

高强度羟基磷灰石纳米陶瓷的构建及其促成骨细胞活性研究
收稿日期: 2020-07-14
修回日期: 2020-08-08
网络出版日期: 2020-08-15
Construction of Hydroxyapatite Nanoceramics with High Mechanical Strength and Efficiency in Promoting the Spreading and Viability of Osteoblasts
Received date: 2020-07-14
Revised date: 2020-08-08
Online published: 2020-08-15
Supported by
National Key Research and Development Program of China(2016YFC1102000);National Key Research and Development Program of China(2016YFC1102003);Sichuan Science and Technology Innovation Team of China(2019JDTD0008)
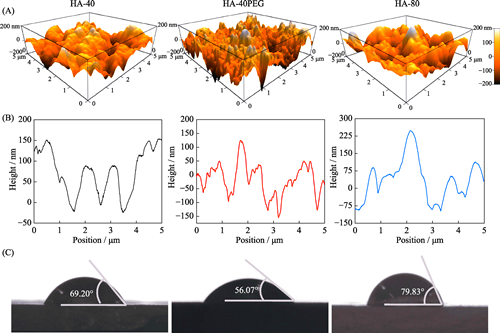
本研究旨在研究羟基磷灰石(HA)前驱粉体与所制备陶瓷之间的关系, 制备具有优良力学性能及成骨活性的HA纳米陶瓷。采用三种HA前驱粉体, 即40 ℃合成的HA-40粉体、以PEG为模板40 ℃合成的HA-40PEG粉体和80 ℃合成的HA-80粉体, 系统研究了前驱粉体对陶瓷性能的影响。结果显示, HA-40、HA-40PEG和HA-80粉体制备的陶瓷晶粒尺寸分别为(217.87±57.53)、(123.22±20.16)和(316.65±68.91) nm, 表明HA-40PEG有利于HA纳米陶瓷的制备。烧结得到的HA-40PEG纳米晶陶瓷表现出良好的力学性能, 与另外两种亚微米晶陶瓷(HA-40和HA-80)相比, 其抗压强度更高(~300 MPa)。细胞研究结果显示, HA-40PEG比HA-40和HA-80更能促进MC3T3-E1前成骨细胞的铺展和增殖。由此可知, 前驱粉体合成是影响HA陶瓷性能的关键因素, 纳米晶构建有利于同时提高其力学性能和生物学性能。
吴永豪 , 李向锋 , 朱向东 , 张兴栋 . 高强度羟基磷灰石纳米陶瓷的构建及其促成骨细胞活性研究[J]. 无机材料学报, 2021 , 36(5) : 552 -560 . DOI: 10.15541/jim20200395
The present study focuses on the construction of HA nanoceramics with excellent mechanical property and osteogenic activity, and the correlation between HA precursor powders and resulting ceramics. Three precursor powders were used, i.e. HA-40 synthesized at 40 ℃, HA-40PEG synthesized at 40 ℃ with PEG as a template, and HA-80 synthesized at 80 ℃. The results showed that grain sizes of three HA ceramics prepared by HA-40, HA-40PEG, and HA-80 precursors were (217.87±57.53), (123.22±20.16), and (316.65±68.91) nm, respectively. It demonstrated that compared with HA-40 and HA-80, HA-40PEG was more beneficial for fabricating HA nanoceramics. Among three resulting ceramics, HA-40PEG displayed the highest comprehensive strength (~300 MPa). In addition, the nano-scale HA-40PEG ceramics promoted better cell spreading and proliferation than those of submicro-scale HA-40 and HA-80 ceramics. These findings suggest that the preparation of HA precursor powders plays an important role in fabricating HA nanoceramics with simultaneous improvement of mechanical and biological properties.

Key words: hydroxyapatite; nanoceramics; initial powder; density; cellular viability
| [1] | HONG Y, FAN H, LI B, et al. Fabrication, biological effects, and medical applications of calcium phosphate nanoceramics. Materials Science & Engineering R, 2010,70(3):225-242. |
| [2] | DOROZHKIN S V. Calcium orthophosphates in nature. Biology and Medicine Materials, 2009,2(1):399-498. |
| [3] | NASIRI-TABRIZI B, HONARMANDI P, EBRAHIMI-KAHRIZSANGI R, et al. Synthesis of nanosize single-crystal hydroxyapatite via mechanochemical method. Materials Letters, 2009,63(5):543-546. |
| [4] | WANG J, SHAW L L. Nanocrystalline hydroxyapatite with simultaneous enhancements in hardness and toughness. Biomaterials, 2009,30(34):6565-6572. |
| [5] | BOSE S, DASGUPTA S, TARAFDER S, et al. Microwave-processed nanocrystalline hydroxyapatite: simultaneous enhancement of mechanical and biological properties. Acta Biomater., 2010,6(9):3782-3790. |
| [6] | FANG Z, FENG Q, TAN R. In-situ grown hydroxyapatite whiskers reinforced porous HA bioceramic. Ceramics International, 2013,39(8):8847-8852. |
| [7] | PROKOPIEV O, SEVOSTIANOV I. Dependence of the mechanical properties of sintered hydroxyapatite on the sintering temperature. Materials Science & Engineering A, 2006,431(1):218-227. |
| [8] | HABIBOVIC P, YUAN H, VAN DER VALK C M, et al. 3D microenvironment as essential element for osteoinduction by biomaterials. Biomaterials, 2005,26(17):3565-3575. |
| [9] | KIM H M, HIMENO T, KOKUBO T, et al. Process and kinetics of bonelike apatite formation on sintered hydroxyapatite in a simulated body fluid. Biomaterials, 2005,26(21):4366-4373. |
| [10] | RICE R W, WU C C, BOICHELT F. Hardness-grain-size relations in ceramics. Journal of the American Ceramic Society, 1994,77(10):2539-2553. |
| [11] | MOSHTAGHIOUN B M, GOMEZ-GARCIA D, DOMINGUEZ- RODRIGUEZ A, et al. Grain size dependence of hardness and fracture toughness in pure near fully-dense boron carbide ceramics. Journal of the European Ceramic Society, 2016,36(7):1829-1834. |
| [12] | GU Y W, LOH N H, KHOR K A, et al. Spark plasma sintering of hydroxyapatite powders. Biomaterials, 2002,23(1):37-43. |
| [13] | KIM B N, PRAJATELISTIA E, HAN Y H, et al. Transparent hydroxyapatite ceramics consolidated by spark plasma sintering. Scripta Materialia, 2013,69(5):366-369. |
| [14] | GUO X, XIAO P, JING L, et al. Fabrication of nanostructured hydroxyapatite via hydrothermal synthesis and spark plasma sintering. Journal of the American Ceramic Society, 2005,88(4):1026-1029. |
| [15] | RAMESH S, TAN C Y, BHADURI S B, et al. Rapid densification of nanocrystalline hydroxyapatite for biomedical applications. Ceramics International, 2007,33(7):1363-1367. |
| [16] | VELJOVIC D, JOKIC B, PETROVI? R, et al. Processing of dense nanostructured HAP ceramics by sintering and hot pressing. Ceramics International, 2009,35:1407-1413. |
| [17] | WANG J, SHAW L L. Transparent nanocrystalline hydroxyapatite by pressure-assisted sintering. Scripta Materialia, 2010,63(6):593-596. |
| [18] | CHEN I W, WANG X H. Sintering dense nanocrystalline ceramics without final-stage grain growth. Nature, 2000,404(6774):168-171. |
| [19] | LIN K, CHEN L, CHANG J. Fabrication of dense hydroxyapatite nanobioceramics with enhanced mechanical properties via two-step sintering process. International Journal of Applied Ceramic Technology, 2012,9(3):479-485. |
| [20] | LUKI? M J, ?KAPIN S D, MARKOVI? S, et al. Processing route to fully dense nanostructured HAp bioceramics: from powder synthesis to sintering. Journal of the American Ceramic Society, 2012,95(11):3394-3402. |
| [21] | THUAULT A, SAVARY E, HORNEZ J C, et al. Improvement of the hydroxyapatite mechanical properties by direct microwave sintering in single mode cavity. Journal of the European Ceramic Society, 2014,34(7):1865-1871. |
| [22] | LI X, SONG T, CHEN X, et al. Osteoinductivity of porous biphasic calcium phosphate ceramic spheres with nanocrystalline and their efficacy in guiding bone regeneration. ACS Applied Materials & Interfaces, 2019,11(4):3722-3736. |
| [23] | LIU D, WU Y, WU H, et al. Effect of process parameters on the microstructure and property of hydroxyapatite precursor powders and resultant sintered bodies. International Journal of Applied Ceramic Technology, 2018,16(2):444-454. |
| [24] | SONG J, YONG L, YING Z, et al. Mechanical properties of hydroxyapatite ceramics sintered from powders with different morphologies. Materials Science & Engineering A, 2011,528(16/17):5421-5427. |
| [25] | LANDI E, TAMPIERI A, CELOTTI G, et al. Densification behaviour and mechanisms of synthetic hydroxyapatites. Journal of the European Ceramic Society, 2000,20(14):2377-2387. |
| [26] | WEINER S, BAR-YOSEF O. States of preservation of bones from prehistoric sites in the Near East: a survey. Journal of Archaeological Science, 1990,17(2):187-196. |
| [27] | FARLAY D, PANCZER G, REY C, et al. Mineral maturity and crystallinity index are distinct characteristics of bone mineral. Journal of Bone and Mineral Metabolism, 2010,28:433-445. |
| [28] | LI X, DENG Y, WANG M, et al. Stabilization of Ca-deficient hydroxyapatite in biphasic calcium phosphate ceramics by adding alginate to enhance their biological performances. Journal of Materials Chemistry B, 2017,6(1):84-97. |
| [29] | MAZAHERI M, HAGHIGHATZADEH M, ZAHEDI A M, et al. Effect of a novel sintering process on mechanical properties of hydroxyapatite ceramics. Journal of Alloys & Compounds, 2009,471(1):180-184. |
| [30] | DASGUPTA S, TARAFDER S, BANDYOPADHYAY A, et al. Effect of grain size on mechanical, surface and biological properties of microwave sintered hydroxyapatite. Materials Science & Engineering C Materials for Biological Applications, 2013,33(5):2846-2854. |
| [31] | PANG Y X, BAO X. Influence of temperature, ripening time and calcination on the morphology and crystallinity of hydroxyapatite nanoparticles. Journal of the European Ceramic Society, 2003,23(10):1697-1704. |
| [32] | KUMAR R, PRAKASH K, CHEANG P, et al. Temperature driven morphological changes of chemically precipitated hydroxyapatite nanoparticles. Langmuir, 2004,20:5196-5200. |
| [33] | TSENG Y H, KUO C S, LI Y Y, et al. Polymer-assisted synthesis of hydroxyapatite nanoparticle. Materials Science and Engineering: C, 2009,29(3):819-822. |
| [34] | LI H, XUE F, WAN X, et al. Polyethylene glycol-assisted preparation of beta-tricalcium phosphate by direct precipitation method. Powder Technology, 2016,301:255-260. |
| [35] | AKAO M, AOKI H, KATO K. Mechanical properties of sintered hydroxyapatite for prosthetic applications. Journal of Materials Science, 1981,16:809-812. |
| [36] | ARIFVIANTO B, MAHARDIKA M, DEWO P, et al. Effect of surface mechanical attrition treatment (SMAT) on microhardness, surface roughness and wettability of AISI 316L. Materials Chemistry and Physics, 2011,125(3):418-426. |
| [37] | DOS SANTOS E, FARINA M, SOARES G, et al. Surface energy of hydroxyapatite and β-tricalcium phosphate ceramics driving serum protein adsorption and osteoblast adhesion. Journal of Materials Science: Materials in Medicine, 2008,19(6):2307-2316. |
| [38] | LI B, CHEN X, GUO B, et al. Fabrication and cellular biocompatibility of porous carbonated biphasic calcium phosphate ceramics with a nanostructure. Acta Biomaterialia, 2009,5(1):134-143. |
| [39] | GUO X, GOUGH J E, XIAO P, et al. Fabrication of nanostructured hydroxyapatite and analysis of human osteoblastic cellular response. Journal of Biomedical Materials Research Part A, 2007,82(4):1022-1032. |
| [40] | MICHIARDI A, APARICIO C, RATNER B D, et al. The influence of surface energy on competitive protein adsorption on oxidized NiTi surfaces. Biomaterials, 2007,28(4):586-594. |
| [41] | YAO C, PERLA V, MCKENZIE J L, et al. Anodized Ti and Ti6Al4V possessing nanometer surface features enhances osteoblast adhesion. Journal of Biomedical Nanotechnology, 2005,1(1):68-73. |
| [42] | WEBSTER T J, SIEGEL R W, BIZIOS R. Osteoblast adhesion on nanophase ceramics. Biomaterials, 1999,20(13):1221-1227. |
/
| 〈 |
|
〉 |