为了提高钙钛矿材料的稳定性, 科学家进行了许多探索。其中, 混合阳离子工程[8⇓-10]是一种提高效率和稳定性的有效方法。典型钙钛矿的化学组成为ABX3型[11], A阳离子一般为+1价的CH3NH3+、CH(NH2)2+或Cs+; B离子一般为+2价金属阳离子, 如Pb2+、Sn2+、Ge2+等; X离子一般为−1价Cl−、Br−、I−等卤素离子。在铅基钙钛矿骨架中, Pb2+位于[BX6]4−八面体晶格的中心, A阳离子占据八面体间隙[12], X离子占据八面体顶角。容差因子
本研究采用简单温和且成本低廉的两步旋涂法在全空气环境中制备了钙钛矿薄膜并组装成器件。同时系统研究了掺杂FA+和Cs+对MAPbI3钙钛矿吸收层微观形貌、物相组成、PSCs光伏性能和长期稳定性的影响并进行机理分析。
1 实验方法
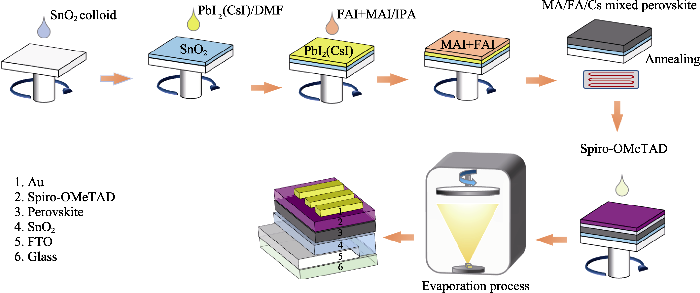
实验过程均在空气环境(相对湿度30%~40%)中完成, 实验流程如图1所示。
图1
图1
在空气中两步旋涂法制备钙钛矿薄膜及器件制造的流程图
Fig. 1
Flow chart for the preparation of perovskite films and device manufacturing by two-step spin coating in the air
1.1 SnO2电子传输层的制备
将FTO玻璃依次用洗涤剂、去离子水、乙醇等分别超声清洗10 min, 随后放入紫外臭氧清洗机处理40 min。取60 μL SnO2(质量分数10%)胶体水溶液旋涂(5000 r/min, 30 s)在FTO基底上, 150 ℃退火30 min, 得到致密SnO2电子传输层。
1.2 钙钛矿薄膜的制备
MAPbI3钙钛矿薄膜(MP): 将PbI2溶于DMF和DMSO的混合溶剂(VDMF∶VDMSO=9∶1)中, 105 ℃下搅拌1 h, 制成1.3 mol/L的PbI2前驱体溶液。称取适量MAI和MACl药品溶解在异丙醇(IPA)中得到混合(0.4 mol/L MAI, 0.07 mol/L MACl)溶液。取30 μL PbI2溶液旋涂(3000 r/min, 30 s)在涂有致密SnO2电子传输层的基底上, 随后取110 μL MAI溶液旋涂 (5000 r/min, 10 s)在PbI2薄膜上, 空气环境中(相对湿度30%~40%)在105 ℃退火10 min, 得到结晶良好的MAPbI3薄膜, 标记为MP。
MA0.4FA0.6PbI3钙钛矿薄膜(MFP): 用适量FAI取代MAI溶解于异丙醇中, 制备MAFA混合有机盐溶液, 与PbI2反应生成MA0.4FA0.6PbI3, 最后在150 ℃退火30 min, 其它制备条件与MAPbI3相同, 得到的薄膜标记为MFP。
Cs0.05MA0.35FA0.6PbI3钙钛矿薄膜(CMFP): 添加适量1.0 mol/L CsI溶液(DMSO为溶剂)到PbI2溶液中, 其他制备条件与MA0.4FA0.6PbI3钙钛矿相同, 得到的薄膜标记为CMFP。
1.3 空穴传输层和背电极的沉积
在钙钛矿薄膜表面旋涂30 μL的Spiro- OMeTAD溶液(4000 r/min, 20 s), 得到空穴传输层, 然后将基片置于干燥器中遮光老化12 h。再利用热蒸发仪在Spiro-OMeTAD层上蒸镀一层80 nm厚的金电极。
2 结果与讨论
2.1 A位阳离子掺杂对钙钛矿薄膜结构和形貌的影响
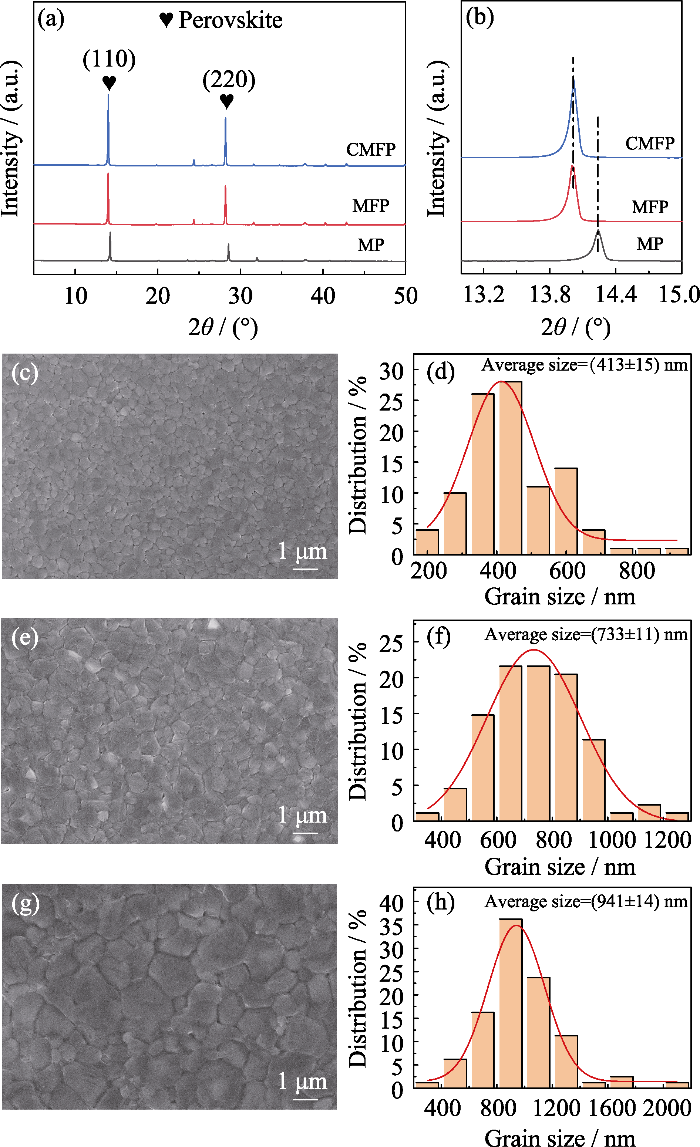
钙钛矿光吸收层是电池的核心, 钙钛矿薄膜的质量对于电池的性能至关重要。为表征薄膜质量, 分别对三种薄膜进行X射线衍射(XRD)分析和扫描电子显微镜(SEM)表征。如图2(a)所示, 位于2θ= 14.24°, 28.25°的衍射峰归属于MAPbI3钙钛矿晶体结构的(110)和(220)晶面[23]; 加入FA+和Cs+后, 衍射峰逐渐增强。从图2(b)可以看出, FA+取代MA+后, 位于2θ=14.24°的衍射峰向低角度偏移(2θ= 14.02°)。根据布拉格方程2dsinθ=nλ(其中d为晶面间距, θ为入射X射线与相应晶面的夹角, λ为X射线的波长, n为衍射级数), 加入Cs+基本没有改变衍射峰角度(相比MFP), 可能是因为Cs+的掺杂量较少。
图2
图2
不同样品的晶体结构和微观形貌
Fig. 2
Crystal structures and morphologies of different samples
(a) XRD patterns; (b) Locally magnified XRD patterns in the range of 2θ=12.8°-15°; (c, e, g) SEM images and (d, f, h) Statistical distributions of grain diameter for (c, d) MP, (e, f) MFP, and (g, h) CMFP
不同样品的SEM照片如图2(c, e, g)所示, 三种钙钛矿薄膜都均匀且致密, 掺杂FA+和Cs+使薄膜的晶粒尺寸逐步增大。图2(d, f, h)中, 掺杂FA+薄膜的平均晶粒尺寸从413 nm增大到733 nm, 进一步掺杂Cs+, 薄膜的平均晶粒尺寸增大到941 nm, 说明加入FA+和Cs+可以促进钙钛矿薄膜结晶, 增大晶粒, 减少晶界。
2.2 A位阳离子掺杂对器件PCE的影响及机理分析
2.2.1 A位阳离子掺杂对器件PCE的影响
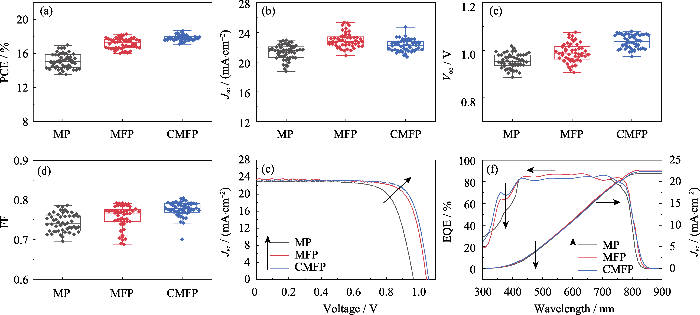
图3
图3
PSCs的光电性能
Fig. 3
Photovoltaic performances of PSCs
Statistical diagram of (a) PCE, (b) Jsc, (c) Voc, and (d) FF; (e) J-V curves; (f) EQE spectra. Colorful figures are available on website
表1 三种钙钛矿太阳能电池的光电性能参数
Table 1
| Sample | Jsc /(mA•cm-2) | Voc/V | FF | PCE/% | PCEmax/% |
|---|---|---|---|---|---|
| MP | 21.32±0.99 | 0.95±0.03 | 0.74±0.02 | 15.11±0.86 | 16.31 |
| MFP | 22.94±0.98 | 0.99±0.04 | 0.76±0.03 | 17.18±0.61 | 18.87 |
| CMFP | 22.24±0.77 | 1.03±0.03 | 0.78±0.02 | 17.83±0.33 | 19.34 |
图3(a~d)和表1显示, 掺杂FA+使MFP平均Jsc由21.32 mA•cm-2(MP)提高到22.94 mA•cm-2, 而平均Voc和FF分别从0.95 V和0.74增大到0.99 V和0.76, PSCs的平均PCE从15.11%提高到17.18%。掺杂Cs+使CMFP的平均PCE进一步提高到17.82%, 这主要归因于电池的Voc和FF得到了提高, CMFP的最大PCE为19.34%。图3(f)是三种电池的外量子效率(EQE)测试曲线, 可以用于校准短路电流密度。通过EQE图谱(图3(f))得到的MP、MFP和CMFP电池的积分电流密度分别为21.87、22.71和22.37 mA·cm-2, 与J-V测试(图3(e))得到的Jsc(22.87、23.48、23.30 mA·cm-2)相差不大, 说明测试结果较准确。
2.2.2 A位阳离子掺杂提升器件PCE的机理
一般来说, PSCs的PCE与Jsc、Voc和FF密切相关。因此, 探索A位阳离子掺杂对这三个参数的影响对于揭示器件光电性能的机理至关重要。Jsc由以下公式[23]决定。
其中,
因为三种PSCs使用相同的透明电极, 所以
为了探索阳离子掺杂对薄膜
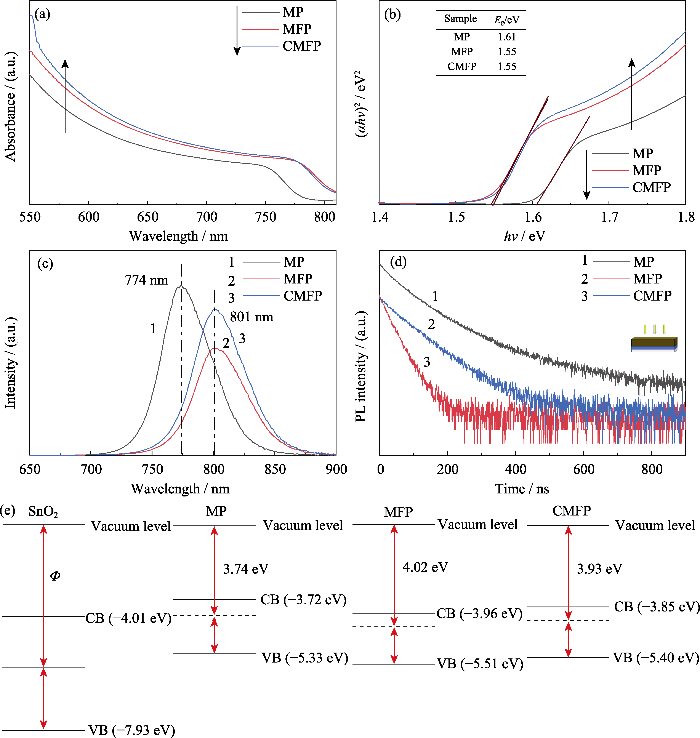
图4
图4
三种钙钛矿薄膜的光电特性和能级结构
Fig. 4
Photoelectric properties and energy levels of three perovskite films
(a) UV-Vis absorption spectra; (b) Tauc plots; (c) PL and (d) TRPL spectra excited from the perovskite layer;(e) Energy level schematics of three samples
式中, τ1表示钙钛矿表面和界面处的光生载流子寿命(快过程), ns; τ2表示钙钛矿晶粒内部的光生载流子寿命(慢过程), ns; A1和A2分别表示这两个过程的相对振幅, y0表示常数。
平均载流子寿命τmean可以用来衡量薄膜整体的缺陷密度,
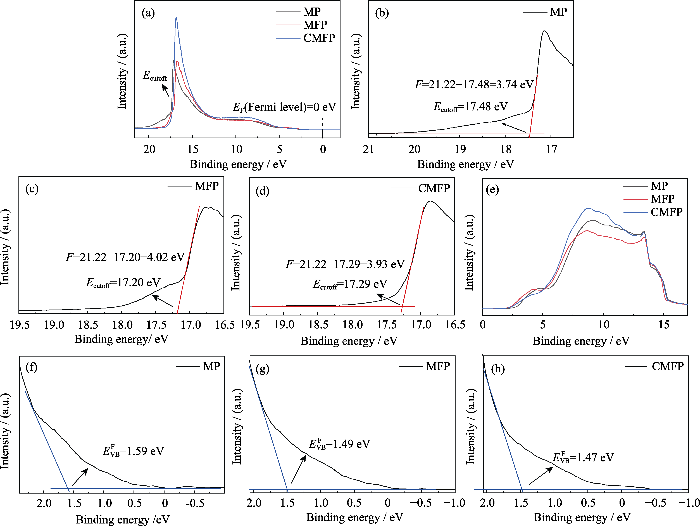
如图4(e)所示, MFP具有最高的功函数(Φ= 4.02 eV)以及最低的ECB(–3.96 eV), 因此MFP钙钛矿与SnO2之间的能级势垒(ΔE)最小, 表明引入FA+可以改善电荷收集, 增大
开路电压Voc可以被定义为[26]:
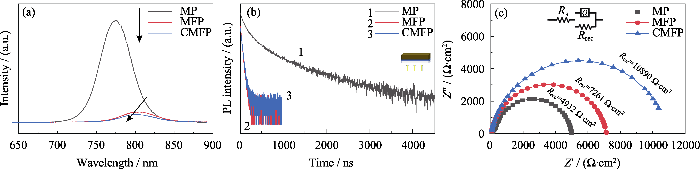
其中, EFermi表示电子传输材料的准费米能级, EN为空穴传输层的能斯特电势。电子传输层中收集的电子越多, EFermi越高, 则Voc越大。因此Voc的大小与界面间的能级匹配、电子的界面传输性质以及器件的复合阻抗(Rrec)密切相关。而填充因子FF本质上代表了抽取光伏器件中光生载流子的难易程度, 串联电阻(Rs)减小和并联电阻(Rrec)增大都有利于提高器件的填充因子。
图4(e)是三种钙钛矿薄膜能带排列情况, CMFP的ECB和Φ相比MFP均向上移动, 这种能带结构有利于减少电荷复合, 因此Cs+掺杂可以增大电池的开路电压Voc。
透过FTO激发进行PL和TRPL测试(图5(a, b)), 拟合结果(表S4)能够反映电子从钙钛矿层传输到SnO2的速率, 即电子的抽取速率。CMFP、MFP和MP的
图5
图5
PSCs的界面传输和载流子复合特性
Fig. 5
Interface transmission and carrier recombination characteristics of PSCs
(a) PL and (b) TRPL spectra excited from FTO layer; (c) Dark-state EIS profiles of the device at 0.8 Vbias with inset showing an equivalent circuit diagram
综上所述, 掺杂FA+可以全面提高器件的Voc、Jsc和FF, 从而将电池的平均PCE从15.11%提高到17.18%; 掺杂Cs+有利于进一步优化器件的Voc和FF, 最终将电池的平均PCE提高到17.83%。
2.3 A位阳离子掺杂对器件稳定性的影响
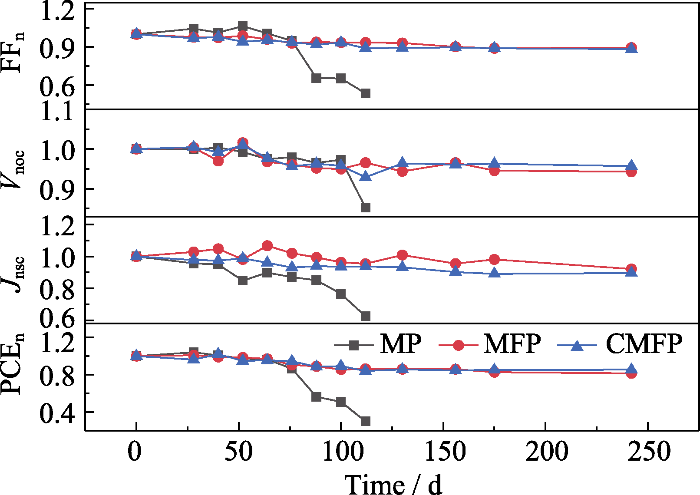
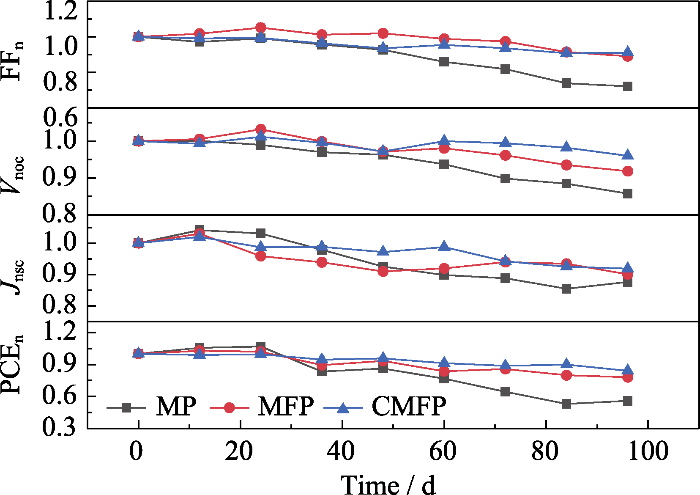
长期稳定性测试结果如图6所示。为了直观观测电池性能, 用相对数值表征各参数的变化, 即后续的测试结果与器件最初相应参数的比值。例如, 将测试过程中得到的Voc与该器件的初始Voc的比值, 记为Vnoc。
图6
图6
三种钙钛矿太阳能电池在(20±5) ℃, 相对湿度<5%, 黑暗条件下的长期稳定性
Fig. 6
Long-term stabilities of three perovskite solar cells at (20±5) ℃, relative humidity <5% in the dark
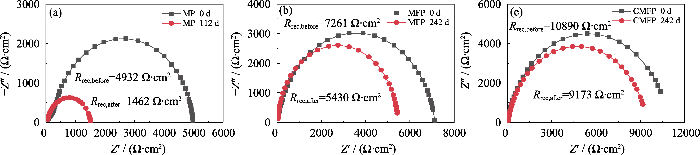
表S6给出了长期稳定性测试结束后各PSCs的J-V相对参数值, MP PSCs放置112 d后PCEn为0.30, MP PSCs的FF和Jsc显著下降, 尤其是FFn为0.54。MFP PSCs放置242 d(5808 h)后, PCEn为0.81, 表明掺入FA+后器件的稳定性可以得到显著改善。CMFP PSCs在测试结束时, PCEn最高, 为0.85, 主要原因是加入Cs+明显延缓了Voc、FF和Jsc衰减。
图7
图7
(a) MP、(b) MFP、(c) CMFP钙钛矿太阳能电池在(20±5) ℃, 相对湿度<5%条件下老化前后的暗态EIS图谱
Fig. 7
Dark state EIS profiles of (a) MP, (b) MFP, (c) CMFP PSCs before and after aging at (20±5) ℃ and relative humidity<5%
图8
图8
三种PSCs在(85±5) ℃, 相对湿度20%~30%, 黑暗条件下的热稳定性
Fig. 8
Thermal stability of three PSCs at (85±5) ℃ and relative humidity 20%-30% in the dark
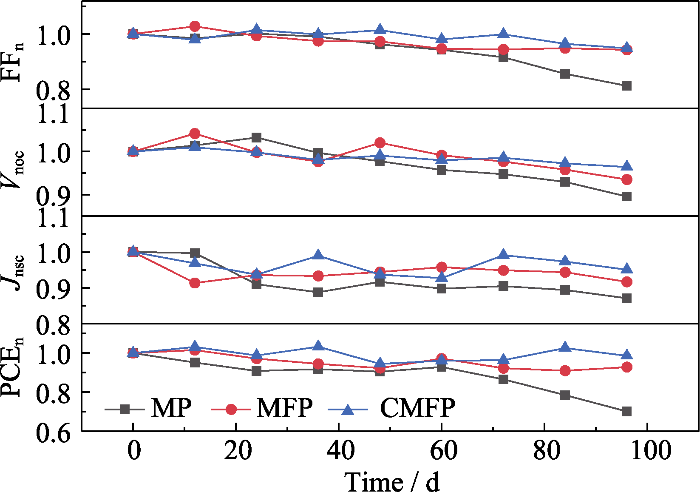
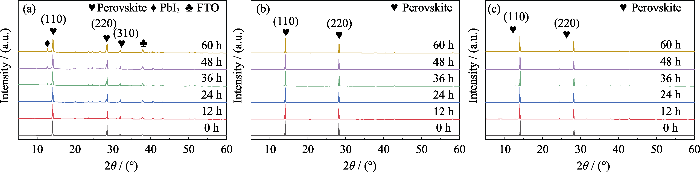
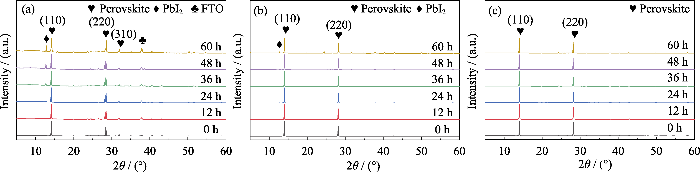
从薄膜的热稳定性测试的归一化XRD变化图谱(图S2(a))中可以看到从24 h开始, MP薄膜开始出现PbI2特征峰(2θ=12.7°), 而且该特征峰的相对强度随加热时间延长而迅速增大, 表明MP膜的受热分解速率较快。然而图S2(b, c)显示, 从加热开始至结束, MFP和CMFP均未在2θ=12.7°处出现明显的特征峰, 表明MFP和CMFP薄膜在整个加热过程中并未发生分解, 这是由于掺杂FA+和Cs+可以显著提高钙钛矿器件的抗热稳定性。
湿度稳定性测试结果如图9所示, 相对数值列于表S8。图9显示, 与热稳定性测试结果相似, 所有PSCs性能均有不同程度的下降。表S8显示, CMFP电池的湿稳定性最佳, 放置96 h后其PCE较原器件仅下降了16%; 而MP和MFP电池的PCE则分别下降了44%和22%。从图S3(a)中可以看出, 放置12 h以后, MP在2θ=12.7°处开始出现PbI2特征衍射峰, 随着在高湿度环境中放置时间延长, 该特征峰的相对强度逐渐增大。到测试结束时其PbI2衍射峰已经非常明显, 薄膜分解现象严重, 表面发黄。图S3(b, c)显示MFP在老化实验60 h时出现了PbI2特征峰, 而从高湿度老化实验开始到结束, CMFP在2θ=12.7°处均未产生PbI2的特征衍射峰。这意味着CMFP薄膜在整个老化过程中均未发生分解, 说明掺杂FA+和Cs+也可以显著提高钙钛矿器件的抗湿稳定性。
图9
图9
三种PSCs在(20±5) ℃, 相对湿度80%~90%, 黑暗条件下的湿稳定性
Fig. 9
Wet stability of three PSCs at (20±5) ℃ and relative humidity 80%-90% in the dark
综上所述, 掺杂FA+和Cs+可以全面提高钙钛矿太阳能电池的抗热、抗湿和长期稳定性。
3 结论
本研究采用两步旋涂法在空气环境中制备了混合阳离子钙钛矿薄膜并组装成电池, 系统研究了A位阳离子掺杂对薄膜微观结构及电池性能的影响。掺杂FA+和Cs+可以增大薄膜晶粒尺寸, 减少晶界, 有利于提高器件的PCE。这是因为掺杂FA+有利于增大薄膜吸光能力, 降低钙钛矿/SnO2的能级势垒, 抑制载流子复合, 从而全面提高电池的Jsc、Voc和FF。而掺杂Cs+有利于降低薄膜的缺陷密度, 提高电子的抽取速率, 抑制非辐射复合, 提高Voc和FF, 使CMFP的最佳PCE为19.34%。此外, 掺杂FA+和Cs+可以增强钙钛矿晶体结构的稳定性, 改善界面接触, 显著提高电池的长期稳定性, CMFP在(20±5) ℃, 相对湿度<5%的黑暗环境中放置超过5808 h(242 d), 效率仍保持初始值的85%。掺杂FA+和Cs+使器件的抗湿和抗热稳定性也得到了全面提高。
补充材料
本文相关补充材料可登录
超长稳定的混合阳离子钙钛矿太阳能电池性能优化研究
马婷婷1,2, 汪志鹏1,2, 张 梅1,2, 郭 敏1,2
(北京科技大学1. 钢铁冶金新技术国家重点实验室; 2. 冶金与生态工程学院, 北京100083)
S1 实验方法
S1.1 实验试剂
碘化铅(PbI2, 99.99%)、碘化甲铵(MAI, 99.90%)、碘化甲脒(FAI, 99.90%)、碘化铯(CsI, 99.90%)、氯化甲铵(MACl, 99.90%)和Spiro-OMeTAD(99.50%), 均购自西安宝莱特光电科技有限公司; N, N-二甲基甲酰胺(DMF, 99.90%)、二甲基亚砜(DMSO, 99.80%)和异丙醇(IPA, 99.80%)购自阿拉丁试剂有限公司; SnO2胶体溶液(质量分数15%)购自阿法埃莎(中国)化工有限公司。FTO导电玻璃(14 Ω·cm-2)购自武汉晶格太阳能科技有限公司; 金颗粒(99.999%)购自中诺新材(北京)科技有限公司。
S1.2 实验表征及测试
在AM 1.5G模拟太阳光的照射下, 采用电化学工作站(CHI660C, CH Instruments, Inc., 美国)测量PSCs的J-V曲线。使用标准硅电池校准光强为 100 mW·cm-2, 以短弧氙灯(CHF-XM-500W, 中国)为光源, 在0~1.2 V范围内, 以100 mV·s-1的扫速线性扫描得到测试结果。电化学阻抗谱(Electrochemical Impedance Spectroscopy, EIS)测试在黑暗中(频率范围:0.1 Hz~0.1 MHz, 0.8 V)进行。采用X射线衍射仪(X-ray Diffraction, XRD, Rigaku Dmax2500, 日本)表征样品的物相结构, 使用Cu Kα1(1.5056 Å)和40 kV高压稳定电源, 扫速为10 (°)/min。使用Supra-55(德国)高分辨率扫描电子显微镜(Scanning Electron Microscope, SEM)研究样品的微观形态, 工作电压为10 kV。外量子效率(External Quantum Efficiency, EQE)测试采用QER-3011 (Enlitech)测试系统测试电池的单色光光电转换效率, 测试波长范围300~900 nm, 步长10 nm。使用紫外-可见分光光度计(TU-1901, Beijing Persee)获得紫外-可见吸收光谱, 扫描范围300~850 nm。采用紫外光电子能谱仪(Thermo Scientifc NEXSA)表征样品的电子结构(Ultraviolet Photoelectron Spectroscopy, UPS), 气压为5×10-9 mbar。使用稳态瞬态荧光光谱仪(FLS980, 爱丁堡)获得稳态光致发光(Photoluminescence, PL)和时间分辨光致发光(Time Resolved Photoluminescence, TRPL)光谱, 激发波长和发射波长分别为470和770 nm(810 nm)。
长期稳定性测试:将三种未封装的电池置于干燥器中((20±5) ℃, 相对湿度<5%), 避光保存, 每隔12 d测试电池的J-V性能。
热稳定测试性:将三种未封装的电池置于85 ℃加热板上加热12 h(空气中), 然后在室温下放置1 h冷却, 为一次循环, 测试电池的J-V曲线。
湿稳定性测试:将三种未封装的电池放在密封容器((20±5) ℃, 80%~90% RH)内, 并置于避光处。每隔12 h, 测试电池的J-V性能。
图S1
图S1
三种钙钛矿薄膜的UPS图谱
Fig. S1
UPS profiles of three perovskite films
(a) UPS full spectra; UPS spectra corresponding to the secondary electron cutoff region for (b) MP, (c) MFP, (d) CMFP;(e) UPS valence band spectra; UPS spectra of the valence band top region with respect to the Femi level for (f) MP, (g) MFP, (h) CMFP
图S2
图S2
在85 ℃, 相对湿度20%~30%条件下, 三种钙钛矿薄膜随时间变化的XRD图谱
Fig. S2
XRD patterns of three perovskite thin films over time at 85 ℃, 20%-30% RH
(a) MP; (b) MFP; (c) CMFP
图S3
图S3
在(20±5) ℃, 相对湿度80%~90%条件下三种钙钛矿薄膜随时间变化的XRD图谱
Fig. S3
XRD patterns of three perovskite thin films over time at (20±5) ℃, 80%-90% RH
(a) MP; (b) MFP; (c) CMFP
表S1
三种性能最佳的PSCs的性能参数(
Table S1
| Sample | Jsc /(mA•cm-2) | Voc/V | FF | PCEmax/% | Intergrated current density/(mA•cm-2) |
|---|---|---|---|---|---|
| MP | 22.87 | 0.97 | 0.74 | 16.34 | 21.87 |
| MFP | 23.48 | 1.05 | 0.76 | 18.66 | 22.71 |
| CMFP | 23.30 | 1.06 | 0.78 | 19.34 | 22.37 |
表S2
从钙钛矿一侧激发瞬态光谱(
Table S2
| Sample | τ1/ns | τ2/ns | B1 | B2 | τmean/ns |
|---|---|---|---|---|---|
| MP | 33.64 | 96.21 | 0.45 | 0.55 | 68.05 |
| MFP | 31.70 | 81.41 | 0.68 | 0.32 | 47.61 |
| CMFP | 26.51 | 77.12 | 0.22 | 0.78 | 65.99 |
表S3 由 UPS图谱(图S1)计算得到的三种钙钛矿薄膜的EVB和ECB
Table S3
| Sample | Ф/eV | EVB/eV | Eg/eV | ECB/eV | |
|---|---|---|---|---|---|
| MP | 3.74 | 1.59 | -5.33 | 1.61 | -3.72 |
| MFP | 4.02 | 1.49 | -5.51 | 1.55 | -3.96 |
| CMFP | 3.93 | 1.47 | -5.40 | 1.55 | -3.85 |
表S4
从FTO一侧激发瞬态光谱(
Table S4
| Sample | ||||
|---|---|---|---|---|
| MP | 75.22 | 422.18 | 0.47 | 0.53 |
| MFP | 35.4 | 21.42 | 0.38 | 0.62 |
| CMFP | 8.21 | 34.52 | 0.76 | 0.24 |
表S5
EIS(
Table S5
| Long-term stability | Rs/(Ω·cm2) | Rrec/(Ω·cm2) | Rnrec | |
|---|---|---|---|---|
| Before testing | MP | 32.40 | 4932 | — |
| MFP | 27.18 | 7261 | — | |
| CMFP | 21.84 | 10890 | — | |
| After testing | MP | 47.99 | 1462 | 0.30 |
| MFP | 39.88 | 5430 | 0.75 | |
| CMFP | 34.16 | 9173 | 0.84 | |
表S6 三种PSCs经长期稳定性测试后的J-V相对参数值
Table S6
| Sample | FFn | Vnoc | Jnsc | PCEn |
|---|---|---|---|---|
| MP | 0.54 | 0.85 | 0.63 | 0.30 |
| MFP | 0.90 | 0.94 | 0.92 | 0.81 |
| CMFP | 0.88 | 0.96 | 0.90 | 0.85 |
表S7 三种PSCs经热稳定性测试后的J-V相对数值
Table S7
| Sample | FFn | Vnoc | Jnsc | PCEn |
|---|---|---|---|---|
| MP | 0.81 | 0.89 | 0.87 | 0.70 |
| MFP | 0.94 | 0.94 | 0.92 | 0.93 |
| CMFP | 0.95 | 0.96 | 0.95 | 0.99 |
表S8 三种PSCs经湿稳定性测试后的J-V相对数值
Table S8
| Sample | FFn | Vnoc | Jnsc | PCEn |
|---|---|---|---|---|
| MP | 0.72 | 0.86 | 0.88 | 0.56 |
| MFP | 0.89 | 0.92 | 0.90 | 0.78 |
| CMFP | 0.91 | 0.96 | 0.92 | 0.84 |
参考文献
Emerging renewable and sustainable energy technologies: state of the art
Organometal halide perovskites as visible-light sensitizers for photovoltaic cells
Two organolead halide perovskite nanocrystals, CH(3)NH(3)PbBr(3) and CH(3)NH(3)PbI(3), were found to efficiently sensitize TiO(2) for visible-light conversion in photoelectrochemical cells. When self-assembled on mesoporous TiO(2) films, the nanocrystalline perovskites exhibit strong band-gap absorptions as semiconductors. The CH(3)NH(3)PbI(3)-based photocell with spectral sensitivity of up to 800 nm yielded a solar energy conversion efficiency of 3.8%. The CH(3)NH(3)PbBr(3)-based cell showed a high photovoltage of 0.96 V with an external quantum conversion efficiency of 65%.
A hole-conductor-free, fully printable mesoscopic perovskite solar cell with high stability
We fabricated a perovskite solar cell that uses a double layer of mesoporous TiO2 and ZrO2 as a scaffold infiltrated with perovskite and does not require a hole-conducting layer. The perovskite was produced by drop-casting a solution of PbI2, methylammonium (MA) iodide, and 5-ammoniumvaleric acid (5-AVA) iodide through a porous carbon film. The 5-AVA templating created mixed-cation perovskite (5-AVA)x(MA)1- xPbI3 crystals with lower defect concentration and better pore filling as well as more complete contact with the TiO2 scaffold, resulting in a longer exciton lifetime and a higher quantum yield for photoinduced charge separation as compared to MAPbI3. The cell achieved a certified power conversion efficiency of 12.8% and was stable for >1000 hours in ambient air under full sunlight. Copyright © 2014, American Association for the Advancement of Science.
Opportunities and challenges for tandem solar cells using metal halide perovskite semiconductors
High-efficiency perovskite solar cells
With rapid progress in a power conversion efficiency (PCE) to reach 25%, metal halide perovskite-based solar cells became a game-changer in a photovoltaic performance race. Triggered by the development of the solid-state perovskite solar cell in 2012, intense follow-up research works on structure design, materials chemistry, process engineering, and device physics have contributed to the revolutionary evolution of the solid-state perovskite solar cell to be a strong candidate for a next-generation solar energy harvester. The high efficiency in combination with the low cost of materials and processes are the selling points of this cell over commercial silicon or other organic and inorganic solar cells. The characteristic features of perovskite materials may enable further advancement of the PCE beyond those afforded by the silicon solar cells, toward the Shockley-Queisser limit. This review summarizes the fundamentals behind the optoelectronic properties of perovskite materials, as well as the important approaches to fabricating high-efficiency perovskite solar cells. Furthermore, possible next-generation strategies for enhancing the PCE over the Shockley-Queisser limit are discussed.
Metal oxide electron transport materials for perovskite solar cells: a review
A review: thermal stability of methylammonium lead halide based perovskite solar cells
Perovskite solar cells have achieved photo-conversion efficiencies greater than 20%, making them a promising candidate as an emerging solar cell technology. While perovskite solar cells are expected to eventually compete with existing silicon-based solar cells on the market, their long-term stability has become a major bottleneck. In particular, perovskite films are found to be very sensitive to external factors such as air, UV light, light soaking, thermal stress and others. Among these stressors, light, oxygen and moisture-induced degradation can be slowed by integrating barrier or interface layers within the device architecture. However, the most representative perovskite absorber material, CH3NH3PbI3 (MAPbI3), appears to be thermally unstable even in an inert environment. This poses a substantial challenge for solar cell applications because device temperatures can be over 45 °C higher than ambient temperatures when operating under direct sunlight. Herein, recent advances in resolving thermal stability problems are highlighted through literature review. Moreover, the most recent and promising strategies for overcoming thermal degradation are also summarized.
Effects of A site doping on the crystallization of perovskite films
Cation engineering on lead iodide perovskites for stable and high-performance photovoltaic applications
Perovskite solar cells (PSCs) based on methylammonium lead iodide (CH<sub>3</sub>NH<sub>3</sub>PbI<sub>3</sub>) have shown unprecedentedly outstanding performance in the recent years. Nevertheless, due to the weak interaction between polar CH<sub>3</sub>NH<sub>3</sub><sup>+</sup> (MA<sup>+</sup>) and inorganic PbI<sub>3</sub><sup>-</sup> sublattices, CH<sub>3</sub>NH<sub>3</sub>PbI<sub>3</sub> dramatically suffers from poor moisture stability, thermal decomposition and device hysteresis. As such, strong electrostatic interactions between cations and anionic frameworks are desired for synergistic improvements of the abovementioned issues. While replacements of I<sup>-</sup> with Br<sup>-</sup> and/or Cl<sup>-</sup> evidently widen optical bandgaps of perovskite materials, compositional modifications can solely be applied on cation components in order to preserve the broad absorption of solar spectrum. Herein, we review the current successful practices in achieving efficient, stable and minimally hysteretic PSCs with lead iodide perovskite systems that employ photoactive cesium lead iodide (CsPbI<sub>3</sub>), formamidinium lead iodide (HC(NH<sub>2</sub>)<sub>2</sub>PbI<sub>3</sub>, or FAPbI<sub>3</sub>), MA<sub>1-<em>x</em>-<em>y</em>-<em>z</em></sub>FA<sub><em>x</em></sub>Cs<sub><em>y</em></sub>Rb<sub><em>z</em></sub>PbI<sub>3</sub> mixed-cation settings as well as two-dimensional butylammonium (C<sub>4</sub>H<sub>9</sub>NH<sub>3</sub><sup>+</sup>, or BA<sup>+</sup>)/MA<sup>+</sup>, polymeric ammonium (PEI<sup>+</sup>)/MA<sup>+</sup> co-cation layered structures. Fundamental aspects behind the stabilization of perovskite phases <em>α</em>-CsPbI<sub>3</sub>, <em>α</em>-FAPbI<sub>3</sub>, mixed-cation MA<sub>1-<em>x</em>-<em>y</em>-<em>z</em></sub>FA<sub><em>x</em></sub>Cs<sub><em>y</em></sub>Rb<sub><em>z</em></sub>PbI<sub>3</sub> and crystallographic alignment of (BA)<sub>2</sub>(MA)<sub>3</sub>Pb<sub>4</sub>I<sub>13</sub> for effective light absorption and charge transport will be discussed. This review will contribute to the continuous development of photovoltaic technology based on PSCs.
Halide segregation in mixed-halide perovskites: influence of A-site cations
Mixed-halide perovskites offer bandgap tunability essential for multijunction solar cells; however, a detrimental halide segregation under light is often observed. Here we combine simultaneous in situ photoluminescence and X-ray diffraction measurements to demonstrate clear differences in compositional and optoelectronic changes associated with halide segregation in MAPb(BrI) and FACsPb(BrI) films. We report evidence for low-barrier ionic pathways in MAPb(BrI), which allow for the rearrangement of halide ions in localized volumes of perovskite without significant compositional changes to the bulk material. In contrast, FACsPb(BrI) lacks such low-barrier ionic pathways and is, consequently, more stable against halide segregation. However, under prolonged illumination, it exhibits a considerable ionic rearrangement throughout the bulk material, which may be triggered by an initial demixing of A-site cations, altering the composition of the bulk perovskite and reducing its stability against halide segregation. Our work elucidates links between composition, ionic pathways, and halide segregation, and it facilitates the future engineering of phase-stable mixed-halide perovskites.© 2021 The Authors. Published by American Chemical Society.
Research progress of perovskite solar cells
Recent progress in high- quality perovskite CH3NH3PbI3 single crystal
Phase behaviour and composition in the formamidinium-methylammonium hybrid lead iodide perovskite solid solution
Halide perovskite materials for solar cells: a theoretical review
Intrinsic thermal instability of methylammonium lead trihalide perovskite
Vapor-assisted deposition of highly efficient, stable black-phase FAPbI3 perovskite solar cells
Formamidinium lead trihalide: a broadly tunable perovskite for efficient planar heterojunction solar cells
Efficient (>20%) and stable all- inorganic cesium lead triiodide solar cell enabled by thiocyanate molten salts
Mixed-organic-cation perovskite photovoltaics for enhanced solar-light harvesting
Efficient perovskite solar cells via improved carrier management
Cesium-containing triple cation perovskite solar cells: improved stability, reproducibility and high efficiency
Insight into the role of guanidinium and cesium in triple cation lead halide perovskites
Effcient and stable TiO2 nanorod array structured perovskite solar cells in air: co-passivation and synergistic mechanism
High-efficiency perovskite solar cells based on the black polymorph of HC(NH2)2PbI3
Formamidinium-containing metal-halide: an alternative material for near-IR absorption perovskite solar cells
Nb2O5 coating on the performance of flexible dye sensitized solar cell based on TiO2 nanoarrays/upconversion luminescence composite structure
Light-induced redox reactions in nanocrystalline systems