无机材料学报 ›› 2026, Vol. 41 ›› Issue (3): 289-294.DOI: 10.15541/jim20250173 CSTR: 32189.14.jim20250173
黄应贺1,2( ), 黄仁兴1, 石宇星1, 雷一杰2, 于涛1, 王诚2, 顾军1(
), 黄仁兴1, 石宇星1, 雷一杰2, 于涛1, 王诚2, 顾军1( )
)
收稿日期:2025-04-24
修回日期:2025-08-10
出版日期:2026-03-20
网络出版日期:2025-08-26
通讯作者:
顾 军, 教授. E-mail: junguca@nju.edu.cn作者简介:黄应贺(1996-), 男, 博士研究生. E-mail: 602023220030@smail.nju.edu.cn
基金资助:
HUANG Yinghe1,2( ), HUANG Renxing1, SHI Yuxing1, LEI Yijie2, YU Tao1, WANG Cheng2, GU Jun1(
), HUANG Renxing1, SHI Yuxing1, LEI Yijie2, YU Tao1, WANG Cheng2, GU Jun1( )
)
Received:2025-04-24
Revised:2025-08-10
Published:2026-03-20
Online:2025-08-26
Contact:
GU Jun, professor. E-mail: junguca@nju.edu.cnAbout author:HUANG Yinghe (1996-), male, PhD candidate. E-mail: 602023220030@smail.nju.edu.cn
Supported by:1. 利用介孔碳的限域效应提升氧还原反应中Pt催化剂的耐久性.mp4(2360KB)
摘要:
铂碳(Pt/C)催化剂是质子交换膜燃料电池(PEMFCs)最具前景的阴极催化剂之一, 但其耐久性不佳。这主要是由于铂纳米颗粒(Pt NPs)会发生团聚和迁移, Pt颗粒尺寸变大, 从而使催化活性丧失。本研究通过构建具有串珠状内部孔隙结构的介孔碳载体(IPMC), 研究了Pt NPs在碳载体中的沉积位置, 并将其精准限域在孔道内部, 利用独特的孔隙结构, 实现了Pt NPs的高效限域, 提升了材料稳定性。分析表明, 介孔碳催化剂中的Pt NPs经过30000 圈加速耐久性测试(ADT)循环后平均粒径仅增加了0.46 nm, 而实心碳催化剂中的Pt NPs增加了0.79 nm。介孔碳催化剂的电化学性能损失较小, 电化学活性面积(ECSA)损失率为24.18%, 显著低于实心碳催化剂(32.33%)。介孔碳催化剂耐久性优异主要归因于IPMC独特的孔隙结构所产生的限域效应, 进而抑制内部Pt NPs的奥斯特瓦尔德(Ostwald)熟化和迁移行为, 显著延缓了氧还原反应(ORR)催化活性衰减。本工作揭示了串珠状介孔对Pt NPs的物理限域机制, 即相互连通且具有局部收缩的孔道结构形成空间屏障, 有效阻碍溶解Pt物种的扩散迁移并锚定颗粒位置, 为设计PEMFCs的最佳碳载体提供了更精确的结构蓝图。
中图分类号:
黄应贺, 黄仁兴, 石宇星, 雷一杰, 于涛, 王诚, 顾军. 利用介孔碳的限域效应提升氧还原反应中Pt催化剂的耐久性[J]. 无机材料学报, 2026, 41(3): 289-294.
HUANG Yinghe, HUANG Renxing, SHI Yuxing, LEI Yijie, YU Tao, WANG Cheng, GU Jun. Enhancing Durability of Pt Catalysts in the Oxygen Reduction Reaction by Confinement Effect of Mesoporous Carbon[J]. Journal of Inorganic Materials, 2026, 41(3): 289-294.
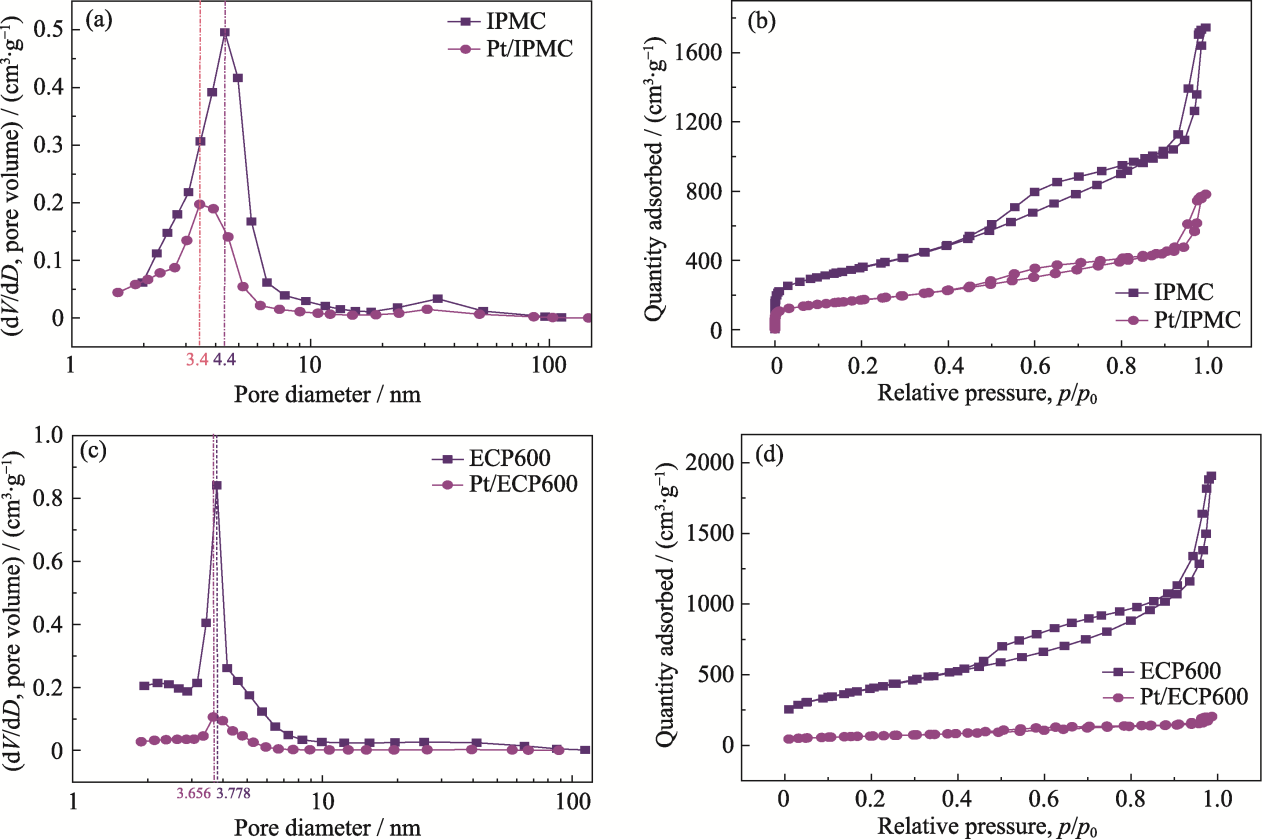
图3 IPMC和Pt/IPMC (a, b), ECP600和Pt/ECP600 (c, d)的孔径变化(a, c)和N2吸附-解吸等温线变化(b, d)
Fig. 3 Pore size changes (a, c) and N2 adsorption-desorption isotherm changes (b, d) of IPMC and Pt/IPMC (a, b), ECP600 and Pt/ECP600 (c, d)
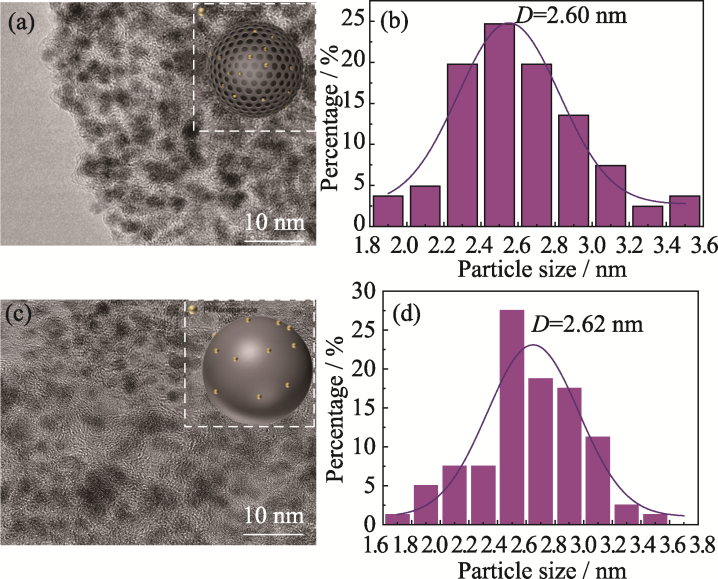
图4 Pt/IPMC (a, b)和Pt/ECP600 (c, d)的TEM照片(a, c)和Pt粒径分布(b, d)
Fig. 4 TEM images (a, c) and Pt particle size distributions (b, d) of Pt/IPMC (a, b) and Pt/ECP600 (c, d)
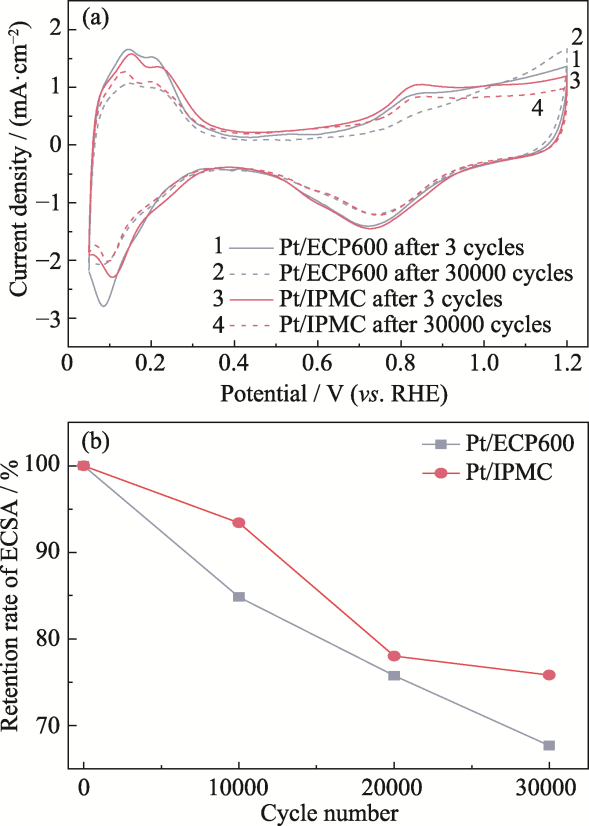
图5 (a) ADT前后Pt/IPMC和Pt/ECP600的CV曲线; (b) 10000、20000和30000圈ADT后的ECSA保留率
Fig. 5 (a) CV curves of Pt/IPMC and Pt/ECP600 before and after ADT cycles; (b) ECSA retention rates after 10000, 20000, and 30000 ADT cycles
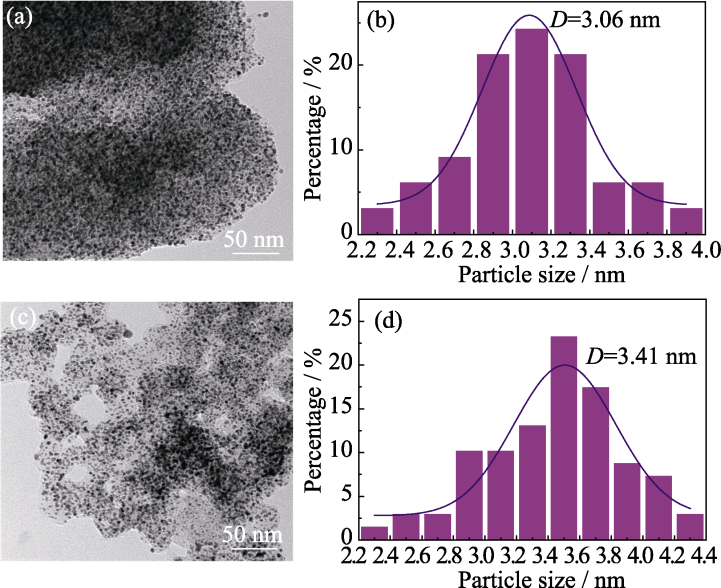
图6 Pt/IPMC (a, b)和Pt/ECP600 (c, d)经过30000圈ADT后的TEM照片(a, c)和粒径分布图(b, d)
Fig. 6 TEM images (a, c) and particle size distributions (b, d) of Pt/IPMC (a, b) and Pt/ECP600 (c, d) after 30000 ADT cycles
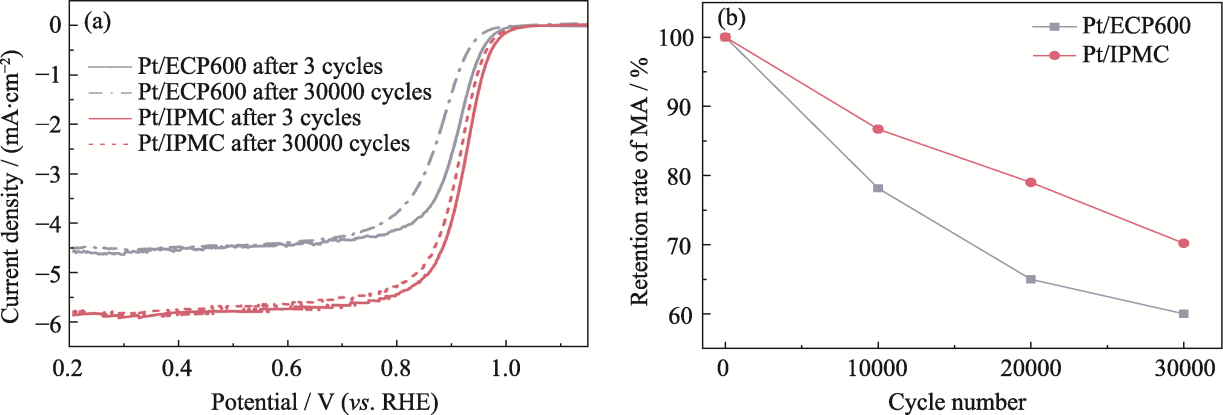
图S5 (a) ADT循环前后Pt/IPMC和Pt/ECP600的LSV曲线; (b) 10000、20000和30000圈ADT循环后的MA保留率
Fig. S5 (a) LSV curves of Pt/IPMC and Pt/ECP600 before and after ADT cycles; (b) MA retention rate after 10000, 20000 and 30000 ADT cycles
| Sample | MA after 3 cycles/(mA∙mg-1) | MA after 10000 cycles/(mA∙mg-1) | MA after 20000 cycles/(mA∙mg-1) | MA after 30000 cycles/(mA∙mg-1) | MA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 144 | 113 | 93.6 | 86.0 | 60.0 |
| Pt/IPMC | 269 | 233 | 213 | 189 | 70.2 |
表S1 Pt/IPMC和市售Pt/ECP600在耐久性能测试中MA的变化
Table S1 Changes in MA during durability test of Pt/IPMC and commercial Pt/ECP600
| Sample | MA after 3 cycles/(mA∙mg-1) | MA after 10000 cycles/(mA∙mg-1) | MA after 20000 cycles/(mA∙mg-1) | MA after 30000 cycles/(mA∙mg-1) | MA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 144 | 113 | 93.6 | 86.0 | 60.0 |
| Pt/IPMC | 269 | 233 | 213 | 189 | 70.2 |
| Sample | SA after 3 cycles/(µA∙cm-2) | SA after 10000 cycles/(µA∙cm-2) | SA after 20000 cycles/(µA∙cm-2) | SA after 30000 cycles/(µA∙cm-2) | SA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 145 | 134 | 125 | 128 | 88.3 |
| Pt/IPMC | 296 | 271 | 295 | 273 | 92.2 |
表S2 Pt/IPMC和市售Pt/ECP600在耐久性能测试中SA的变化
Table S2 Changes in SA during durability test of Pt/IPMC and commercial Pt/ECP600
| Sample | SA after 3 cycles/(µA∙cm-2) | SA after 10000 cycles/(µA∙cm-2) | SA after 20000 cycles/(µA∙cm-2) | SA after 30000 cycles/(µA∙cm-2) | SA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 145 | 134 | 125 | 128 | 88.3 |
| Pt/IPMC | 296 | 271 | 295 | 273 | 92.2 |
| Sample | ECSA after 3 cycles/(m2∙g-1) | ECSA after 10000 cycles/(m2∙g-1) | ECSA after 20000 cycles/(m2∙g-1) | ECSA after 30000 cycles/(m2∙g-1) | ECSA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 99 | 84 | 75 | 67 | 67.67 |
| Pt/IPMC | 91 | 86 | 72 | 69 | 75.82 |
表S3 Pt/IPMC和市售Pt/ECP600在耐久性能测试中ECSA的变化
Table S3 Changes in ECSA during durability test of Pt/IPMC and commercial Pt/ECP600
| Sample | ECSA after 3 cycles/(m2∙g-1) | ECSA after 10000 cycles/(m2∙g-1) | ECSA after 20000 cycles/(m2∙g-1) | ECSA after 30000 cycles/(m2∙g-1) | ECSA retention rate/% |
|---|---|---|---|---|---|
| Pt/ECP600 | 99 | 84 | 75 | 67 | 67.67 |
| Pt/IPMC | 91 | 86 | 72 | 69 | 75.82 |
| Sample | ECSA/(m2∙g-1, initial) | ECSA retention rate | Pt particle size change/nm | Testing environment |
|---|---|---|---|---|
| Pt/ECP600 | 99 | 67.67% (after 30000 cycles) | 2.62→3.41 | 0.1 mol·L-1 HClO4, 0.6-0.1 V, 100 mV·s-1 |
| Pt/IPMC | 91 | 75.82% (after 30000 cycles) | 2.60→3.06 | |
| Pt/C[S2] | 58 | ~47.5% (after 6000 cycles) | - | 0.1 mol·L-1 HClO4, 0.6-1.0 V, 50 mV·s-1 |
| Pt/HCSs[S2] | 63 | ~58.6% (after 6000 cycles) | - | |
| Pt/Vulcan[S3] | - | (after 3600 cycles) | ~3.48→~4.00 | 0.1 mol·L-1 HClO4, 0.4-1.4 V, 1 V·s-1 |
| Pt@HGS[S3] | - | ~3.80→~3.75 | ||
| Pt/VC[S4] | 69 | 54% (after 2000 cycles) | - | 0.5 mol·L-1 H2SO4, 0-1.1 V, 20 mV·s-1 |
| Pt@CS[S4] | 81 | 21% (after 2000 cycles) | - |
表S4 本研究与文献中介孔碳催化剂的耐久性对比
Table S4 Comparison of durability of mesoporous carbon catalysts in this study and literatures
| Sample | ECSA/(m2∙g-1, initial) | ECSA retention rate | Pt particle size change/nm | Testing environment |
|---|---|---|---|---|
| Pt/ECP600 | 99 | 67.67% (after 30000 cycles) | 2.62→3.41 | 0.1 mol·L-1 HClO4, 0.6-0.1 V, 100 mV·s-1 |
| Pt/IPMC | 91 | 75.82% (after 30000 cycles) | 2.60→3.06 | |
| Pt/C[S2] | 58 | ~47.5% (after 6000 cycles) | - | 0.1 mol·L-1 HClO4, 0.6-1.0 V, 50 mV·s-1 |
| Pt/HCSs[S2] | 63 | ~58.6% (after 6000 cycles) | - | |
| Pt/Vulcan[S3] | - | (after 3600 cycles) | ~3.48→~4.00 | 0.1 mol·L-1 HClO4, 0.4-1.4 V, 1 V·s-1 |
| Pt@HGS[S3] | - | ~3.80→~3.75 | ||
| Pt/VC[S4] | 69 | 54% (after 2000 cycles) | - | 0.5 mol·L-1 H2SO4, 0-1.1 V, 20 mV·s-1 |
| Pt@CS[S4] | 81 | 21% (after 2000 cycles) | - |
| [1] | STAMBOULI A B. Fuel cells: the expectations for an environmental-friendly and sustainable source of energy. Renewable & Sustainable Energy Reviews, 2011, 15: 4507. |
| [2] | LI J R, LIU M X, LIU X, et al. The recent progress of oxygen reduction electrocatalysts used at fuel cell level. Small Methods, 2024, 8: 2301249. |
| [3] | ZHAO W H, XU G T, DONG W Y, et al. Progress and perspective for in situ studies of oxygen reduction reaction in proton exchange membrane fuel cells. Advanced Science, 2023, 10: 2300550. |
| [4] | LE T D, AHEMAD M J, KIM D S, et al. Synergistic effect of Pt-Ni dual single-atoms and alloy nanoparticles as a high-efficiency electrocatalyst to minimize Pt utilization at cathode in polymer electrolyte membrane fuel cells. Journal of Colloid and Interface Science, 2023, 634: 930. |
| [5] | FISSEHA G, YU Y N, LU S J, et al. A novel core-shell nanostructure of Ti-Au nanocrystal with PtNi alloy skin: enhancing the durability for oxygen reduction reaction. Chinese Journal of Catalysis, 2024, 56: 81 |
| [6] | SHU Q H, ZHANG J, HU B, et al. Rational design of a high-durability pt-based ORR catalyst supported on Mn/N codoped carbon sheets for PEMFCs. Energy & Fuels, 2022, 36: 1707. |
| [7] | MOGHADAMESFAHANI R A, VANKOVA S K, EASTON E B, et al. A hybrid Pt/NbO/CNTs catalyst with high activity and durability for oxygen reduction reaction in PEMFC. Renewable Energy, 2020, 154: 913. |
| [8] | LI M, LIU F, PEI S, et al. Synthesis of platinum nanocrystals dispersed on nitrogen-doped hierarchically porous carbon with enhanced oxygen reduction reaction activity and durability. Nanomaterials, 2023, 13(3): 444. |
| [9] | CHEN L G, ZHAO W, REN Y C, et al. Study on attenuation mechanism and durability improvement of platinum-carbon catalysts for proton exchange membrane fuel cells. Journal of the Electrochemical Society, 2024, 171: 094509. |
| [10] | SONG Z X, WANG B Q, CHENG N C, et al. Atomic layer deposited tantalum oxide to anchor Pt/C for a highly stable catalyst in PEMFCs. Journal of Materials Chemistry A, 2017, 5: 9760. |
| [11] | BOGAR M, YAKOVLEV Y, SANDBECK D J S, et al. Interplay among dealloying, ostwald ripening, and coalescence in PtXNi100-X bimetallic alloys under fuel-cell-related conditions. ACS Catalysis, 2021, 11: 11360. |
| [12] | LI S B, YIN P, XU C, et al. Entropy-driven Ostwald ripening reversal promotes the formation of low-platinum intermetallic fuel cell catalysts. Small, 2024, 20: 2401134. |
| [13] | DONG Y H, ZHANG D, LI D G, et al. Control of Ostwald ripening. Science China-Materials, 2023, 66: 1249. |
| [14] | LI X F, SU F Y, XIE L J, et al. Carbon corrosion induced by surface defects accelerates degradation of platinum/graphene catalysts in oxygen reduction reaction. Small, 2024, 20: 2310940. |
| [15] | GARCIA-CARDONA J L, SIRES I, MAZZUCATO M, et al. On the viability of chitosan-derived mesoporous carbons as supports for PtCu electrocatalysts in PEMFC. Electrochimica Acta, 2023, 442: 141911. |
| [16] | JAVED H, PANI S, ANTONY J, et al. Synthesis of mesoporous carbon spheres via a soft-template route for catalyst supports in PEMFC cathodes. Soft Matter, 2021, 17: 7743. |
| [17] | SHAHGALDI S, HAMELIN J. Improved carbon nanostructures as a novel catalyst support in the cathode side of PEMFC: a critical review. Carbon, 2015, 94: 705. |
| [18] | CAO L J, ZHU H W, LI Y D, et al. Anchored and confined Pt nanoparticles in radial mesoporous hollow carbon spheres enhancing oxygen reduction reaction stability. New Journal of Chemistry, 2023, 47: 10866. |
| [19] | GALEANO C, MEIER J C, PEINECKE V, et al. Toward highly stable electrocatalysts via nanoparticle pore confinement. Journal of the American Chemical Society, 2012, 134: 20457. |
| [20] | MASHINDI V, MENTE P, PHAAHLAMOHLAKA T N, et al. Platinum nanocatalysts supported on defective hollow carbon spheres: oxygen reduction reaction durability studies. Frontiers in Chemistry, 2022, 10: 839867. |
| [21] | YARLAGADDA V, CARPENTER M K, MOYLAN T E, et al. Boosting fuel cell performance with accessible carbon mesopores. ACS Energy Letters, 2018, 3: 618. |
| [22] | PENG L, PENG H, HUNG C T, et al. Programmable synthesis of radially gradient-structured mesoporous carbon nanospheres with tunable core-shell architectures. Chem, 2021, 7: 1020. |
| [23] | LIU H, CUI W H, SHEN Y F, et al. Hierarchical carbon with microporous core and mesoporous shell for efficient oxygen reduction application. Microporous and Mesoporous Materials, 2024, 368: 113011. |
| [24] | QIAO Z, HWANG S, LI X, et al. 3D porous graphitic nanocarbon for enhancing the performance and durability of Pt catalysts: a balance between graphitization and hierarchical porosity. Energy & Environmental Science, 2019, 12: 2830. |
| [25] | WANG S J, NIU M, ZHENG J, et al. Hollow porous CoP-carbon nanocages for hydrogen evolution reactions. ACS Applied Nano Materials, 2024, 7: 12821. |
| [26] | XIE M, CHU T K, WANG X L, et al. Effect of mesoporous carbon on oxygen reduction reaction activity as cathode catalyst support for proton exchange membrane fuel cell. International Journal of Hydrogen Energy, 2022, 47: 28074. |
| [27] | HUANG L, WEI M, QI R J, et al. An integrated platinum-nanocarbon electrocatalyst for efficient oxygen reduction. Nature Communications, 2022, 13: 6703. |
| [28] | HUANG Y H, GU J, HU Y D, et al. Preparation of mesoporous carbon with adjustable diameter and pore size. Diamond and Related Materials, 2022, 130: 109515. |
| [29] | FERRO G, ROIRON C, WANG H S, et al. Designer electrocatalysts for the oxygen reduction reaction with controlled platinum nanoparticle locality. Advanced Energy Materials, 2024, 15(25): 2403165. |
| [30] | JEONG Y, CUI M C, CHOI J, et al. Development of modified mesoporous carbon (CMK-3) for improved adsorption of bisphenol-A. Chemosphere, 2020, 238: 124559. |
| [31] | ZOU J, FAN C Y, JIANG Y, et al. A preliminary study on assessing the Brunauer-Emmett-Teller analysis for disordered carbonaceous materials. Microporous and Mesoporous Materials, 2021, 327: 111411. |
| [32] | ABBASLOU R M, VOSOUGHI V, DALAI A K. Comparison of nitrogen adsorption and transmission electron microscopy analyses for structural characterization of carbon nanotubes. Applied Surface Science, 2017, 419: 817. |
| [33] | GARSANY Y, BATURINA O A, SWIDER L K E, et al. Experimental methods for quantifying the activity of platinum electrocatalysts for the oxygen reduction reaction. Analytical Chemistry, 2010, 82: 6321. |
| [34] | ZHU S J, HUANG Y H, YU T, et al. Enhance the durability of the oxygen reduction reaction catalyst through the synergy of improved graphitization and nitrogen doping of carbon carrier. International Journal of Hydrogen Energy, 2024, 51: 956. |
| [35] | ZHANG C, FENG Z X, LEI Y J, et al. Batch synthesis of high activity and durability carbon supported platinum catalysts for oxygen reduction reaction using a new facile continuous microwave pipeline technology. Journal of Colloid and Interface Science, 2022, 628: 174. |
| [1] | 杨代辉, 孙甜, 田合鑫, 史晓斐, 马东伟. 铁氮共掺杂介孔碳材料的简易制备及其氧还原反应催化性能[J]. 无机材料学报, 2023, 38(11): 1309-1315. |
| [2] | 高秀丽, 王丹丹, 李硕, 邢伟, 阎子峰. 介孔碳微球的氢醌改性及电容性能研究[J]. 无机材料学报, 2018, 33(1): 48-52. |
| [3] | 徐顺建, 罗玉峰, 李水根, 钟 炜, 黄明道. 低温制备介孔碳对电极构建的染料敏化太阳电池优化研究[J]. 无机材料学报, 2012, 27(1): 83-88. |
| [4] | 贾 巍, 徐茂文, 包淑娟, 贾殿赠. 多孔TiO2的合成及其电化学性能研究[J]. 无机材料学报, 2010, 25(12): 1335-1339. |
| [5] | 徐顺建,乔冠军,王红洁,李涤尘,卢天健. 酚醛树脂聚合相分离热解制备介孔碳[J]. 无机材料学报, 2008, 23(5): 971-974. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||