion and Flame, 2010, 157(11): 2086.
DOI URL |
| [16] | 舒荣禄. 氧化铈催化剂的掺杂改性及其对碳烟颗粒催化氧化的研究. 西安: 长安大学硕士学位论文, 2023. |
| [17] |
ANANDHA KUMAR S, SHAHANAS T, HARICHANDRAN G. Morphologically controlled preparation of CoCr2O4 nanomaterials for high-performance asymmetric supercapacitor application. Journal of Energy Storage, 2024, 77: 110011.
DOI URL |
| [18] |
BAI Y T, WANG S, ZONG X P, et al. Insights into the effect of Cu and Fe dispersion state over Ce0.1Al on the catalytic performance of NO reduction by CO. Journal of Environmental Chemical Engineering, 2023, 11(5): 110460.
DOI URL |
| [19] |
XIAN G, ZHANG N, ZHANG G M, et al. FeNiCeOx ternary catalyst prepared by ultrasonic impregnation method for diclofenac removal in Fenton-like system. Water Science and Technology, 2019, 79(9): 1675.
DOI URL |
| [20] |
OUYANG R M, XU J W, ZHOU J Q, et al. Defect BaSnO3 perovskites for oxidative dehydrogenation of ethane. Fuel, 2025, 384: 134022.
DOI URL |
| [21] |
ZHANG S, LIU S J, ZHU X C, et al. Low temperature catalytic oxidation of propane over cobalt-cerium spinel oxides catalysts. Applied Surface Science, 2019, 479: 1132.
DOI URL |
| [22] |
HE J S, YAO P, QIU J, et al. Enhancement effect of oxygen mobility over Ce0.5Zr0.5O2 catalysts doped by multivalent metal oxides for soot combustion. Fuel, 2021, 286: 119359.
DOI URL |
| [23] |
HOSSEINI S A, ALVAREZ-GALVAN M C, FIERRO J L G, et al. MCr2O4 (M=Co, Cu, and Zn) nanospinels for 2-propanol combustion: correlation of structural properties with catalytic performance and stability. Ceramics International, 2013, 39(8): 9253.
DOI URL |
| [24] | ZHANG M J, WANG Y, YU M, et al. Preparation of aluminum borate whiskers/CoxCr3-xO4 catalysts on channel surface of cordierite honeycomb ceramic for soot catalytic combustion. International Journal of Applied Ceramic Technology, 2025, 22(3): e14994. |
| [25] |
XU L F, LI Y N, ZHANG P, et al. Preparation and characterization of bimetallic Ni-Ir/C catalysts for HI decomposition in the thermochemical water-splitting iodine-sulfur process for hydrogen production. International Journal of Hydrogen Energy, 2019, 44(45): 24360.
DOI URL |
| [26] |
BHOSALE S R, BHOSALE R R, PATIL V L, et al. Superior antibacterial performance of surfactant-assisted ZnO nanoflakes produced via co-precipitation method. Chemical Physics Letters, 2024, 846: 141318.
DOI URL |
| [27] |
GAO Y B, TENG S C, WANG Z P, et al. Enhanced catalytic performance of cobalt and iron Co-doped ceria catalysts for soot combustion. Journal of Materials Science, 2020, 55(1): 283.
DOI |
| [28] |
YUE J L, XIONG F, SHADIKE Z, et al. A layer-structured high entropy oxide with highly reversible Fe3+/Fe4+ redox as advanced cathode material for sodium ion batteries. Journal of Power Sources, 2025, 627: 235735.
DOI URL |
| [29] |
MALYUKIN Y, KLOCHKOV V, MAKSIMCHUK P, et al. Oscillations of cerium oxidation state driven by oxygen diffusion in colloidal nanoceria (CeO2-x). Nanoscale Research Letters, 2017, 12(1): 566.
DOI URL |
| [30] |
LUO X, HONG Y, ZHANG H L, et al. Highly efficient steam reforming of ethanol (SRE) over CeOx grown on the nano NixMgyO matrix: H2 production under a high GHSV condition. International Journal of Energy Research, 2019, 43(8): 3823.
DOI URL |
| [31] |
TAN T, CHEN Y S, WANG Y Y, et al. Insight into the effects of the CO2/H2O activation and Ce redox cycle over Ni/CeO2/ hydrotalcite catalyst surface on biogas Bi-reforming for methanol friendly syngas. Energy, 2024, 313: 133954.
DOI URL |
| [32] |
LIU J, JIA Y Z, HAN C, et al. Effect of oxygen vacancy on the performance of Ce-promoted CuZn catalysts for ethanol synthesis from syngas. International Journal of Hydrogen Energy, 2024, 92: 1059.
DOI URL |
| [33] |
ZHANG J R, YANG T L, RAO Q, et al. Enhancement of Fe/Ce oxygen carrier performance in chemical looping dry reforming of methane. Fuel, 2024, 366: 131344.
DOI URL |
| [34] |
XIAO J Y, ZHANG C, WANG Y L, et al. Homogeneously Co/Ce Co-doped amorphous MnOx microspheres with abundant oxygen vacancy for catalytic combustion of propane. Fuel, 2025, 382: 133631.
DOI URL |
| [1] | 刘江平, 管鑫, 唐振杰, 朱文杰, 罗永明. 含氮挥发性有机化合物催化氧化的研究进展[J]. 无机材料学报, 2025, 40(9): 933-943. |
| [2] | 朱文杰, 唐璐, 陆继长, 刘江平, 罗永明. 钙钛矿型氧化物催化氧化挥发性有机化合物的研究进展[J]. 无机材料学报, 2025, 40(7): 735-746. |
| [3] | 张瑞阳, 王壹, 欧博文, 周莹. α-Ni(OH)2表面羟基协同Ni3+位点催化氧化甲醛机理研究[J]. 无机材料学报, 2023, 38(10): 1216-1222. |
| [4] | 朱俊逸, 张成, 罗忠强, 曹继伟, 刘志远, 王沛, 刘长勇, 陈张伟. 脱脂工艺对光固化3D打印堇青石陶瓷性能的影响[J]. 无机材料学报, 2022, 37(3): 317-324. |
| [5] | 孙扬善, 杨治华, 蔡德龙, 张正义, 柳琪, 房树清, 冯良, 石丽芬, 王友乐, 贾德昌. 粉末烧结法制备α-堇青石基玻璃陶瓷的析晶动力学和性能[J]. 无机材料学报, 2022, 37(12): 1351-1357. |
| [6] | 黄谢意,王鹏,尹国恒,张绍宁,赵伟,王东,毕庆员,黄富强. 掺磷非晶氧化钛负载铂用于高效催化氧化挥发性有机化合物[J]. 无机材料学报, 2020, 35(4): 482-490. |
| [7] | 李思汉, 张超, 吴辰亮, 张荷丰, 严新焕. 低负载量Pd/CeO2/γ-Al2O3催化剂用于低温催化氧化VOCs[J]. 无机材料学报, 2019, 34(8): 827-833. |
| [8] | 郭瑞华, 莫逸杰, 安胜利, 张捷宇, 周国治. 氧化铈空心球可控合成及其对Pt基催化剂电催化性能的影响[J]. 无机材料学报, 2018, 33(7): 779-786. |
| [9] | 严世胜, 彭鸿雁, 赵志斌, 潘孟美, 羊大立, 阿金华, 叶国林, 王崇太, 郭心玮. 掺氮金刚石电极性能及其氧化降解硝基苯研究[J]. 无机材料学报, 2018, 33(5): 565-569. |
| [10] | 翟春阳, 孙明娟, 杜玉扣, 朱明山. 贵金属/半导体光电阳极在直接甲醇燃料电池中的应[J]. 无机材料学报, 2017, 32(9): 897-903. |
| [11] | 李 敏, 洛 园, 许伟佳, 刘家祥. DMFC阳极催化剂Fe3O4@Pt的制备及其催化性[J]. 无机材料学报, 2017, 32(9): 916-922. |
| [12] | 张 艺, 张晓凤, 何晓蕾, 黄火娣, 乐丽娟, 林 深. Pt/{GN/CuPW11}n复合膜的制备及其对甲醇氧化电催化性能[J]. 无机材料学报, 2017, 32(10): 1075-1082. |
| [13] | 金朝, 陈其汉, 郑梦佳, 赵鹏, 李倩, 崔小强. Au纳米粒子/双层TiO2周期结构的制备及其对乙醇的电催化性质[J]. 无机材料学报, 2016, 31(3): 241-247. |
| [14] | 郑遗凡, 仲淑彬, 吕德义, 周 环, 郇昌永. 焙烧温度对Cu-Mn/γ-Al2O3催化剂催化氧化甲苯性能的影响[J]. 无机材料学报, 2015, 30(7): 694-698. |
| [15] | 华成江, 王明辉, 栾国有, 刘 岩, 吴 华. 原位晶化法在铜网上快速合成Cu3(BTC)2基膜及其催化性能研究[J]. 无机材料学报, 2015, 30(5): 529-534. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||